SPECIAL SECTION: Message For Our “Friends” In The Middle Kingdom
You knuckle-dragging barbarians are still trying to muck with this site, so I’ll just repeat what I said last time.
Up your shit-kicking barbarian asses. Yes, barbarian! It took a bunch of sailors in Western Asia to invent a real alphabet instead of badly drawn cartoons to write with. So much for your “civilization.”
Yeah, the WORLD noticed you had to borrow the Latin alphabet to make Pinyin. Like with every other idea you had to steal from us “Foreign Devils” since you rammed your heads up your asses five centuries ago, you sure managed to bastardize it badly in the process.
Have you stopped eating bats yet? Are you shit-kickers still sleeping with farm animals?
Or maybe even just had the slightest inkling of treating lives as something you don’t just casually dispose of?
中国是个混蛋 !!!
Zhōngguò shì gè hùndàn !!!
China is asshoe !!!
And here’s my response to barbarian “asshoes” like you:
OK, with that rant out of my system…
Loop it if you like; I will wait.
Richly deserved.
Justice Must Be Done
The prior election must be acknowledged as fraudulent, and steps must be taken to prosecute the fraudsters and restore integrity to the system.
Nothing else matters at this point. Talking about trying again in 2022 or 2024 is hopeless otherwise. Which is not to say one must never talk about this, but rather that one must account for this in ones planning; if fixing the fraud is not part of the plan, you have no plan.
Lawyer Appeasement Section
OK now for the fine print.
This is the WQTH Daily Thread. You know the drill. There’s no Poltical correctness, but civility is a requirement. There are Important Guidelines, here, with an addendum on 20191110.
We have a new board – called The U Tree – where people can take each other to the woodshed without fear of censorship or moderation.
And remember Wheatie’s Rules:
1. No food fights
2. No running with scissors.
3. If you bring snacks, bring enough for everyone.
4. Zeroth rule of gun safety: Don’t let the government get your guns.
5. Rule one of gun safety: The gun is always loaded.
5a. If you actually want the gun to be loaded, like because you’re checking out a bump in the night, then it’s empty.
6. Rule two of gun safety: Never point the gun at anything you’re not willing to destroy.
7. Rule three: Keep your finger off the trigger until ready to fire.
8. Rule the fourth: Be sure of your target and what is behind it.
(Hmm a few extras seem to have crept in.)
Spot (i.e., paper) Prices
Last week:
Gold $1,926.60
Silver $24.72
Platinum $994.00
Palladium $2,365.00
Rhodium $20,400.00
This week, 3PM Mountain Time, markets have closed for the weekend.
Gold $1,947.70
Silver $24.86
Platinum $987.00
Palladium $2,503.00
Rhodium $20,000.00
Gold is moving around within a fairly tight range lately.
James Webb Space Telescope Update
A lot of material has been written by the JWST team. The mirrors are lined up and focused, but they need to be focused for all five of the sensors behind the mirror before they can do the next iteration of fine-tuning. But that has only been done for four of them. What’s the issue? The issue is that the Mid-InfraRed Instrument (MIRI) has been blind until very recently.
The other four instruments have gotten down to their desired operating temperatures of 34 to 39 kelvins. (And I discussed kelvins last week. Very convenient timing!) But they all work in either long wavelength visible light (oranges and reds) or “near infrared.” “Near” because it’s right next to visible wavelengths, just a bit longer than our eyes can see. But objects at room temperature actually glow at some of these frequencies, so in order to keep the sensor itself from glowing in the very light it’s trying to see, we keep them cooled off.
But the longer the wavelength (and lower the frequency) we want to see, the colder we have to make the instrument. Mid Infrared is longer than near infrared, so we have to cool MIRI down to about 7 kelvins or less. From MIRIs point of view the rest of the spacecraft, at -390 to -399 F, is scorching hot!
This is done with cryogenic cooling, using helium. Apparently at least some of the power on board the spacecraft runs what is essentially a refrigerator, and we’ve been running that and allowing MIRI to slowly, slowly cool down to -450 F. We don’t want to cool it off too fast lest frost form on the sensor (which would FUBAR it).
This whole process is described here.
From what I can see MIRI is actually at this temperature now, so the mirror adjusting fun can resume.
What use is MIRI? It’s expected to be able to look at stars–and their accompanying planets–as they form. For instance, we’ve all seen “the Pillars of Creation” as imaged by Hubble, which is actually a part of the Eagle nebula.

This isn’t the original picture; they had Hubble go back and take another picture after they upgraded its cameras.
As presented the pillars run vertically, but the “Eagle Nebula” name was given to it based on the appearance of that area seen rotated 90 degrees clockwise. Or, here is a picture of the entire nebula, rotated only about 45 degrees. The dark area in the center is shaped a bit like an eagle, you can see the wings to the left, the body in the center, and the talons to the right…and of course, blown up, it’s “The Pillars of Creation.”

The Pillars of Creation picture is actually in false color, with green used to show hydrogen, etc., the picture of the eagle nebula is more like what it would look to us if we were close enough to it for it to be bright enough for us to see color.
But here’s what the Pillars look like in infrared:

Of course the colors here are ALL false; an infrared image in true color would look black.
But notice you can see a lot more stars.
Infrared can cut through dust clouds, including the dust clouds that condense to form stars and planets. So MIRI is expected to help us greatly in our quest to study “solar systems” that are forming Out There. This is all described in this blog post, from which I ripped off these pictures.
Fuck Joe Biden
Biden, you don’t even get ONE scoop of ice cream today.
(Please post this somewhere permanent, as it will continue to be true; the SOB will never deserve a scoop.)
Incidentally, I’m writing this on Friday. This morning I saw a full-sized pickup truck in the oncoming left turn lane with a forest of flags in the bed. Once he turned and drove left-to-right in front of me, I could see two American flags near the cab, and SIX “Let’s Go Brandon” flags near the tailgate. That was unusual; usually there’s a mix of FJB (only not abbreviated) and “Trump Won” and “Trump 2024” flags when someone does this.
I haven’t seen such a display in a few weeks, by the way. Even the guy at the office complex where I work with the FJB flag on his pickup seems to have removed it (or doesn’t work there any more–trucks all look alike to me from the back, and all crossovers look like cockroaches).
T3 Heat Engines
In the early 1800s, steam engines were the cutting edge tech. We didn’t talk of them changing the world, because before that time, really the world didn’t change much over a person’s lifetime. People in the late 1700s pretty much lived as they had hundreds of years earlier. In fact, living conditions in Europe at the time (for everyone except the nobility and a very small wealthy merchant class) were described as “Third World Europe.” Except that unlike the third world today, we had no concept of sanitation, vaccination (real vaccination, not this current crap), the germ theory of disease, etc., so we had high infant mortality rates and all the rest of the stuff that today’s Third World is getting away from.
The steam engine, and the prosperity it would bring to Europe, would ultimately change that, and usher in an era where we are actually used to seeing rapid technological change generating wealth that even the not-so-rich and not-so-connected could accumulate.
If you recall my first Thermo post, I talked about turning work (energy doing something macroscopic like moving objects around) into heat, and how heat and work were both forms of energy.
A steam engine is one of a large class of devices called heat engines, that turn heat back into work.
A common misconception is that James Watt “invented” the steam engine. No, he didn’t. Actually the first known thing that was something sort of like a steam engine was put together by Hero of Alexandria (10-70 CE), and it’s either called Hero’s Engine or the Aeolipile (from Greek αιολουπυλη). It looks something like this:
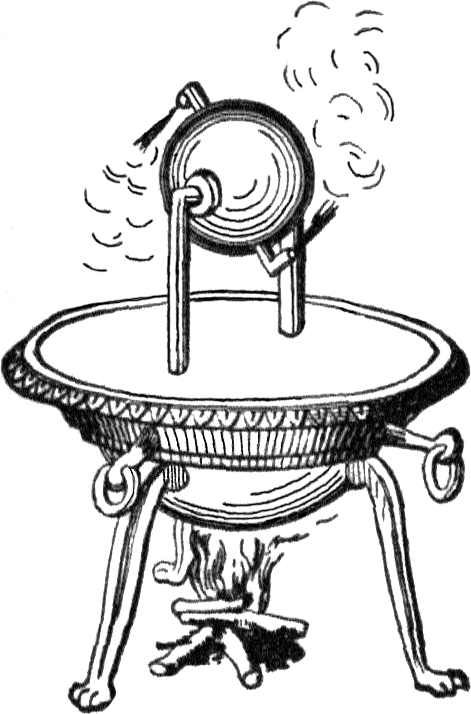
A cauldron of boiling water is kept under pressure, steam can only get out by going into an object free to move around a pivot (like a bird on a spit over a fire). The steam can only escape by going through the bent arms; it generates a thrust that causes the doohickey to spin.
So Hero was actually able to get heat to do work–some fifteen or so centuries before we had our scientific concept of “work.”
The next step was to actually put something like this to work. It took a while. In 1698 Thomas Savery invented a pump for water that worked by steam power. Steam was allowed to cool and condense , which created a near vacuum which could be used to suck water upwards against gravity. Then more steam could be used to push that water.
So the first commercial application for steam power was to pump water.
Small copies of this device were reasonably efficient, larger ones were not. And lift height was severely limited (since it was a vacuum pump). And the power wasn’t continuous.
In 1712 Thomas Newcomen came up with a steam engine that could deliver continuous power and it used a cylinder, but was still very inefficient because parts of the engine itself (not just the steam) ended up being heated and cooled, heated and cooled, over and over again…which took time. These things could pump water out of mines…slowly. Still, it was better than paying some grunt to do it.
What Watt did was vastly improve the Newcomen engine. And now it could be used not just to pump water, but to pull trains, and power steamships, and even (very primitive) automobiles. And also to power factory equipment, which up until then had been almost entirely reliant on water power or (worse) animal power.
A good steam engine could supply LOADS of power.
And of course this was something scientists investigated. And what they learned could be used to make steam engines even more efficient, delivering more work for the same amount of fuel, which could be either wood, or coal that was pulled out of some of the mines the steam engine had been invented to pump water out of.
One “model” of an engine that could turn heat into work was the Carnot Engine, basically an idealized model, a thought experiment.
What’s needed is a piston with gas in it, a huge mass at high temperature (called the hot reservoir) that we can attach to the piston, and another huge mass at cold temperature (the cold reservoir) which we can attach to the piston as well. It runs through four phases, then from the fourth phase, it returns to the first phase.
Phase one starts with the piston pushed in all the way. There’s a tiny space inside filled with hot gas, under pressure. The gas is at exactly the same temperature as the hot reservoir.
Now you could let the gas under pressure expand, and push on the piston and use that to do work. But as the gas expands, it will lose pressure and the temperature will drop. Instead, what we want to do is let the gas absorb heat from the hot reservoir, so that it stays at the same temperature as it expands. Ideally the temperature is exactly the same but in practical terms this is unachievable (unless you are willing to take literally forever for it to happen).
Phase 2 is to allow the gas to continue to expand and push the piston, but now you let the gas cool off…until it reaches the temperature of the cold reservoir.
Phase 3 is to compress the gas, and keep it at the same temperature as the cold reservoir. Compressing the gas would normally raise its pressure and temperature, but we let the increased heat bleed into the cold reservoir. Do this infinitely slowly, and the gas in the cylinder stays at the cold reservoir temperature.
Phase 4, finally, is to continue compressing the gas, but let it heat up…all the way up to the temperature of the hot reservoir. We’re now back to having compressed gas at the temperature of the hot reservoir.
So what has happened here? We’ve extracted a net amount of work from the system…more came out letting the gas expand and cool than we had to put into it recompressing the gas. Also, some heat energy has been taken from the hot reservoir and put into the cold reservoir.
In reality, this would result in the hot reservoir cooling off just a tiny bit, and the cool reservoir heating up just a tiny bit, but this ideal model assumes these reservoirs are of infinite size.
So what’s going on here? Heat has moved, and some (notice: some, not all) of that heat got turned into work. The rest simply moved from hot to cold and did no work for us at all. And since heat moves from hot to cold naturally, not the other way, that heat energy we didn’t use is gone for good.
As it turns out this “Carnot Cycle” is the most efficient possible way, even in principle, to turn the heat differential between a hot and cold body into work. And it gets more efficient the greater the difference between the two. (And this is true even for engines that are steam turbines with no piston at all, or where the working fluid changes from a gas to a liquid…they can’t beat the Carnot engine.)
The maximum theoretical efficiency is 1-TC/TH. Note that those temperatures need to be measured against absolute zero (so kelvins and Rankines work). You’ll note that if TC and TH are far apart, that fraction gets smaller, and the efficiency climbs, if you can make that fraction 0, your efficiency is 1 (i.e., 100 percent). As a practical matter, though, we don’t have a cold reservoir at 0 K, so we can’t reach 100 percent efficiency even in principle. And whatever this number is…our current power plants don’t even achieve that.
But this is why power plants (coal, nuclear, natural gas, even geothermal) run so hot; it’s to raise the temperature of the working gas as high as possible. And this is why geothermal usually sucks as a power source. You have to drill way, way, way down to get to rocks hot enough to be worth the effort. Of course in some places, like Iceland the hot stuff is closer to the surface. Yellowstone has, if anything, even more potential than Iceland does for geothermal power.
And of course with nuclear and fossil fuels, we control the temperature difference and you can bet we run them as hot as we can.
Since a Carnot engine is as efficient as you can get, a lot of engine design since the mid 1820s has been to try to get as close to a Carnot engine as possible. (That was, for example, the initial idea behind the Diesel engine; eventually Diesel had to start making design compromises.)
In every power plant we build (other than geothermal) the heat reservoir is not only not infinite in size, but it’s actually rather small. It would cool off rapidly as the engine cycles, and the heat is transferred out. To keep it hot, we have to burn fuel. So we’re basically burning fuel to create heat to maintain the temperature of the hot reservoir so we can then turn the heat into work.
With geothermal, the reservoir is huge, perhaps even yuge, but still finite; using geothermal does suck heat energy out of the earth and dump it somewhere colder. It’s just that there’s so much of it we’ll never notice.
A typical coal fired plant uses the work extracted from burning the coal (and keeping the hot reservoir hot) to turn a generator. And the efficiency is roughly 30 percent. 30 percent of the energy in the coal becomes electricity. In fact the engineers and designers are overjoyed when they get that number.
But that does mean the other 70 percent is wasted as waste heat…and it can’t be any other way.
Waste heat is inevitable, and it can’t be recovered. Not without reversing the process. We can push heat energy from the cold to hot reservoir by supplying work. If it’s a Carnot engine, we can run the process in reverse, put the work we got out back in, and move all the heat back where it came from. For any real engine, it will take more work to move less heat than we lost creating the work in the first place. In other words, a Carnot engine is a reversible process. But no real heat engine is reversible this way. But if you try, you will move some heat from cold, back to hot.
This is the idea behind a heat pump.
In reality, there are always losses, and (again) there’s no such thing as a Carnot engine. So if, say, you ran your (real) heat engine and got 100 joules of work out of it, and 200 joules of heat ended up in the cold reservoir, you’d probably need to put 150 joules into it to move those 200 joules back to the hot reservoir. Where would you get the extra 50 joules from? Maybe from another engine, which has its own losses and its own inefficiencies, and even more energy flows from its hot reservoir to its cold reservoir.
In fact, there’s no way to completely reverse heat flow from hot to cold. Any attempt actually moves even more heat from hot to cold in the process. Which might be worth it, if you want to cool the inside of your refrigerator. You suck some heat energy out…at the cost of more heat energy being permanently lost somewhere else.
But on net, heat flows one way, one way only: From hot to cold. Even a theoretically perfectly reversible process like a Carnot engine just holds the total flow to zero. Never negative.
This is what was originally behind the concept of entropy. It denotes that fact that as processes go forward, temperatures even out, they get more and more evened out as time goes on, never the other way around.
(If you think you know of a case, like the inside of your refrigerator, recognize that that’s at the cost of more heat flow in the other direction somewhere else. This is why you can’t cool your house by opening the refrigerator door and leaving it open–it dumps the heat through the coils in back…more heat than it sucks out of the inside. Your air conditioner, on the other hand, does work…by dumping the heat (again, more than it removed from your living space) outside your house and making the outside warmer than it was before.)
Entropy is, conceptually, a measure of how much heat energy is irretrievably lost, and it increases. (It can even be quantified and measured…but that’s WAY beyond the scope of this post.) Entropy never shows a net decrease.
(Entropy is also second only to quantum mechanics as a source of pop philosophizing with a scientific/geeky bent to it. More on this in future posts, because the concept of entropy would eventually be greatly extended beyond mere “waste heat” type considerations.)
So what happens at some distant day when all of the energy in the universe has become waste heat and is irretrievably lost? Well, it’s game over.
Thinking in the long term, the big picture is the entire universe is running down. It will ultimately be one big thing of uniform temperature…and though there will be a lot of heat energy out there, none of it will be usable, not just for industry, but for life itself.
The concept of entropy, and the accompanying realization that the universe as a whole must run down, was the dawn of the science of thermodynamics. The true “dismal science.” Economics is sometimes called the dismal science, but it doesn’t hold a candle to thermodynamics when it comes to “dismal.”
Obligatory PSAs and Reminders
China is Lower than Whale Shit
Remember Hong Kong!!!
中国是个混蛋 !!!
Zhōngguò shì gè hùndàn !!!
China is asshoe !!!
China is in the White House
Since Wednesday, January 20 at Noon EST, the bought-and-paid for His Fraudulency Joseph Biden has been in the White House. It’s as good as having China in the Oval Office.
Joe Biden is Asshoe
China is in the White House, because Joe Biden is in the White House, and Joe Biden is identically equal to China. China is Asshoe. Therefore, Joe Biden is Asshoe.
But of course the much more important thing to realize:
Joe Biden Didn’t Win
乔*拜登没赢 !!!
Qiáo Bài dēng méi yíng !!!
Joe Biden didn’t win !!!

00:13 or so.
Glad you caught it – I got distracted at 00:09!!!
I was writing an email to my mom, who was complaining about radio talk and radio music being too fast. I was saying that the internet had items on a variety of tempos and used
as an example.
After I hit send, I read the comments. Sheepishly.
I had a music history professor who referred (jokingly, of course) to E. Power Biggs as E. Bower [Bauer] Pigs… 🙂 (not sure what Cherry Rhodes and Ladd Thomas think of that 🙂 )…. [ https://music.usc.edu/two-lifetimes-of-music ]
And then, there’s Rocky’s music 🙂
Helene
@mostlyorganic
1h
·
BREAKING: Missouri Supreme Court orders Soros-funded prosecutor Kim Gardner to turn over communications with George Soros and Missouri political insiders related to her false case against Eric Greitens
https://gab.com/emoji/1f517.svg
Source
SHARE this far and wide
https://gab.com/emoji/1f1fa-1f1f8.svg
Join Us
https://gab.com/emoji/1f449.svg
@SGTnewsNetwork
1 like
1 repost
Interesting clip from some medical seminar (probably an iPhone video) about proving that the clot shot vaccine can shed and transfer by sexual intercourse.
https://t.me/SGTnewsNetwork/20173
Also on Gab:
https://gab.com/SGTnewsNetwork/posts/108048370166264555
[video src="https://media.gab.com/system/media_attachments/files/103/011/186/original/192bd1e2112d548a.mp4" /]
According to the presenter on the video, it appears there’s discoloration of the blood, clotting, and Candida present — in addition to impairment of the immune system (one is assuming that a separate test for T-cell and B-cell levels was performed.) It also appears that the blood in question is from an “UN-vaccinated” person who had intercourse with a “vaccinated” person.
Which then begs a question: when two persons who are BOTH “vaccinated” have intercourse, can it be assumed they are “transferring” elements of the spike protein POISON between each other? What does this do to their blood, immune systems, and so forth — does it accelerate / aggravate the damage from the “vaccine”?
From there: Can it be posited that if a child is conceived from intercourse after one (or both) of the parents is/are “vaccinated”, that the spike protein POISON is transferred to the child at conception?
This is actually part of why I was kicked off Twitter and my account hidden, under the excuse of “QAnon”.
I had pushed some stuff discovered by George Webb – that the alleged Trump “communications from Trump Tower with a Russian bank” were likely CREATED by the people accusing Trump, or somebody working with them – that it was a set-up.
WELL LOOKIE HERE……
Vickie B reposted
techno_fog
@techno_fog
7h
·
New – Letter from Special Counsel John Durham to Michael Sussmann’s attorneys:
The Special Counsel’s expert may testify to the possibility that the Alfa Bank/Trump data “was fabricated, altered, manipulated, spoofed, or intentionally generated…”
1,693 likes
98 comments
815 reposts
13 quotes
Here is the image that I was sharing on Twitter…..
Retired Navy Corpsmen
@Devildoc696
3h
·
14 likes
1 comment
17 reposts
Charged for disseminating truth. She’s a heroine.
Amen! Orwellian – when speaking TRUTH is a CRIME.
So, I guess since all deplorables and patriots knew this would be the result in advance of the result being disclosed – that makes us guilty in the Orwellian World
Knowing that to be true there and self evident as a fact of our existence on this planet, what do we expect the result of decades of open borders without retribution will be?
America came into existence through the blood and toil of people who trusted God and believed in liberty and freedom. IMO, that will be the only way it can return to its purpose and promise. We need the Lord’s hedge of protection around us again or we will be destroyed from within. We cannot expect His protection if we do not do as He commands as a people.
As far as I’m concerned, GAB is the center of Godly civilizational resistance right now.
Everyone realizes this will come to the US with being invaded?
Linda
@Guest4Ever
13m
·
https://t.me/GeneralMcInerney/2518
General McInerney
Coming soon..
t.me
View Link Feed
1 like
1 repost
Never loose the fire in your heart Steve
Possibly a propos of nothing esp as I can’t get a good look at it.
there is a bus shelter advertisement I pass on the way home which has a picture of a plastic bag and the banner wasting food is worse than using plastic.
disclaimer- I can’t read who’s the advertiser- print too small
i don’t see it every day as it’s one of those changing ones and I don’t always see it on the way home.
BUT- is this about trying to implement a mental sea change?
is famine or food insecurity looming large in the near future?
im giving the side eye
Like the Stalinists of old, “they” are trying to change the system by central control, which WILL cause famine and food insecurity. They are preparing us for it, so that we don’t choose to fight back. They want us to accept death peacefully.
Democrats are communists.
Communists will kill us before they will give up power.
Do the math.
Or they are trying to reset us mentally to the mindset of our grandparents or great grandparents ,who survived the Great Depression ,.
seriously my olds would waste nothing. Every scrap of food was scooped up to become bublensqueak before reluctantly being consigned to the chooks. And any chook who went off the lay because of age literally became an old boiler. Nothing would bring out the inner demon of my grandmother more than the picky kid who rejected food because “I don’t like it”
Yup. I remember my folks doing what needed to be done to get me to eat my vegetables.
No desert and you don’t leave the table till your plate I clean.
and they wonder why , when food became plentiful and cheap we got fat
and now with appliances, cars, phones being so Smart + Alexa, ppl are getting even fatter and lazier and dumber.
💖 Luddites, Amish 💖
A study of Amish showed their diet isn’t particularly “ good” – they have full cream milk, eggs etc. no low fat shit for them.
researchers discovered the key was 10,000 steps a day. You walk that much and your metabolism kicks in, you have a higher proportion of muscle / fat – it’s muscle that burns calories,even when resting.
I’m not talking about their diets…that’s their business.
I am talking about their ability, generally, to sustain themselves with out over reliance on technology and Smart stuff…and their strong sense of community and knowing how to help like-minded survivors..know-how…survivalist know-how.
and the “soverign citizen” type of attitude towards Big Govt.
I spent many a night sitting at the table until bedtime. I was NOT eating peas or cooked carrots or the peas/carrots/corn medley and I MEANT IT!! I eventually won that war. Mom couldn’t finish the dishes or do other chores and finally grew tired of the monitoring, pleading, threatening, and bribing drama night after night. Negatory on the brussel sprouts too. They went in the fake plant in the corner and I got busted after they started reeking the atmosphere. 😱 😉 😊
LOL!!! You gotta get ’em to the john – they’re the perfect size to FLUSH!!!
The problem with Brussels sprouts (I hated them as a kid – love them now) is that our parents didn’t know how to serve them to make them tasty.
The Brussels sprouts of my youth were limp, overcooked, tasteless or worse, and mildly stinky. The WORST vegetable of them all. My parents gave up QUICKLY.
Modern Brussels sprouts are oil-cooked, fried or quick-roasted, crisp and firm, peppered, tasty, and often split. They capitalize on the best features of many other vegetables, stolen by the prep.
I can eat a ton of the good ones now. Perfect with MEAT and even BACON, which makes them taste fantastic!!! 😉
You’re right about the taste and no seasoning thing. From frozen bag to overcooked mush in pan w/water on stove to table, HECK NO!!
I actually would probably like them now prepared tastily but the childhood trauma prevents me from going there. I’ll keep busy with the broccoli, FRESH asparagus, cabbage, kale, spinach, chard and other veggies that I grew into liking.😉😁
Yeah, don’t try it yourself at first. I let the PROS tempt me back.
My advice is to go to some places with real chefs, that serve luxury pub food, and pick a great meat item served with the little death cabbages.
These chefs KNOW that many if not most people can’t stand Brussels sprouts, so they pull the ultimate makeup jobs on ’em.
Heck, I’m getting hungry just talking about ’em!
And I just ate lunch!
👍😁😁
Tip. Cook kale with coconut oil. It makes them easier to scrape into the bin
Brussel sprouts were my line in the sand. Tried them as an adult and, yep, they still taste foul.
my brother was a bitch. Every meal it was – this was a chicken, this was a baby lamb.
Exactly, my parents were the same, I think it’s because they lived through ww2 and rationing.
It never left them though and I think it must have rubbed of on us kids, waste not want not.
Same here, too. My parents survived the depression and WWII in the USA and England, as did my grandparents, and NOTHING went to waste. My father had tools, for instance, that looked like the day they were bought, despite being a generation or two old, and used a lot during that time. My wife’s parents and grands here in Germany, same thing…
“Eat your befores before your afters” was often heard, as was “you’ll not leave this table until you’ve finished your meal” (or veg, or whatever).
It also encouraged planning, thriftiness, circumspection before purchasing, and, at least in my case 🙂 a formidable spare parts bin… which I’ll probably spend the next few years trying to sell on eBay, etc.
[Anyone need a 5¼ floppy drive, with A/B IDE cable? Or an Epson 2100 A3+ printer, complete with roll-cutter, basket, roll photo paper, etc.? And that’s some of the NEWER stuff 😀 ]
Funny thing is, stores are now back to selling unpackaged fruit, veggies, etc. (they haven’t gone to selling hardware that way, drat), and acting as if they’ve invented something new. Hmmm. Tack the word “sustainable” (nachhaltig) onto anything and it becomes a new trend…
OMG, I just spent an hour rummaging through tubs of my old computer parts and cables the other day! I (we) really need to toss that junk. Btw, you need some 3 1/4 diskettes? I got DOS and Win 3.1 too.😜😁
Historical artifacts!
Send them to the Greenfild Village Museum in Dearborn, Michigan …after all they have Edsel automobiles 🚗 in there ‘snicker .. 😜
🤣
There are actual computer museums out there.
Guessing. Coothie has some relics, the museums would be interested in.
😂 😅 😂
They’re full.
https://computerhistory.org/
And, if you note their “covid” policies, full of it.
I’ve actually never been.
🙂
I’ve got HP NewWave, HP Internal Edition, factory sealed, 5¼ floppies (maybe 3½ diskettes, too)… good match for that Win 3.1 OS 😆
I have MSDN going all the way back to then, in many MANY languages… Brazilian Portuguese, Dutch, Chinese(simplified), Czech, Hebrew, Italian…. every product that Micro$haft makes… LOTS of coasters there…
My Mom used to make ornaments and artwork out of old CDs… Oh, btw, do you need any AOL CDs? 😀
Murphy (must’ve been one of my Irish ancestors) says that as soon as I get rid of any of this, I’ll need it, and it’ll be nowhere to be found…
At least that’s what he says… between sips of Guinness 🙂

Cheers! Sláinte! Prost! 🍻
UNCLE!!!!🤣🤣🤣
The cd on a string rear vision mirror!
Found a good use for old or blank CD’s. Hang them with some garden twine or wire to keep the birds and critters away from garden areas. They blow in the wind and the sun reflects off of them.
We have been attacked by flocks of birds the last few years. For the previous two years it was starlings nesting. This year it’s robins. Big, fat, ornery robins. Had tried fake owls and lately, rubber snakes. The snakes do better than the owls, which they just roost and crap on. 😂 But even the snakes get ignored after awhile if you don’t move them around frequently. Enter CD’s. Problem gone.
Remember when you brought nails/ screws loose by the pound?
It does rub off.
It actually pays off as well 😉
Yes 🙂
I was pretty much raised by my Dad’s mom, my Nanny, I called her. She lived through the Depression. She only attended school through the third grade, and couldn’t drive a car, but she fed three families with vegetables she grew and preserved. I literally never ate a store-bought vegetable at home.
She made “bread pudding” that was just like brownies out of leftover biscuits and/or cornbread. She could make the roughest greens taste like heaven. She used every scrap of fabric in something. My Dad grew up wearing underwear made by her out of feedsack material, which used to be nice cotton.
She saved everything. Plastic bags, rubber bands, string. Nails, nuts, bolts. Tools were lovingly cared for.
I will say I was never forced to eat anything I didn’t like. But her cooking was so good that there wasn’t much to complain about!
I learned at her knee. I’m gonna make it real hard for these fuckers to kill me and mine.
Your Nanny sounds like my Oma who would save and use everything. She also could do everything. Baked heavanly plum cakes and other simple cakes. She cooked wounderful food grew her own chicken and had a garden. The garden was not very big but she grew a lot in it and preserved. Women from yesterday were very compitend less educated but resourceful and kind.
Yes, they were.
I make a pretty mean plum cake, myself, lol!
Great 🙂 Yummy
Love it.
My wife and I are in the process of doing memory books for our families. My paternal grandmother, Mamaw, sounds a lot like your Nanny and singinsoul1’s Oma. She was sharp, earned her teaching certificate back when women did not get to go to higher learning schools much back in 1916. Taught elementary school kids in the old coal mining camp towns. Came from a poor, hard working coal mining family. Knew no other way than to work hard, scrimp, save everything that could be of value, make do. Made their clothes, worked in the veggie garden, canned, etc. Best country cooking you would ever want to eat. Simply made magic.
Add in being an Appalachian poet of note in Who’s Who in American Poetry back in her day. I brought one of her poems to the QTree a couple weeks back, Probably will bring more in the future. When her and Papaw died they only had about $700 in their savings account, no home (rented), never had a vehicle (walked, took a bus or train), etc. They were wealthy beyond imagination in what matters.
Shining lights in most of our families for future generations if we would simply pay attention.
Lovely story, and I’m glad you are doing memory books. That is so important.
I called my great-grandma Mamaw.
Somewhere in all family lines, no matter how far back, somebody knew how to subsist, otherwise their progeny would have died out. I think we are about to learn how to do it again. Then our families can write memory books about us 🙂
Please bring more poetry from your Mamaw.
Will do. Probably on Sundays. Seems appropriate with Bakocarl’s messages. Mamaw’s wording clearly conveys faith, family, friends and love of nature throughout.
Sounds like the people arround where I live. Grandparents you describe live in the Hallows of Appalachia. Nothing much has changed for them. They are a close family God fearing happy people. The music that come from the hills stirs the heart. They are also not trusting of strangers but since many have been in military they are open to me. They seldom accept help. When there was a flood I brought church supplies to them they took it but rejected it from another woman who brought items to them. They are self reliant. They have reasons to be not trusting outsiders have exploited them in Steel mills and coal mines. They are hardworking good people.
I had to work on that not to become to upset when my kids did not wanted to eat. They did using same method I was taught. No struggle just get to eat next day what you did not eat the day before. When people are hungry they eat.
Still today when I make a comment when ives me a list what his wife is not eating when they visit I said one time ” the reason is she never was hungry.” He gets upset with me when I say it. Going out with my kids spouses is a nightmare. Seeing them pick over food one does not eat this the other that and leave half the food on the plate.
If I get to much food I set it asside ask for a box and take it home. We do not through food out and I use everything.
Such a simple, truthful concept. I am clueless why the average American does not understand it and resist yet. I don’t know what else to do that has not already been done.
They will understand when they are hungry. Then we will see some shit we would rather not have to see. But it will be interesting.
Americans have become unaccustomed to struggle.
My Jewish cello professor and her sister escaped death by the Nazis in WWII via the Dutch Underground. They wrote out their music exercises on blotter paper, by memory, and practiced silently to keep in shape. Fortunately they were able to escape Germany and end up in the USA.
She said to me one time, “the problem with your generation is, you haven’t suffered enough”… then explained some of the hardships they endured. I was floored, and humbled, and, yes, corrected.
That was almost 50 years ago. I shudder to think what the two or three generations after mine must (not) think…
When I was a wee bairn, I used to follow my Dad around like a lost puppy, sometimes getting in his way, but he was always patient, kiend, and loving with me. And almost every time I went exploring the garage, I’d find something new (to me) or unusual, and ask him what it was or was for. One time there was something made out of steel, painted green, that looked like a rounded foot on a frame like an upside-down leg. I asked what it was, and he replied that it was a last, which one used to make shoes.
He could make his own shoes!
Coming from Appalachia, he could make (and fix) pretty much anything and everything, and not waste a tool, or time, or money. And he could grow things, be it animals or plants. We used to have one of those over-the-toilet cabinet things on spring-loaded poles, and there would always be some plant(s) wound around the poles with SaranWrap or a baggie filled with (a little) peat moss, soil, rooting compound in the middle, where he was grafting, making a root from a cutting, etc. He could probably have made a brick take root… I was always surprised and fascinated with what he (and my Mom) knew and could do. Educated in the University of Life and Hard Knocks, so to speak… and despite all of that, we never wanted for anything, and appreciated what we had, which was infinitely more valuable that what passes for life these days…..
I think maybe in these times GOD is nudging us back in that direction; to be a little (OK, a LOT) more self-sufficient, humble, THANKFUL, and observant of the provenance that GOD has showered upon us.
The Greens completely misinterpret “subdue the earth” as destroying the earth (and its contents, as it were). The point of the passage is shepherding, husbanding, managing, and preserving, something of which the Greens, et. al. have no clue. But we seem to have gotten so far from our roots, from the land, that we have no (or little) connection anymore.
Grating the Greenhearted and her ilk continuously yammer about “the Earth” and everything, yet they wouldn’t know what to do with a 20×50 plot of land, some fertilizer, some seeds, some water, a rake, a shovel (trowel some call it), and a lot of elbow grease. Indeed they wouldn’t know what to do with a Farmers’ Almanac or a Western Garden Handbook if it fell on them…
Talk about “gotta get back to the garden”… Joni was talking about the wrong garden…..
There is also a Southern Garden Handbook which is almost identical to the Western….with the major change being how they do climate.
The Southern one uses USDA cold maps, then developed their own “hot maps”, and they’ll rate plants as being cold zone X-Y and hot zone A-B.
The Western has microclimates.
I used to work in the building that the Lane Brothers built (Lane Publishing) in Menlo Park. STUNNING grounds doesn’t even come close. It was a great campus, but, sad to say, it’s been sold; not sure if they tore everything down or not…
I was one of the few permanent employees at ADIA, the temp agency (internal IT ops mangler) and we shared some of the building. I suspect they were on their way downhill even then, as we were just renting (smart move on their part).
My parents got each new edition as it came out; I have a few of them, including one of the last that was published. Also some of their design books, which give great information, and a taste of the 60s, 70s, and “Western Life”… ahhh, those were the days…..
I know the gardens well. They used to have annual “open houses” (with about 10% of the actual buildings open).
I guess the last edition of the book was the Ninth, in 2012….but you never throw the old ones away.
Oh, yeah. Gotta get back to the garden is right!
I am getting ready to teach a beginning gardening class next week. I hope my students are ready for my “folk gardening” approach. Our forebears didn’t have the internet, artificial fertilizers, expensive equipment, or any of the gadgets and gewgaws people want today. I garden mostly with a few hand tools and found items like buckets for containers. I want to teach people NOT to spend a fortune to get a whole bunch of food. Wish me luck!
(1) Good luck!
(2) Rubbermaid tubs can survive a LOT of abuse. They can be used to raise fish, or you can drill holes in the bottom for plant containers.
(3) I have found that a good place to start was an understanding of dirt. We currently have adobe clay which we’ve supplemented with some river sand and lots of compost over the years.
Thanks!
I do have some Rubbermaid tub planters!
I have spent over a decade on the soil I’m on. I use nothing that will damage an earthworm, that’s my rule. I dug up one today that was so big it startled me! About ten inches long. Way down, I have had two-footers.
Good compost is garden gold.
Speaking of gardens- where are they?
it’s all alfresco outdoor living areas etc.
presidential land is often excellent soil and all we put on it is lawns and garden furniture
Average American, Go Along, To Get Along.
Spoonfed Pravda’s Official Narrative by TW, FB, Big Tech, Fox, CNN, NBC… hollyweird talking heads, hollyweird actors… AND UNIPARTY LIARS.
Remember Chyna and their ‘one plate’ campaign?
How about these plates? 😀
(Lucy and “Unbreakable Dishes” at 8.50 in, if it didn’t work right)…
I LUV LUCY!🤸♀️😍🤸♀️
Excellent short video by Robert Malone who basically says that there is a big difference between saying that the vaccines cause HIV (which is not true) and saying that they cause AIDS (which is syndromatic, and appears to be true).
FRANK QUOTES https://gab.com/emoji/1f69a.svg🩸https://gab.com/emoji/1f69b.svg
@FRANKQUOTES
3d
·
FOLLOW https://gab.com/emoji/1f449.svg @FRANKQUOTES
https://gab.com/emoji/2139.svg
Dr. Robert Malone: “The Vaccines Are Causing a Form of AIDS” | A condition called VAIDS. (Vax induced AIDS) https://gab.com/emoji/1f489.svg
“We have lots of scientific data now that are demonstrating that these vaccines, particularly RNA vaccines, are damaging T cell responses…
They’re causing a form of Acquired Immunodeficiency Syndrome”
[video src="https://media.gab.com/system/media_attachments/files/103/440/530/original/c4045106ca69c9f0.mp4" /]
1,542 likes
121 comments
1,331 reposts
25 quotes
The photos of The Pillars of Creation are beautiful. (Yes, I looked just for the pics, LOL.) I’m assuming the JWST will be able to take even better ones; at least I hope so. Thanks for the update on it.
I assume that would be a very distant day, at the rate we’re going. 🤔
Fire and Ice Launch Audio in a New Window
BY ROBERT FROST
Some say the world will end in fire,
Some say in ice.
From what I’ve tasted of desire
I hold with those who favor fire.
But if it had to perish twice,
I think I know enough of hate
To say that for destruction ice
Is also great
And would suffice.
How true!
Billions, maybe even trillions of years.
The sun will run out of fuel long before then…but then, “the sun run[ing] out of fuel” is also entropy at work.
No. Not our problem
Saved by Google, again. 🙂
I read the end of the Book. God says He’s gonna create a new Heaven and a new Earth. If He said it, I believe it.😍
EFF all these Climate Groomers!
Bless you for that comment.
🤗🤗PGROUP🤗🤗
AMEN!!!!!
this is so sickenng !
#BoycottDisney
Child-Grooming DISNEY Linked To Ghislaine Maxwell & Epstein’s PEDO Island…
article link…
https://www.naturalnews.com/2022-04-08-child-grooming-disney-linked-to-ghislaine-maxwell-and-jeffrey-epsteins-pedo-island-boycott.html#
4/8
why was Maxwell doing fundraising for Disney in 1985 ??
more on this + photos etc, at the link.
YIKES.
Hello Smiley,
Good to see you.
A kind and respectful FYI on the down low.
7 years ago, during Trump’s first campaign, I was an admin for ReTrumplicans, about 50K members and extremely active. We were dealing with all the Macedonians, etc., posting crazy crap on our page.
I noticed Natural News was one of the worst and got curious.
Did a major deep dive and tracked them back to a source in Thailand. Almost 100% of their content is crap. I had such a bad experience with this particular site, I was able to ban them from our page….. and I only did that with a few.
Be very careful of anything from Natural News. Please.
Thank you!!! (And HI THERE!!!!)
We need to be on the lookout for disinformation purveyors that are supposedly on “our” side. (E.g., Dianne Marshall, but also these folks.)
ok well if you could also provide some links to support your opinion, that would be helpful
I like that site and I will continue to post articles from it if I think they are valid.
Some of the info there can be a bit hyped sometimes in which case I won’t post it for fear of getting replies like yours….and screams of “disinformation”.
Your bad experience was from 7 years ago.
I don’t necessarily doubt what yr saying but the site may have improved since then ?
i don’t know…7 years ago, I did not even know about the site.
Much of what he says is substantiated by other sources.
he is not a “disinformation purveyor”…but he does get carried away, at times.
I really don’t appreciate the suggestion that I am putting “disinformation” in the comments section here…unless you can show me exactly what I posted that is “crap” and/or “disinformation”.
This article gives a bit more information on the two phony DHS agents.
Seems their downfall was brought about by an almost accidental set of circumstances.
https://www.rt.com/news/553502-fake-dhs-agents-arrested-bidens/
There’s something so off about this. The article characterizes it as a bizarre charade but at some point the secret service and other govt employees surely at least looked for their credentials.
Have the govt agencies become so cloaked in separate authority powers that each doesn’t bother checking thoroughly even though they clearly bragged about powers that should have raised eyebrows long before this happened?
This quote immediately made me think of Pelosi’s new Capitol Police Force. It’s becoming believable, even to supposedly seasoned secret service agents ,that there can be clandestine rogue operatives snooping into an entire building of citizens ?
The quote FTA ~ Not only could they not resist boasting of their bogus Homeland Security connections, they also bragged that they were deputized “special police” with the Washington DC government and involved in undercover gang investigations, as well as the January 6th probe.
It’s got an Awan Brothers smell around it.
Speaking of Capitol Police, a short bio on Dingy Harry [wiki?] that I read described him getting a job there as his introduction to DC. As political patronage. IOW, qualifications to be a police officer were not needed.
Ashley Babbitt can certainly vouch for the accuracy of that statement. Oh wait, …
Sundance has an update about Truth Social. Sounds like they really have been following the GAB model, and making sure they cannot be cancelled, by robust independent hardware and infrastructure acquisition.
https://theconservativetreehouse.com/blog/2022/04/08/ceo-devin-nunes-provides-an-update-on-truth-social-media-platform-web-based-access-likely-in-next-few-weeks/
Liking, web based available in a couple weeks.
Immune to Big Tech tyranny. 🙂
Android…guessing months.
From the CTH article, a sobering Truth in today’s world… (Emphasis added)
“They are going to need revenue that is not subject to the whims of other third-party payment processors and develop advertising systems outside the behemoth overlords at Google. These are not easy challenges to overcome.”
Exactly!!!
Alas, Windows access!!! Now they’re cooking with gas.
Flatulence? Or some other type of gas?
😂
GaAs… Laser heat 😀
Israel Is Bleeding
the real PM...
“Israel Is Bleeding ” : Right Rallies After Government Destabilized
article link…
https://www.jpost.com/breaking-news/article-703418
4/6
the shocking resignation of Idil Silman (pictured above) from PM Bennett’s coalition administration….and defection to Netanyahu’s opposition Right.
👉 rising wave of terrorism
👉 Silman’s sudden resignation
…are quickly destabilizing Israel , causing the demise of Bennett’s coalition.
pro-Netanyahu rally of supporters turned out en masse last Wednesday,
more, at the link.
Binyamin Netanyahu will always be overshadowed by his older brother, Yonaton, who was killed during the raid on Entebbe in 1976. He is a fanatic for the nation of Israel, despite any minor personal enrichment he may have accrued while leading it.
his brother is not at issue here or with the information posted.
as to your opinion about Netanyahu being a “fanatic” about the nation of Israel, many say the same thing about Trump and Make America Great Again.
I think fanatic as in die hard patriot and believer
and, of course, there’s the bigger picture…
Will Political Uncertainty In Israel Lead To Iran Conflict ?
Analysis…
article link…
https://www.jpost.com/middle-east/article-703412
4/6
Just some random weirdness…..
Totally
AWESOME
‼️‼️‼️‼️
bwahahahahaha .. bwahahahahaha 😂
definitely sharing
That was fantastic! Thanks for that.
everything’s a scam…
Guess Who Scores Billion$$$ In Biden’s Iran Deal ?
answer, at the link…
https://hotair.com/ed-morrissey/2022/04/08/guess-who-scores-billion-in-bidens-iran-deal-n461002
4/8
sweet !
(forgot the /s/ after sweet ! )
Marxist Reset: Federal “Derogatory Geographic Names Task Force” (Yes, Really) Announces The US Will Rename 660 Mountains, Rivers, and landmarks to Remove “Racist” Language – More Surely to Come
https://www.thegatewaypundit.com/2022/04/marxist-reset-federal-derogatory-geographic-names-task-force-yes-really-announces-us-will-renaming-660-mountains-rivers-landmarks-remove-racist-language-surely-come/
Surely they could have come up with six more?
LOL !!
To be added later! Reserved for Hitler, Stalin, Mao, Marx, Engels and Gramsci!
LOL! Yeah!
Those telescope photos are amazing!
The universe seems endless and the phrase – worlds without end – comes to mind.
And I wonder if elements and substances not yet discovered – not present on earth – are there on or in those planets, stars, star-space dust clouds, constellations….
and the universe is ever-expanding…
Elements? Probably not, unless there really is an “island of stability” and it’s *stable*. But then we’d have to figure out how those elements could have been created in the first place.
(I say probably because one should never say never.)
Substances? That’s a much broader category and I don’t doubt for an instant we’ll see stuff not seen before. We keep finding new stuff right here on Earth, after all, where we’ve been all along.
I knew you’d have a good answer.
Or, if you see blue, you need to back away from the computer at about half the speed of light.
Or just flip the channel to “Red Shift” 🙂
Breitbart: SLAVE LABOR’-FUELED CHINESE FLEET DESTROYING FISHING INDUSTRY IN WEST AFRICA
China is really a bad world citizen.
From pollution to over-fishing the world’s oceans – to aggression in the South China Sea – from to cyber-theft to dangerous toxic products – China is a bully, thief, destroyer.
YUP.
Verse of the Day for Saturday, April 9, 2022
✟
“Wherefore, my beloved brethren, let every man be swift to hear, slow to speak, slow to wrath:”
James 1:19 (KJV)
Thank You, Jesus, for blessings received and prayers answered!!!
BE MY VOICE
BABY LIVES MATTER
PRAYING ON THE ARMOR OF GOD
Father God, I now follow your command to put on the full armor of God, because my battle is not against flesh and blood but against rulers, authorities, the powers of this dark world and against spiritual forces of evil in the unseen world.
I first pray on the Belt of Truth that it may be buckled around my waist, may I be centered and encircled by your truth dear Lord. Hem me inside all that is true and right, and may I be protected and held up by the truth of your living word, in my Lord Jesus name.
I pray on the Breastplate of righteousness, please protect my vital organs and my inner man, cover my integrity, my spirit, and my soul. Guard my heart for it is the wellspring of life, please strengthen and guard the most vulnerable places in my life with that which is right, good, and noble that I might not receive a fatal blow from the enemy, in my Lord Jesus name.
I pray on the Gospel Shoes of Peace. I choose to stand in the shoes of your good news, and on the firm foundation of my Lord and Savior Jesus Christ, the solid eternal rock. All other ground is sinking sand, I pray that I will not slip or fall, but that my feet would be firmly fitted on your lordship, my Lord Jesus. I choose to stand on you, so that the peace of God, which transcends all understanding will guard my heart and mind in Christ Jesus, the eternal Rock of Ages. I receive your holy peace now my Lord, from the sole of my feet to the crown of my head, in my Lord Jesus name.
I pray the Shield of Faith into my hand now. As I take up the shield of faith, I ask that you might extinguish every dart and arrow, that is launched from the enemy to take me down spiritually, physically, mentally, emotionally, and every attempt of the enemy to destroy my joy. I ask that my faith in you would make it flame out. Extinguish every flaming arrow that would come against me, my life, my family, my home, or my ministry. May my faith always be out in front of me like a shield. Give me the courage to “faith my fears” by choosing to walk by faith and not by sight, in my Lord Jesus name.
I pray on the Helmet of Salvation, that you might protect my mind from the thoughts that can lead me astray. I choose to take every thought captive, and arrest all intentioned ideas and motives that would harm others, or distract me from your holy will for me. I submit every captured thought to the Lordship of my Lord Jesus Christ, and ask that you would imprison those thoughts that are not of you my Lord. Transform my mind and renew my thinking that I may think God thoughts, and have a sober mind that is focused on your glory. Please protect me from being double minded that I may allow my mind, I reject to live an earthly life, because I choose to live a holy one, governed by you My Lord Jesus, the prince of peace, please have my mind to be saturated with the holy mind of Christ, in my Lord Jesus name.
Finally, I take up the Sword of the Spirit which is the holy word of God, I pray this powerful offensive weapon into my hand, and ask that your holy word would be fitting for every encounter I face. As the enemy gets close to me, please give me the insight, wisdom, and skill to wield the word of God to drive away the enemy, in my Lord Jesus name.
May the enemy and his team flee from me, upon hearing the word of God spoken by the power and direction of the Holy Spirit. Give me the sword of the spirit to cut through the wiles of the devil, so that I may discern the schemes of the enemy when he is near.
With all kinds of prayers, supplication, and intercession I pray to you my Lord God as the one who fights my battles. Now that I’m in your holy powerful armor, I walk away covered and ready to face my day as you go before me, and please protect me in the midst of the spiritual warfare in this unseen world, in my Lord Jesus name.
Thank you my Lord, for the spiritual weapons of armor and prayer that you have given me. It is written no weapon formed against me shall prosper, and you will refute every tongue that accuses me.
Thank you Father God, my Lord Jesus and the Holy Spirit, that I am more than a conqueror in my Lord Jesus. I pray all of this in the mighty name of my Lord God and Savior Jesus Christ.
AMEN.
man trapped sounds awfully racist
😉
https://www.soundboard.com/sb/sound/76639
Chuck Norris lost his virginity before he was born.
Good stuff, Steve. Thanks for the update and the “dismal science” discussion. So, it appears there is a plan and that plan provides for obsolescence of all things. Hmmm….
SteveInCO
Thank you for a great lesson on steam. The Carnot engine and Newcomen are new to me.
50+ years ago, the student “sundries” and bookstore at the old Carnegie-Mellon Institute of Technology (long since morphed into Carnegie Mellon University) was called “Entropy.”
Ukraine: A new battle in the old war of the “New Normal” – The Burning Platform
Worthy read.
part of a thread…
Interesting TW from the guy who just bout ~9.2% of TW.
Indications TW censoring, banning, shadow banning taking its toll on TW Twats posting.
Looks like Elon IS going to shake things up in TWLand.
Impose Free Speech.
LEFTY World IS going to, in glorious fashion, melt down.
EdiT. “Most of these “top” accounts tweet rarely and post very little content.”
^^^ Pointing out, empty heads have little of value to say, post?
perhaps pointing out those numbers are fraudulent? Tweet is part of a thread, in another tweet Musk mentions most rarely tweet (hmmm … wonder if they are even around?) Tons of Bots on Twitter I’m sure
ABSOLUTELY. He’s showing the “effects of grooming”.
…and the ever vigilant twatter watchdogs who would demote/delete the wrongthink accounts regardless of followers and activity but the righthink accounts stay up forever
YUP. Never suspended. Never lost followers. Always protected.
Is he trying to clean up twitters act because they fear the future threat from Truth Social?
Haven’t a clue 😉 we shall see …
https://www.wsj.com/articles/china-is-accelerating-its-nuclear-buildup-over-rising-fears-of-u-s-conflict-11649509201
Information warfare.
https://t.me/TommyRobinsonNews/34446
short vid clip
Crowds gathering for Le Pen (never before for Macron)
Hopefully little MacSoyBoy’s draconian WuFlu lockdowns, mandates, passports will push her into victory this time.🙏
People of the World, RISE UP!! There are more of US than there are of THEM!!!
99% more!
Viva La France,
Vive Le Pen!!!
Ok, Abbott bombed on the Charter busses. Maybe he used that as a distraction from the RED MEAT? In a Trumpian move, he has slowed down transportation on 3 major bridges where Mexico trucks goods into Texas to a craaaaawwwl. He added extra inspection conditions to incoming trucks which now takes 45min/truck. Laredo is the HUGEST import route into the country. Bensman said on Warroom that a drone followed the waiting trucks for over 10 miles. 🤸♀️ US companies and Mexico are losing mega $$$ and are pizzled. Look for even more food and goods shortages. Abbott started this last week and expects to continue this coming week. THIS should get attention quick. 👀
This is EXCELLENT sleight of hand. The Bidenese commies prepared for Washington busing, and now they DIE AT THE BORDER, where they can do nothing.
FUCK JOE BIDEN.
A double whammy! Abbott hits both Biden and Mexico – trying to stop the illegal invasion and the bad policies.
I’d like a couple of tons of that, maybe with some maple syrup and caramel added to the mix 😀
Oh, and MOAR BACONNNNNNN!!!!
3, 2, 1… Dodge and HLS WILL conjure up some BS reasoning to stop slowing trucks into the US.
https://www.reuters.com/business/finance/russia-calls-integrating-brics-payment-systems-2022-04-09/
❤❤💓
Stash of assault rifles, body armor, passports with multiple visas, and sham uniforms found in penthouse of ‘fake’ Homeland agents – including one with ‘links to Pakistani intelligence’ – who ‘infiltrated Biden, Kamala and Jill’s Secret Service details’
By KATELYN CARALLE, U.S. POLITICAL REPORTER FOR DAILYMAIL.COM
PUBLISHED: 10:07 EDT, 8 April 2022 | UPDATED: 15:18 EDT, 8 April 2022
https://www.dailymail.co.uk/news/article-10700055/USSS-agents-Biden-Kamalas-probed-fake-DHS-agents-showered-gifts.html
Las Vegas anyone. Something similar timed with the J6 Kangaroo hearings.
Trying to gift the SS guy with a rifle could have been setting him up to be the patsy.
NOTHING would surprise me at this point.
Partying with the guy is perfect for setting up MK sessions.
Don Bongino said something about them gifting an apartment to an agent. He thinks it was likely bugged so they could get more intel. This appears to be something akin to the Arwan Brothers.
Oh, man – THAT is how you get a guy to help pull an assassination.
JFK case, Secret Service participation theory…….
Who’s behind this Op? Who busted them?
Who busted them? Guessing, pure dumb luck by Feds.
A postman got assaulted and the postal people thought the two might have witnessed it, the postal investigators must not have been happy with them and got on to the FBI.
The freakin’ POST OFFICE broke the case!!!
buggest own ga in history
seeing as post office is cabal stronghold
LOL! Yeah, that was a bit of a surprise.
Happen FJB just had a lucky break, they could have rid themselves of him and blamed it on extreme white nationalists.
???
Awan➡Wasserman-Schulz ➡DNC➡Nasty➡O’bummer
Hmmm… Interesting.
“Awan”… for some reason, popped into my brain when I first saw this story a few days ago.
This whole thing STINKS !
Ditto.
😉
Mo Ham Head Ali…
They could get jobs in Kalifornistan for the Highway Patrol or something 😡 😡 😡 😡
Could be the DEMONRATS are showing da bad guyz just how easy it is to get in…..
Patriots everywhere are rolling (and roiling) in their graves…..
Quote of the Day – Doug Ross – https://directorblue.blogspot.com
I think we’ve pretty much proven that DODGE and FIB are criminal arms of the UniParty communists.
Ø politicized and weaponized every agency of government.
There, FIFY, Jack…..
Yep!
Another win for 45!! ⛳⛳⛳🤸♀️🤸♀️🤸♀️
Judge: New York City Illegally Canceled Trump Contract (thegatewaypundit.com)
A state Supreme Court judge ruled Friday that the city did not have the right to cancel Trump’s contract to run the Ferry Point Golf Course. Mayor Bill de Blasio had nixed the deal after the Jan. 6, 2021, Capitol riot, arguing that Trump would not be able to attract golf tournaments.
In her decision and order filed on Friday, Justice Debra James said there was nothing in the contract that required a tournament — only that the city would share in any proceeds. That means either Trump gets to continue to run the course, or the city needs to pay him to leave. Trump’s son Eric said they’ll stay.
That decision by the Trumps will go well with de Blasio living in exile in some communist hellhole.
DAYUM!!!😍😍😍
WOAH: DeSantis Raises Over $100 Million For Re-election Campaign (thegatewaypundit.com)
EXCELLENT!!!
He’s fantastic. Happy to see him rolling in dough!!!
The Second American Civil War is Underway
Quote – “The confirmation of Ketanji Brown Jackson to Supreme Court is only historic in the sense that she is an illegitimate justice, nominated by an illegitimate occupant of the White House, and a nomination in violation of U.S. law prohibiting hiring people on the basis of race and gender. She is also a Groomer, friendly to pedophiles and child pornographers, and placed on the Court to normalize that behavior.”
MORE – https://www.thegatewaypundit.com/2022/04/lawrence-sellin-second-american-civil-war-underway/
Is it possible to actually know what has been and is going on in Ukraine? — Puppet Masters — Sott.net
Another great find! Thanks!
barker, would it be possible for you (or someone one else) to follow up this post from Sott.net, by posting Part 2???
Part two and three are in the original link — one is right after the badge graphic and the other is about a page or two past the map graphic.
cthulhu, Thanks!!! I immediately put this post in a file for later reading, not having read it.
Barker hit a gold-mine there. Lots of nuggets. The West Point article linked is a must read exposing the Nazis in Ukraine. They call them ‘right-wing’…so I guess the media and politicians all of a sudden support Nazis. Go figure.
TRUCKERS WERE RIGHT: Freedom Convoy protest was legal, judge says, and Ottawa is now being sued | FarmersForum.com
“The LPR forces have found a US-made radiation dosimeter in the liberated Ukrainian territories, supplied exclusively to American law enforcement agencies.
The LPR believes the equipment was abandoned by US instructors as the Ukrainian forces retreated.”
(With some photos.)
https://t.me/intelslava/24833
Biden Admin Prioritizes Climate Fantasies While Global Famine Looms (thefederalist.com)
EVERYTHING! wonder what the po’ folks are doing?Think gas is high?
Have ya been to the grocery store? Might prepare for a shock.
Chocolate now costs more than alcohol (even the good stuff) … and it’s
my drug of choice! So glad I prepped!
Fellow shopper told me her kids might get a few eggs, definitely no candy…
and price increase is 100% in some instances… and not just a few items…
^^^ REALITY, Northern NV. ^^^
I’ve been surviving on the big bags of chocolate chips at Costco. Last bag about 10 days ago still reasonably priced. But yeah, with hyper inflation no telling what to expect.
Cover all the bases: get alcohol-filled chocolates 😀 hic!
Yes, you are a hoot! I’ve already stocked up on the wine … think I’ll make it through 3 mos.
And then there’s Mr. Nezzer from Veggie Tales, with “The New and Improved Bunny Song” [originally Rack, Shack, and Benny, but now on the Silly Songs Countdowns] 😀 (whoever sings Mr. Nezzer is great…]….
Sort of a “case study” I guess? 😀 or barreling on through 🙃
Either way!
https://www.rt.com/russia/553599-zelensky-eu-leader-wants-proof-bucha-killings/?utm_source=rss&utm_medium=rss&utm_campaign=RSS
RT
EU leader wanted proof Bucha wasn’t staged – Zelensky
Ukrainian President Volodymyr Zelensky told journalists an EU head of state asked for proof that the Bucha killings weren’t staged
Forwarded from Intel Slava Z
https://s.w.org/images/core/emoji/13.1.0/svg/1f1ed-1f1fa.svghttps://s.w.org/images/core/emoji/13.1.0/svg/1f1fa-1f1e6.svg
Hungary backs an independent probe into what happened in the Ukrainian town of Bucha, the spokesman of Hungarian Prime Minister Viktor Orban says.
I don’t know how to post telgram here. But in the Intel Slave Z posts just saw a some Russian Soldiers in a small convoy going through a Ukr town. People out on the side of the road waving Russian Flags. Then near the end of the vid a young teenage girl flags the truck down and hands the soldier a colorful card. “Amen” written in English across the top. She had the nicest smile when she handed it to him. It was the most recent post.
https://t.me/s/intelslava
Para, this is link # https://t.me/intelslava/24843
It’s difficult to get the link# when there is a viv clip 😉 otherwise, with text, just click on the post anywhere and it will come up… T’gram is NOT user friendly… no dates on the posts and no #; most of their stuff has to be viewed thriu their app. I have Apple phone and they don’t allow T’gram apps! SMH…
Here’s more from Intelslava:
“More captured Ukrainian troops They took them in the city of Rubezhnoye. They make a pitiful sight. Thin, hungry, exhausted by fear and despair. By all indications and the first data from the interrogation, they belong to the soldiers of the National Guard.
They themselves, apparently, do not understand what they came out to fight for and what is happening in general. They don’t even know how the West uses Zelensky and all of Ukraine. These young soldiers will no longer be sent to the slaughter by the criminal authorities of the Nazis. Fortune turned to face them at the most critical moment, when death breathed in their faces. It was lucky that our soldiers, under the leadership of Apta Alaudinov, offered them to surrender.”
I hope and pray that all the innocent young (and old, of course) are able to get out from under Azov, Zelensky, and the rest of the WEFfenNaZis…
Someone once said, “War is old men sending young me off to die”.
Here’s hoping and praying that that war, and other wars (e.g. COVIDIOCY, WEF and the DEMONRATS versus TRUE PATRIOTS, etc.) end, and for GOD’S WILL and GOD’S GLORY…
Also the war of the clot shots. All those youngsters (OK less than 50-ers) (youngsters to me 🙂 ) dying IS TRAGIC, CRIMINAL, AND COMPLETELY UNNECESSARY!!!!! May Almighty GOD put and end to the clot-shots and their effects, and Heal those afflicted by them.
Praying along with you Cuppa…
Amen 🙏🏼
Thanks! See’s now if I open the images in a new tab I get the link to the image! I’m a happy camper now😁
One can also right click on the timestamp bottom r/h corner, then left click copy link.
As one scrolls through there is a divider for each day, I’d been going on the assumption that the posts were news that the author had received on that particular day.
I guess I move too quickly… thanks for the tip RAC… once in a while I encounter a channel with daily dates… it must be an “option”?
https://t.me/intelslava/24808
https://t.me/intelslava/24821
Published today, serial numbers.
Thanks RAC
The Cabal lies at every opportunity, and the Ukatranny is 100% Cabal.
AGREED. Don’t believe him.
Via IntelSlava Z
Fights in Mariupol are coming to an end
According to units of the DPR and the RF Armed Forces have reached the sea and are fighting in the port of Mariupol. Previously, it was under the control of the Ukrainian national battalions.
“We went to the sea. We are cleaning up,” one of the people’s militia fighters told us.
A significant part of the port is already in the hands of the allies. If the defense of the Ukrainian Nazis there finally collapses, then it will be possible to safely state the capture of Mariupol.
We are waiting for our victory.
views
telegram-cdn.org/file/o3B33S3w1IlxQubGAhj0rJSJdQNBSh6wZjs9dSKMFNP9s5ZZYgZGxCH9GfpzlE_L7uqKKiLd8AuhTprqxHY_nu3FkEoKF_41Mtj_TGv1QXUn60RGanw3oDY5kC-AqqHNsU3JAtj50FNzxWnSEbuHbjdqOf9bSYlkSRiVzjf-iCUtMeP2-CHGX75714E7ft_7tEuDGkZXyC_Mw394QkCH1eM2ApJIaq_n3pE6ByzrEIWR7FonXopa3yaJCkmNUGfdKZo480T2oOIx_wVtZK5ufOjH0QqTdpHs_T1OKkX-rGyk85hqQI3s5YmZAQ8Oq7RA2t-htgdTZYQ0KJg2lLA.jpg
https://www.rt.com/news/553568-psaki-nbc-journalists-concerned-credibility/
RT
NBC employees fear for brand integrity if Psaki hired – media
Reports of White House press secretary Jen Psaki’s hire at MSNBC has her potential colleagues worried about the appearance of impropriety
Pot meets Kettle.😂
Yeah, not sure what integrity they are talking about.
You can go blind and stir crazy looking for it. 😜
This is one more reason we know that Obama is running things. This was an OBAMA move, to send David Axelrod to NBC, to become the political “czar” behind the curtains.
RED JEN will be doing the same thing for him.
This is for the election. OBAMA is preparing for November.
The DemComs have some kind of a massive cheat, and they need an insider there to enforce perceptions after it goes down.
what’s up w/reports of employee concerns?
My guess is that the staff crawled out from under post-Axelrod “wokeism”, and they know that Red Jen is going to help re-institute (or provide cover for management to reinstitute) those same partisan politics as part of the editorial process. All their freedom gains will be lost, as she helps shift the culture back to DEMOCRAT.
Journalists understand that WE are stealing their jobs. Especially the ones who are picking up red pills and trying them out.
WE are beating mainstream journalists to the truth EVERY DAY.
NBC employees are going back under the PROGGIE WHIPS.
I hope they cancel her … (not gonna happen I know)
Would even be nice if the employees could get some guarantees…… but like you said….. not gonna happen.
We can dream 😉 (or pull the covers over our head… Nah!)
FIGHT!
FIGHT!
FIGHT! 😀
right! 😉
hussein has, will ensure Lying Red Jen has prominent role at NBC.
Forwarded from WhipLash347
Look up 1MBD – Malaysia – Goldman Sachs, Former Leaders of Australia and many others. 1MDB Scandal ties to all the horrors that will come out of Haiti. etc Scandal then read THE STORM by Martin Geddes it is in here. It lists the Scandals that will rip Planet Earth as you know it apart. Once you have you read this you will under why i say HUMPTY DUMPTY.
https://www.google.com.au/amp/s/www.bbc.com/news/world-asia-46341603.amp
https://www.justice.gov/opa/pr/over-1-billion-misappropriated-1mdb-funds-now-repatriated-malaysia
https://www.google.com.au/amp/s/www.aljazeera.com/amp/news/2021/8/16/malaysias-political-crisis-five-things-to-know
https://www.martingeddes.com/thestorm-how-to-prepare-for-a-global-corruption-purge/
https://www.martingeddes.com/wwg1wga-thestorm-and-qanon-red-pill-websites-now-available/
BBC News
1MDB: The playboys, PMs and partygoers around a global financial scandal
A wealth fund is alleged to have been looted of billions. The extraordinary story can be told through the characters caught up in it.
61
views
16:03
https://www.rt.com/news/553574-1mdb-banker-convicted-bribery-goldman/
RT
Verdict issued in ‘Wolf of Wall Street’ financier case
A former Goldman Sachs investment banker has been convicted for embezzling $4.5 billion from the Malaysian government
Greetings! and a quick ground report from South Alabama.Farmers are cutting their first Hay crop of the year in the field next door today.Must mean we have several days of fair weather ahead. 😃 I’ve been keeping myself immersed with outdoor Spring clean-up chores. Lots of downed limbs as a result of passing Gulf storms heading North. Last week on a drive to Pensacola ( a 200 mile round trip) fuel $3.77 per gal. for regular “in town”,when we crossed the Fla line the price was $4.11 per gal.reg This week it was $3.68 per gal. in town. I suppose the difference is a result of FJB robbing the strategic reserve eventually hitting the local market.FJB! 😉 Most notable: Dr.s office has removed their ‘mask’ requirements altogether. So nice to see complete facial expressions during conversations. Noticeable smiles and relief on entire staff. 😃
Latest words of wisdom/advice from Martin Geddes via my inbox..what timing !I hope it posts.
Remember! It’s OK to be wrong sometimes, we just have to have the humility too openly say so, then correct our mistakes as soon as we recognize them. 😉
Stay Vigilant
Hold the Line !
——————————————————————————————————————–Surviving Splitzkrieg
How lovingly ‘holding the line’ keeps us sane during insane times
Martin Geddes
Apr 9
In WW2 the Germans perfected the military doctrine of the lightning strike — “blitzkrieg” — applying overwhelming mechanised force to smash enemy defence lines. In March of 2020 we experienced the WW3 equivalent in terms of bio-information war — “splitzkrieg” — using a fear narrative to divide people into the “Covid compliant” and the resistance.
The wickedness had begun long before, infusing our society with wokeness, Cultural Marxism, and perverted ethics. The corrosion was intended to weaken our social and intimate bonds before initiating collapse, thereby enabling the theft of the whole planet from those with a legitimate birthright to inhabit it. This whole transhumanist nightmare of enslavement — with total control of money and mankind — is now juddering to a halt.
I have been watching the Q operation since its public debut in 2017, and how the “where we go one, we go all” ethos is designed to (eventually) reunite society and reverse the shattering impact of “splitzkrieg”. The long-awaited moment of mass awakening — with endless megascandals breaking — is tantalisingly near as “The Bidan Show” finally wraps up. Yet we must continue living in the present, not knowing on which day our loved ones will “wake up” from the black magic spell cast upon them.
My own life faces many “splitz” from the “krieg”. I have found myself over and over caught up in a process of defining my own locus of responsibility, so that I am neither involved in coercion, nor being coerced. This “holding the line” means managing my own wellbeing, while simultaneously not conceding to evil, yet also relating to those who behave badly from a loving place. The times I have strayed from the path of righteousness — and “broken the line” — only reinforce the value of staying on the path.
This “holding the line” at the personal level is difficult in a context of pervasive persecution. We face simultaneous psychological war, economic war, biological war, cultural war, and even genocide. At an individual level those who dissent from the mass psychosis suffer isolation, ostracism, and slander. “Splitzkrieg” heaps injustices upon those who act according to holy principles, and resolution is not available on the impatient timescale of the victim.
One of my key “Splitzkrieg” learnings is that “Satan” is actually a role — of the accuser or opponent — and my #1 enemy is the speaking of untruths against me behind my back. These gossiping cowards refuse to confront me, and deny me any due evidential process of natural justice. They have taken up the power to define my reality for themselves, and exclude any possibility that they might be doing me a terrible wrong — as a consequence of their actions based on accepting lies.
“Splitzkrieg” is the end game of Satanic war: we must “think mirror” because the enemy has to invert everything in order to keep the “lockstep” pseudoreality internally consistent. Fact and fiction, right and wrong — they are swapped over and over. Moral relativism is an enemy because the self-image of being righteous enables people to self-justify all kinds of wrongdoing. This leads to the spread of totalitarian ideology, resting on false morality and false logic, which divides society into True Believers™ and the perplexed.
Whether our loved ones have been deceived, hypnotised, poisoned, demonised, or even hijacked with nanotech doesn’t matter. We have to confront the reality of “splitzkrieg” — where we have to “hold the line” even when our most intimate spaces have been invaded by unseen enemy forces. For the battlefront of this war can run down the middle of the marital bed: it is just not sexy for men to fail to protect children at risk! One of the hardest sights to see is when someone has found the path, understood that there is a holy war on, and then failed to fight.
I have many stories of having to “hold the line”, and am learning “personal splitzkrieg” from each encounter:
In each case I have had to “locate the line” before I could hold it — where “the line” is the geometry of the spiritual and social split. What is genuinely my concern versus theirs? Is one party projecting — and if so who? Have I got some triangulation on the situation that is not merely confirmation bias? Most of all, am I dealing with everyday lapses of good manners, entrenched foolishness, or psychopathic evil?
Shockey’s Law [ref — PPT] states that “‘Money’ is the answer — now what is your question?”. That cynical truism describes “living in Babylon”, but perhaps we can flip it to a more benevolent outlook? I cannot put my own name to it as a Law, as I suspect this is already taken by past prophets: “‘Love’ is the answer — now what is your question?”. However, the loving thing to do with each split due to psychological and spiritual warfare is not always obvious.
It is tempting to see everything as either an offer of love or a call for love, even terrible behaviour, and to try to cross the divide and heal the split. If we are not careful that can make us complicit: real evil has to be extinguished, not negotiated with. For all lesser splits we must act in love — a unifying force — as it is the only possible antidote to “splitzkrieg”. Insisting on accountability, for instance, may superficially appear harsh and uncaring, but “tough love” is far more loving than floppy accommodation of sloth.
I am finding that I have to prioritise my efforts in this “social fragmentation bomb” conflict thus: firstly, peace in my own heart; then, in my own home; next, in my wider community including family; and lastly, peace in the world at large. In the context of the “jabicide”, I am slowly accepting that death is an essential part of a genetic and cultural clean-up process. Their passing, however sad, doesn’t change the peace I have in my heart and home.
I have to welcome the likelihood that loved ones may become stark warnings rather than shining examples: my control over their free will is pointless at best. Many relationships are anchored to a particular place and time, and the shifting foundations of society mean that basis of connection is no longer relevant. I am even rethinking what I consider to be family: it is people who both love us for who we are, and practise the “no man left behind” doctrine.
“Splitzkrieg” has already divided humanity into the “organic” and “GMO” varieties. This may not be reversible or healable, and it may be necessary to grieve in ways we never have had to grieve before. For me the consolation has been to see all kids as our kids — so my own “warrior mode” can be activated wherever a predator seeks to pick off one of the tribe. I have a clear purpose anchored in a life-giving force, which gives me comfort.
If we put truth, light, and love at the heart of everything we do — seeking the highest morality despite our many lapses — then there is something of lasting value to salvage from the nightmare we have been living through. Only a reinvigorated striving towards Oneness can ultimately give meaning to the cruel horrors of bio-information “splitzkrieg”.
That’s an excellent lesson in setting appropriate boundaries.
Moderna recalls 764,900 doses of contaminated vaccine –
https://www.theepochtimes.com/moderna-recalls-764900-covid-19-vaccine-doses-after-contamination-found_4394113.html
And that somebody will be guest speaker at tonight’s Trump Rally! 😁
LOL! Nice.
Hi y’all! Happy Rally Night!!!! 💥
Remember when we were being told that there are no Nazis fighting for Ukraine against Russia…that it is a ‘right-wing’ conspiracy theory?
Well I guess West Point Academy’s Center for Counterterrorism must be a bunch of conspiracy theorists too.
Ironically, who is it now supporting the far-right extremist Nazis in Ukraine? You cannot make this stuff up. 😂 😂 😂
From April 2020 they write:
“Drawing on nearly a dozen reporting trips to Ukraine between 2014 and 2019, Tim Lister examines the nexus between far-right extremists in Ukraine and the United States. He writes: “In recent years, some Americans and Europeans drawn to various brands of far-right nationalism have looked to Ukraine as their field of dreams: a country with a well-established, trained, and equipped far-right militia … that has been actively engaged in the conflict against Russian-backed separatists.” He notes that in some instances, “U.S.-based individuals have spoken or written about how the training available in Ukraine might assist them and others in their paramilitary-style activities at home.”
https://ctc.usma.edu/wp-content/uploads/2020/04/CTC-SENTINEL-042020.pdf
The article starts on page 30.
She thinks she’s looking at the front of the tank….
(yep, she’s that incompetent)…
She says if Ukraine wants the weapons then they have to pay for them. 😮😆🤣😂🤣
In Rubles 😆
I reached my limit the moment they announced the first $450 million weapons shipment. After 5 weeks we’re now at over $2 BILLION. It makes my blood boil. Where are they getting that money from? Taking from our Defense budget? That’s the only reason Creepy submitted a budget increase for DoD, they’re planning on dragging this out. Dems ALWAYS slash the Defense budget. They wouldn’t even let 45 reallocate DoD money for the wall!!! More concerned about Ukraine’s borders than US borders!?!?! Who authorized the expenditure from what budget? Doesn’t Congress control the purse and where it gets spent? Haven’t heard them passing any new bills for Ukraine weapons. Who voted for that, NOT ME!!! And they’re talking about this dragging out for a long time????? EFF THAT, NO MORE DAMN MONEY TO UKRAINE!!!! NO MORE WUFLU MONEY!!! Inflation will never come down if we keep spending MOAR money!!! Stoopid Groomers, ARGH!!! 🤬 🤬 🤬 🤬 🤬
Sorry for the rant PR, Luv U🤗😘🥰😍💞💞…….Mooching German cheepskates set me off. Europe wants the war, they can bloody well pay for it.🤑🤑🤑
Who authorized the expenditure from what budget?
^^^ U N I P A R T Y. To the tune of ~$14 BILLION. ^^^
Two or three weeks ago.
IIRC.
$14?!?!?!?!?!?!?!!!😱😱😱🥵🤢🤮
Yes, SADLY, AND I do mean, SADLY.
When folks trash Dumrats, I increasingly TRASH UNIPARTY. .
FTA
Biden Signs $1.5 Trillion Spending Bill With $14 Billion For Ukraine Aid—Here’s What’s In It
https://www.forbes.com/sites/jonathanponciano/2022/03/15/biden-signs-15-trillion-spending-bill-with-14-billion-for-ukraine-aid-heres-whats-in-it/?sh=9a467d622149
14 Billion to Ukraine means 14 million back to Democrats.
Probably a cut for the RINOs in there, too.
Always 10% for the Big Guy.
That was in the quickie overnight bill? I missed that. Those evil mothertruckers!! We need to get in the grills of every last one of them.
Thanks for educating me K-Bo. 🤗😢
Just like all the quickie overnight bills…..
We still don’t respect the DEMONRATS and the RINOs in the morning 😀 😡 😡 😡 😡
Agree… enjoyed the rant!
Sorry, who’s mooching?
Not the folks here in Germany. We’re paying for this twice. We’ve paid with weapons and other things (which probably don’t work; our military is a mess), and we’ve already taken in ONE MILLION refugees (REAL refugees, at least) and Baerbock is expecting up to TEN MILLION, total… THAT in a country with just over 80 million people (and who knows how many “Syrian” refaugees)….
The only good thing is that the Ukrainian refugees are overwhelmingly Christian, as opposed to those coming from Merde-Kuh’s “favored lands”… And, they are mostly women and children, also a stark contrast from the Moslem invaders…
Since there is no requirement (at present) for Ukrainians to have visas, etc., and many other requirements have been relaxed, there is no real idea of how many have actually entered Germany. So far, Poland has borne the brunt of the crisis, with an extimated three million refugees so far. Sadly, there was a young Ukraininan woman who was brutally raped here in Germany by a Mozzie refauxgee. She returned to Poland from Germany after that. Sad that she was one who TRULY needed help and that animal refauxgee ended up taking both the help and possibly her future away from her… The legacy of Merde-Kuh lives on 😡 😡 😡 😡
The Moslem refauxgees are now complaining about being “second-class refauxgees”… Maybe they should quit their crimes and not try to make Germany like the hellholes they left…..
(It could also be a sneaky way for Kanzler Scholz to get rid of our defective equipment… most of our stuff doesn’t work (thanks, not, von der Leyen). Of course we’d be left with nothing, but that might be an improvement in terms of getting stuff that works… and an odd sort of net gain for Russia…shades of the Polish MIGs debacle)….
I think the WEFfen are the only ones gaining, as usual… wealth redistribution, once again…
Okay … that ^^^^ is Biden…
NOT those old men with dementia stumbling around
Yes, a simple facelift does not make a man look like a completely different person. (as opposed to the Kardashian regimen)
Kenny Rogers
Wayne Newton
I did say a facelift, singular. Not repeated surgeries, cheek and jaw fillers, botox, and whatever else is going on with Wayne Newton. And didn’t Kenny Rogers speak publicly about regretting all the work he had done? Unfortunate.
The Biden photo looks to be maybe 15+ years ago. Looks like a different person to me. Just an opinion.
According to plastic surgeons, Biden has not had just a simple facelift but probably has had lots of things done: hairline, filler, Botox, teeth implants, facelift…(See the article I posted below.). And he has done some of that repeatedly.
Yes, Kenny Rogers has said he regrets having the work done.
IMO, Biden does look different, but not so different that it isn’t him. Wayne Newton looks very different, but it is the same man.
Kenny Rogers died two years ago.
WOW! I had never seen it that clearly before.
HUNK TO HIGH OFFICE Joe Biden looks unrecognizable before taking office after plastic surgeons point to multiple cosmetic procedures
https://www.the-sun.com/news/4480500/joe-biden-looks-unrecognizable-plastic-surgery-cosmetics/
Applies to schools and teachers also…
Do you know who is teaching your child? What textbooks they have? And daycare? Have you vetted them, talked to other parents?
MED BEDS ARE AVAILABLE
https://t.me/s/Whiplash347/104132
I’d like to know more about those. For $20,000 you can get a Tesla MedBed generator to help with sleep!
Tesla MedBed Generators $19,999.0
https://www.teslabiohealing.com/products/tesla-medbed-generators
This is a very smart scam. It’s a brilliant marriage of traditional Chinese ideas about “chi” or “qi” or however they spell it, and the allure and mystery of Nikola Tesla, augmented by the success of Elon Musk’s ventures under the name, which have given “Tesla science” cultural validity as an idea.
The scammers are very careful to skirt outside of the law, and the
BidenChina Joe FDA is unable to do anything to people who so artfully deny that their product works, using all the right language.A lot of elderly Chinese fall for this, I’m sure.
TRUMP IS IN THE HOUSE!!!!
RALLY THREAD!!!!
I just listened to the Trump rally, which was mostly So Wonderful, except for one big problem. Pres. Trump is endorsing Dr. Oz for Pennsylvania U.S. senator, and Oz says he will give up his TURKISH citizenship If he’s elected. You’ve got to be kidding. Besides which, he’s not very conservative. Besides which, I hope someone can research if he has any connections with the Gulen schools, which I think started in Pennsylvania. I also would not want a former hedge-fund exec. If elected, Oz will also be the richest person in the senate, and he has hobnobbed with dems. I would vote for Kathy Barnett, and if I lived in Pennsylvania, I’d be trying hard to help her win the primary. Welll, I got that off my chest. It just upset for me a fantastic rally. Pres. Trump, I wish you would reconsider.
Trump’s guy Sean Parnell had to drop from the race. I see Parnell was endorsing David McCormick but the polls must look not that good for McCormick? And the race is being considered a bell weather for the Democrats. It’s likely Trump’s choice is more strategic/realistic than ideal here.
Agreed. Oz is optimal right now – not perfect.
I understand your concerns and have not thought Dr. Oz was very conservative. It might be that PDJT is endorsing the person he thinks has the best chance of winning, due to name recognition — and of course that Oz would vote the right way on a lot of issues. Trump himself is rich and used to hobnob with Dems. I think that can work both ways. 😊 Maybe Trump sees some things they have in common.
Steve Bannon used to be an investment broker at Goldman Sachs.
Trump wants to end the insanity in the House and Senate. Oz will win and help do that. It’s a chess play.
We agree…was at a rally watching gettogether. When he said that we all looked at each other like ‘are we crazy, did he just give full support to Oz’…I think it’s a bad choice
Hes getting bad endorsement advice. Not the first one and he endors3d a bunch of rinos then lamented before.
His connection at rallies vs these decisions dont make sense.
If Oz turns into a capricious, self-centered RINO, you will be proven correct! We had best hope that Oz follows through on all promises.
Watch what Oz does if he wins. If he backtracks on giving up Turkish citizenship, the bad choice will be evident. If he follows through on it, though, he may have been a better choice than we realized.
Molly, thank you for saying it. Yes, very bad choice and both Oz and McCormick o are trans-friendly, too and nothing is worth voting for someone who is a Turkish citizen and says he will renounce it If he’s elected. Politics and money, I hate it.
Politics.
Thought I read Oz, quit a political race. Now he’s endorsed for Senator. Hmmm.
From bb comments:
It’s just about over LetJusticePrevail116 minutes ago
“Trump likes to be on the side of winners and he has been convinced by pollsters and other assorted wizards of smart that Oz can win so he’s backing him. He fails to realize that if we had all followed that pattern he never would have been the nominee and then president. He needs to start thinking outside the box again, I don’t like this swampy direction.”
I’m just kind of disgusted. I like Kathy Barnett, she is the real conservative, and I didn’t mean Oz had been a hedgefund exec, that is McCormick, and both he na and Oz are libs, wolves in sheep’s clothing, though McCormick is for fracking. I just hope hope something happens to help Barnett.
It seems like there should be a political tie-in with this, but I just can’t finger it out. At any rate, it’s still Caturday.
Oops!
Don’t ya hate it when that happens ?!
I’m suspicious of the Google involvement in preventing the image from showing. **shifts eyes left** **shifts eyes right**
If the image will download to your computer, you can always upload it with your comment using the little picture icon in the lower right corner. That works better than the URL sometimes.
Thanks. I wasn’t aware of that.
P F M…
Couple weeks ago, I posted Worldometers dropped Active USA Covid ~four million over night.
Yesterday, the USA was at ~14 million Active Covid.
Earlier today, USA ~11million Active Covid.
Now, USA has 9,369,891 Active Covid.
Now that’s interesting.
CTH
BRICS Ministers of Finance Hold a Meeting – It Is Time to Replace Western Financial Trade Mechanisms and Remove The Dollar
This is not some grand conspiracy, ‘out there‘ deep geopolitical possibility, or foreboding likelihood as an outcome of short-sighted western emotion. No, this is just a predictable outcome from western created events that pushed specific countries to a natural conclusion based on their best interests.
You can debate the motives of the western leaders who structured the sanctions against Russia, and whether they knew the outcome would happen as a consequence of their effort, but the outcome was never really in doubt. Personally, I believe this outcome is what the west intended. The people inside the World Economic Forum are not stupid – ideological, yes, but not stupid. They knew this would happen.
https://theconservativetreehouse.com/blog/2022/04/09/brics-ministers-of-finance-hold-a-meeting-it-is-time-to-replace-western-financial-trade-mechanisms-and-remove-the-dollar/#more-231465
This is kinda of heady stuff to think about, but we are seeing the parallel system on a world scale being implemented in a roughly east west divide. Just thinking, they keep talking about what makes the American dollar so strong is we follow the follow the rule of law. Unfortunately we see that as being less true than true at times. Wonders if that will make a difference on which one survives as Lavrov has been making the same noise saying the US is no longer following the rule of law?
We need more work on our parallel system, Gab, Parler, and sharing garden secrets with the neighbors might not cut it. 🙃
Australia admitting the Injections are NOT safe…
https://twitter.com/CiceroConsulta1/status/1512917167011885062
FTA
Life expectancy falls yet again in the United States
Americans are living shorter and shorter lives. Life expectancy fell in 2021 in the United States — but not the rest of the world. Lifespans fell by an average 0.39 years in America, even as lifespans rose in the rest of the world by 0.28 years in 2021, reports the Washington Post. Life expectancy fell more for whites than for blacks and Hispanics. The average U.S. life expectancy is now 76.6 years.
The fall in U.S. life expectancy is particularly striking, because observers had assumed life expectancy could only go up after dropping in 2020 due to the outbreak of the coronavirus epidemic, and then bottoming out in 2021 due to new coronavirus vaccines. A public health physician said he had “assumed the newly approved vaccines would reverse the overall downward trend,” and had been “naively optimistic” that lifespans would “rebound” in 2021 — which has not happened.
The Washington Post notes that the “total US decline in 2021 of .39 years is larger than the decline among Whites of .34. The difference is likely caused by lower life expectancy among Native Americans and Alaska Natives,” which likely declined faster than for other racial groups. “U.S. men suffered a larger decline than women, widening the difference in life expectancy from just over five years in 2019 to just under six years in 2021.”
https://libertyunyielding.com/2022/04/07/life-expectancy-falls-yet-again-in-the-united-states/
Sounds like the vaccines.
https://twitter.com/elonmusk/status/1512966135423066116?s=20&t=pYvtcNq4GdGjr8BmjEG37w
HOLY SHEET – this dude is raising a RUCKUS.
HEE HEE 😂
Twitler needed a good shaking up – and it’s getting it!
Nah, he’s just getting warmed up.
When he gets rolling, he does, “the old rules don’t apply anymore.”
When he’s HOT, he does, “the old rules never applied the way you thought they did.”
E.G. — “We don’t have to rely on NASA any more”….morphing into “Heinlein had better ideas for space launches than NASA did.”
Also, “We no longer have to stay in range of a nation-state-style geosynchronous satellite”….morphing into “we’ll just shift a few Starlink sats over Ukraine.”
Elon just launched a paid private trip to the ISS. It was the first stage’s fifth trip, and it landed upright on its barge just like God and RAH meant it to. This is the third excursion to the ISS for the Crew Dragon Endeavour capsule. Everything is modular, capable, and reusable.
Obviously the ideal here would be a space elevator…which, of course can’t be built until we get to space cheaply, so to get there we have to fall back on something less ideal. And the best of THAT world would be a single stage to orbit that just needs to be refueled and checked for mechanical problems and put through a maintenance regimen…like an airliner.
If you can’t do that, then something that disassembles but can be reassembled readily is best. No need to buy a new whatsis for every flight. The Shuttle was only partially reusable; the solid rockets had to be practically rebuilt every time and the big external tank was expended. The shuttle itself, the most reusable item, still took MONTHS of expensive work to turn around because those heat tiles needed constant care.
We’re getting there. Slowly, slowly, but we’re getting there.