Another week, another deluge of BS from the White House and from the Controlled Opposition.
The Audit is over, now the spin doctoring begins. Other efforts are afoot in other states. Good. The more the merrier.
The collapse of the Covidschina continues.
No doubt much will be said about those today. (And I have missed a lot this past week.)
To my mind the audits are the last hope for a within-the-system fix to what happened last November. “Within the system” meaning the audits find fraud, the various states decertify the results, and some dang judge rules that Biden must step down and Trump must be installed.
That last step is crucial. The way our system works, “fraud” isn’t a fact until some “competent authority” (i.e., meaning “one that has jurisdiction,” not “one that won’t end up with an ice cream cone on its forehead”) rules it is so. That must happen before the system will accept that the election is vitiated by fraud. No finding of fraud means, as far as they are concerned no fraud, no fraud means nothing vitiated. We sit and fume, because the system has failed.
I’ll leave it to you to decide how likely you think it is that a judge will rule against the Left given the riots that would likely endanger his/her family.
As for the military stepping forward and doing the job instead? Well, that’s technically “outside of the system” and besides…this military, that’s being made woke as we speak?
What do we do in the likely event that fraud is found, but no judge will find it to be “fact” as far as the Federal Government is concerned? I keep hoping someone will come up with a suggestion, and so far “general strike” (H/T Scott) is the only one I’ve seen.
Justice Must Be Done.
The prior election must be acknowledged as fraudulent, and steps must be taken to prosecute the fraudsters and restore integrity to the system. (This doesn’t necessarily include deposing Joe and Hoe and putting Trump where he belongs, but it would certainly be a lot easier to fix our broken electoral system with the right people in charge.)
Nothing else matters at this point. Talking about trying again in 2022 or 2024 is pointless otherwise. Which is not to say one must never talk about this, but rather that one must account for this in ones planning; if fixing the fraud in the system is not part of the plan, you have no plan.
This will necessarily be piecemeal, state by state, which is why I am encouraged by those states working to change their laws to alleviate the fraud both via computer and via bogus voters. If enough states do that we might end up with a working majority in Congress and that would be something Trump never really had.
Lawyer Appeasement Section
OK now for the fine print.
This is the WQTH Daily Thread. You know the drill. There’s no Poltical correctness, but civility is a requirement. There are Important Guidelines, here, with an addendum on 20191110.
We have a new board – called The U Tree – where people can take each other to the woodshed without fear of censorship or moderation.
And remember Wheatie’s Rules:
1. No food fights
2. No running with scissors.
3. If you bring snacks, bring enough for everyone.
4. Zeroth rule of gun safety: Don’t let the government get your guns.
5. Rule one of gun safety: The gun is always loaded.
5a. If you actually want the gun to be loaded, like because you’re checking out a bump in the night, then it’s empty.
6. Rule two of gun safety: Never point the gun at anything you’re not willing to destroy.
7. Rule three: Keep your finger off the trigger until ready to fire.
8. Rule the fourth: Be sure of your target and what is behind it.
(Hmm a few extras seem to have crept in.)
Spot Prices
Last week:
Gold $1751.20
Silver $22.49
Platinum $986.00
Palladium $2047.00
Rhodium $15,750.00
This week, 3 PM MT on Friday, markets closed for the weekend
Gold $1762.00
Silver $22.65
Platinum $981.00
Palladium $2000.00
Rhodium $14,050.00
Minor shifts in almost everything. Gold and silver up a bit, the PGMs down a bit. I, as always, intend to hold.
Part XX – The Little Neutral One
Let us start off by recapping our list of “as of 1894” mysteries and conservation laws, and bring things up to date including the neutron.
Conservation of mass- Conservation of momentum
Conservation of energy- Conservation of electric charge
- Conservation of angular momentum
- (ADD:) Conservation of mass-energy
The following mysteries were unanswered at the end of 1894. I’ve crossed out the ones that have been answered up to this installment.
Why was the long axis of Mercury’s orbit precessing more than expected, by 43 arcseconds every century? Was it, indeed, a planet even closer to the sun? If so, it’d have been nice to actually see it.Why was Michelson unable to measure any difference in speed of light despite the fact we, being on planet Earth that is orbiting the sun, had to be moving through the medium in which it propagates?- What makes the sun (and other stars) shine (beyond the obvious “they shine because they’re hot” answer). What keeps the sun hot, what energy is it harnessing?
- How did the solar system form? Any answer to this must account for how the planets, only a tiny fraction of the mass of the solar system, ended up with the vast majority of the angular momentum in the system.
What is the electrical “fluid” that moves around when there is an electric current, and that somehow seems imbalanced when we perceive that an object has a charge? Were there both negative and positive fluids, or just one fluid that had a natural neutral level; below it was negative (deficit), above it was positive (excess)?Why are there so many different kinds of atoms? How did electrical charges relate to chemistry? How is it that 94 thousand coulombs of charge are needed to bust apart certain molecules (though it often had to be delivered at different voltages depending on the molecule)?Why were the atomic weights almost always a multiple of hydrogen’s? Why was it never quite a perfect multiple? Why was it sometimes nowhere near to being a multiple?Why does the photoelectric effect work the way it does, where it depends on the frequency of the light hitting the object, not the intensity?Why does black body radiation have a “hump” in its frequency graph?
As of 1930, we had a notion of the rough answer to #3, thanks to Arthur Eddington. I hinted at it last time. But details still needed to be worked out.
Number 4 was still a mystery back then, as far as I know.
Recaps and Refreshers
There are some preliminaries to get out of the way here, some of them are refreshers on what came before. Back in part 17, when I told the story of the discovery of the neutron, I brought in the concept of “spin” in regards to electrons and protons. But it’s not spinning like a top, it’s something else, still measured as angular momentum. But apparently one “rotation” doesn’t bring the particle back to where it was before, it’s somehow upside down, and another rotation is needed to get it back to where it was. (Yes, that does not make sense to us here in our macroscopic world.) It takes 720 degrees of rotation, not 360, to put the particle back the way it was. (And yes, that doesn’t make sense.)
As I said, it does get measured in units of angular momentum, and Planck’s constant, h, has the same dimensions as angular momentum. Angular momentum can be thought of in terms of whole revolutions of whatever is spinning, or in rotations through an angle of one radian (which is preferred), so Planck’s constant is often divided by 2π to give a “reduced Planck’s constant” called ℏ (pronounced “h-bar”).
It turns out that electrons and protons have a spin of 1/2ℏ. Physicists will often drop the ℏ and just say that electrons and protons have “spin 1/2”. Two electrons, side by side, might be oriented the same way, or in opposite ways, in which case one of the electrons is assigned +1/2 spin and the other one is “upside down” and has spin -1/2. Similarly for protons. And neutrons. All have +/- 1/2 spins.
Like angular momentum, spin is expected to be conserved, because it is a funky form of angular momentum.
If you have many protons (and neutrons) in a nucleus, the nucleus itself has a total spin, which is just the sum of all those half spins. The combined number of protons and neutrons is the atomic mass number, so for an atom of nitrogen-14, there are seven protons (because it’s nitrogen, and nitrogen by definition has seven protons) and seven neutrons, total 14. So the spins of seven protons and seven neutrons have to be added up. When actually measured, the total spin is 1.
It turns out there’s a rule here: If the mass number is even, the total spin is an integer. If it’s odd, the total spin has a 1/2 (or -1/2) in it. This makes sense, if you think about it. You can go through the protons and neutrons in a nucleus and group them, arbitrarily into pairs. Each pair will consist of two +1/2 spins (total 1), a +1/2 and a -1/2 spin (total 0), or two -1/2 spins (total -1). And it doesn’t matter which protons and neutrons you arbitrarily choose to pair together. The result is that all of the pairs put together will make up a whole number spin since you’re adding 1s, 0s, and -1s. So if the nucleus has an even mass number, its spin is the sum of a bunch of pairs of nucleons and will be an integer. If it has an odd mass number, there will be one proton (or neutron) left over after you make up your pairs, again no matter which pairings you use, you have one left over. It will have spin -1/2 or +1/2, so you’ll end up adding or subtracting 1/2 from the integer spin you get from the pairs. In general, a spin with a 1/2 in it is called a half-integer spin, because when you double it, you get an integer.
Before the discovery of the neutron in 1932, there was an idea that a nucleus consisted of protons–as many as the mass number–and some electrons to cancel the charges on some protons. So the nitrogen-14 nucleus would have 14 protons and 7 electrons, leaving a total charge of 7, and that total charge made it a nitrogen nucleus. On one level (considering electrical charge) this makes sense, but it turns out to make no sense at all when considering spin. That (hypothetical) nucleus would have 21 particles in it, all with half spins (remember that electrons too have a half spin), so it should have a total spin with a 1/2 in it, a half integer spin. Yet the nitrogen-14 nucleus, as I mentioned earlier, had a measured spin of 1. That was a powerful argument used as support for the existence of a hypothetical “neutron” and indeed the discovery of the neutron made the math work out; now there were an even number of particles in the nucleus so it could have an integer total spin. However, as we shall see below, this solved one problem but left another problem in place.
When the anti-electron or positron was discovered, it turned out to have the opposite spin of an electron. Even when oriented the same way as an electron, its spin was -1/2 compared to the electron’s 1/2. And this is true of anti-protons and anti-neutrons as well; in fact the difference in spin is the only obvious difference between a neutron and an anti-neutron (but it’s enough!).
In 1925, Wolfgang Pauli enunciated the “Pauli exclusion principle.” At first he applied it only to electrons, but in 1940 it was generalized to all particles with half-integer spins. In short, it states that no two such particles can occupy the same quantum state. An example of this is the lowest energy level of an atom, the “1s” orbital. An electron in that orbital is in a certain quantum state. But spin is part of the quantum state, so you can put a second electron in that orbital, so long as it’s oriented the other way and has spin -1/2. But after that, no more. You have to put a third and fourth electron in the “2s” orbital, then six subsequent electrons into the three “2p” oribtals (two each), and so on. (This is why the periodic table “rows” (or periods) all have even numbers of elements in them; if you subdivide them into blocks corresponding to the s, p, and d orbitals, those blocks also each have even numbers of elements in them.)
This is fundamentally the reason why two material objects can’t be in the same place at the same time. They’re made up of electrons, protons, and neutrons with half spins.
This principle does not apply to particles with integer spins, like photons (spin 0). Photons can pile onto the same quantum state by the billions, and it’s no problem. That’s very “non-matter” behavior, and indeed photons aren’t considered to be “matter” as we know it. They can occupy the same place at the same time, and often do. Beams of light can cross through each other without a problem.
Eventually, the name “fermion” (for Enrico Fermi) was given to all half-integer spin particles as a class, and “boson” (For Satyendra Nath Bose) to particles with integer spin.
OK, that’s enough about spin (for now).
I’ve also already told the story of how physicists had discovered the “Strong Nuclear Force,” often just called the Strong force. It’s responsible for keeping nuclei together, and when it’s just not strong enough to do the job, you get alpha decay, where the nucleus ejects an alpha particle (after 1932, known to consist of two protons and two neutrons), total four mass units. When this happens to a uranium-238 atom, it becomes a thorium-234 atom; four mass units less. And the charge has decreased by two, from 92 (uranium) to 90 (thorium).
It was beginning to look, by the way, as if the total number of nucleons (be they protons or neutrons) was something that was conserved, a new conservation law. Eventually this would be called “conservation of baryon number” (other similar particles would be discovered after 1950, they ware all called baryons). Protons and neutrons each had a baryon number of 1. The only way to wipe them completely out was to hit them with antimatter, but the antimatter was regarded as having negative baryon numbers, an anti-proton or anti-neutron had baryon number of -1. So proton-anti-proton annihilation took a +1 and -1 and turned them into zero. Nice and tidy.
Beta Decay Spells Doom?
But there was another kind of radioactive decay, beta decay.
And it was causing headaches for physicists.
As a reminder, this is when a nucleus spits out an electron. The charge of the nucleus goes up by one, but its mass number stays the same. For example, that thorium-234 atom undergoes beta decay to become protactinium-234. Same mass number, but the charge has changed from 90 (thorium) to 91 (protactinium). That process repeats to get you to uranium-234.
The old notion of the nucleus as consisting of one proton per mass number, counterbalanced partially by electrons, seemed plausible because of this. One of those electrons could be kicked out, “uncovering” a proton and increasing the charge. But as I described above, this notion of electrons in the nucleus left an issue with spin. This was solved with the discovery that the nucleus in fact contained protons and neutrons, and no electrons, but there was still a problem with beta decay.
That thorium-234 nucleus has to have an integer spin (just like the nitrogen-14 nucleus) because 234 is an even number, and the discovery of the neutron explained why. (According to wikipedia the spin happens to be 0.) The resulting protactinium-234 nucleus similarly has to have an integer spin (also happening to be zero). But in moving from thorium-234 to protactinium-234, an electron was ejected; it has a spin of 1/2. Shouldn’t the resulting nucleus have a half spin as well?
It doesn’t. So it appears that beta decay violates conservation of angular momentum.
Another issue popped up when energy was measured. With alpha decay, the masses (considered as their energy equivalents) and kinetic energy of the particles before and after the event balanced, once you added everything up, including the recoil of the nucleus like a rifle firing a bullet.
With beta decay, some of the energy disappeared. The energy of the nucleus (including its recoil) plus the kinetic energy of the electron, did not add up to what was there before. There was always some missing energy, but the amount could vary from very tiny to most of it.
This of course looked very much like a violation of mass/energy conservation.
[I’ll pause here to note that physicists typically simply speak of “energy conservation”, not “mass/energy conservation” because they consider matter to just be another form of energy; so a change in mass of a nucleus due to binding energy and so forth, is just another change of energy to them. I’m going to follow that convention from here on out.]
Yet another issue was noted when the recoil of beta decay was considered. With alpha decay, the alpha particle and nucleus recoiled in exactly opposite directions, much like a cannon firing a cannonball is shoved back in the opposite direction. The two new momenta (a relatively light alpha particle traveling quickly, versus a relatively heavy nucleus traveling in the opposite direction slowly) cancel out, leaving the total momentum unchanged.
But with beta decay, the recoil was not in the opposite direction from the velocity of the beta particle that was ejected. If the two directions aren’t opposite one another they cannot cancel completely–there’ll be some slight motion to the side left over–so there’s some “new” momentum where there had been none before.
And this looks a lot like a violation of the conservation of momentum.
So beta decay broke not just one, but THREE conservation principles!
Oh, dear.
This was hard to stomach. Sure, these conservation principles are generalizations. We see them work all the time without fail, but there’s always a smidgen of a chance that we’ll discover that they don’t really hold true. After all, we had once had conservation of mass, and conservation of energy, but then realized they weren’t true after all. But in that case, they were still true afterwards when combined into the conservation of mass/energy (or conservation of energy could be kept if mass was simply regarded as another form of energy, but that still involved “scratching” conservation of mass).
But three such violations at once was hard to believe.
At least, baryon number was safe.
And the problem persisted in 1934, when a second form of beta decay was discovered as part of the discovery of antimatter. A phosphorus-30 nucleus (one which does not exist in nature) would decay by spitting out a positron not an electron. It would end up moving to the left on the periodic table (because a proton had turned into a neutron) and become a nucleus of silicon-30, which is stable. This new mode of decay is now known as beta-plus or beta-positive decay, and it suffered from the same issues with conservation of angular momentum, energy, and momentum.
There was another violation on top of all of these but one much less troublesome. It had been suggested that the total number of electrons was fixed. Before the neutron was discovered, back when the nucleus was thought to contain protons and electrons, beta decay was just considered ejecting a pre-existing electron from a nucleus, so it wasn’t unreasonable to think that there might be some law conserving electrons. So we never saw electrons being created from nothing. But with the new understanding, beta decay consisted of a neutron turning into a proton and ejecting a brand-spanking, made-from-nothing electron. And beta positive decay transformed a neutron to a proton, creating a positron from nothing. So conservation of electrons, never very well established to begin with, seemed to have been scotched.
Wolfgang Pauli (of the Pauli exclusion principle) pondered this problem and realized there might be a way to rescue all of these conservation numbers. He wrote a very famous letter in 1930 (two years before the discovery of the neutron), in which he suggested there might be a totally new particle, one that was very light (lighter than the electron) and had no charge. As such it would be very difficult to detect.
This could solve all of these problems and save all of the conservation laws.
The spin issue could be solved by positing that the particle had the opposite spin as the electron (or positron), so that the two ejected particles together added up to zero spin, so the nucleus didn’t have to have a spin change at all in order to comply with conservation of angular momentum.
If the new particle carried away some of the energy, it would cover the “missing energy” that seemed to suggest a violation of conservation of energy.
And if beta decay resulted in three entities, not just two, then any two of them could move in directions not opposite each other, with the third particle serving to cancel the sideways momentum.
Pauli named his proposed particle the neutron. (Remember this was two years before Chadwick detected “the” neutron, and the name wasn’t taken yet.) Enrico Fermi, the next year, renamed the hypothetical particle the neutrino, This is Italian for “little neutral one,” as opposed to the big neutral one, the expected but still undiscovered neutron we know today.
And in 1933 Fermi proposed a new force, the weak nuclear force (to contrast it from the strong nuclear force). This force would cause a neutron to turn into a proton, neutrino, and electron, or alternatively a proton to turn into a neutron, neutrino and positron. In other words, it would be the force that governs beta decay–both kinds of beta decay.
In fact if we assume that there are also anti-neutrinos, we can even create a new conservation law from the ashes of the conservation of electrons. If we classify electrons and neutrinos as “leptons,” then a hypothetical “conservation of lepton number” might work better.
“Regular” beta decay, then, would turn a neutron into a proton, electron, and an anti-neutrino. Baryon number is preserved (neutron +1, proton +1, electron and anti-neutrino both 0), and the new lepton number is too; no leptons before the decay balances with the situation after the decay where there is one electron (+1 lepton) and one anti-neutrino (-1 lepton). Beta plus decay spits out a positron; to balance this anti-lepton we need a neutrino lepton.
This is very tidy. Not only are the three old conservation laws saved, a new one is created. It must have been very tempting to just assume it’s true. But it’s not enough.
We need to find the particle.
The neutron, after all had been a suggestion that would solve a lot of problems, but few were willing to take its existence on faith. Fortunately it was found, fairly quickly.
But this particle was going to be a cast-iron bitch to find. Because it didn’t have an electric charge, so it wouldn’t interact with the electromagnetic force. And it, like the electron, wouldn’t be affected by the strong force. It could only “feel” the weak nuclear force. (Just forget about gravity, it’s so weak.)
The weak force has a very, very short range. In fact, now we know its range is less than a tenth the diameter of a proton. So the only way a neutrino could interact at all is when it is directly in contact with a nucleus.
Remember that a nucleus is about 1/10,000 the width of an atom, so even with solid matter like lead, only 1/10,000 x 1/10,000 x 1/10,000 or one trillionth of the space is actually nuclei. So a neutrino, travelling through a block of solid lead at pretty basically the speed of light, is only in contact with a nucleus one trillionth of the time. So, only a trillionth of the time could it interact with a proton or neutron. So for some reaction that has a half life of (say) 1/10,000th of a second, the neutrino would have to spend 100,000,000 seconds travelling through the lead before it had a fifty percent chance of interacting one of the lead nuclei. That works out to be about three years. In those three years about half of the neutrines will have traveled three light years, or basically 18 trillion miles, and come out the other side, intact.
It’s a common trope that neutrinos will pass through light years of solid lead (if you could arrange for that to be set up) without interacting, and this is basically why. They need to spend a fairly long time (1/10,000 of a second is an eternity inside a nucleus) near very small things and they’re buzzing along at the speed of light. More realistically, the vast majority of neutrinos would simply drill right through the earth without affecting it. In fact, sixty five billion neutrinos pass through every square centimeter of the arth every second, and no one notices. More to the point, that many neutrinos also pass through every square centimeter of you every second, and you don’t notice, because they pass through, and don’t get stopped by your body.
The first detection was actually of antineutrinos generated in a nuclear reactor by the huge number of beta decays occuring in the reactor core.
When a an antineutrino does condescend to react with a proton, the proton becomes a neutron and a positron is spat out; it’s basically anti-neutrino induced beta decay, except that this decay absorbs an antineutrino instead of creating a neutrino (the books balance either way). And this was how neutrinos were eventually detected…in 1956.
Clyde Cowan, Frederick Reines, Francis B. Harrison, Herald W. Kruse and Austin D. McGuire did this by parking their apparatus–which largely consisted of large tanks of water–near a nuclear reactor, which generates a lot of antineutrinos, then waited for the tiny fraction of them that would interact with their detector. The reaction, as noted turned a proton into a neutron and spits out a positron; the positron finds an electron and mutually annihilates it, generating two gamma rays. If the nucleus hit is one of the hydrogen atoms in a water molecule, it turns from a single proton to a single neutron, which will wander off until it hits a nucleus and is absorbed–in this case cadmium was used since it absorbs neutrons readily. When this happens the neutron also generates a gamma ray. By watching for both of these events, two gamma rays from the electron-positron annihilation followed shortly after by a different-strength gamma ray from the neutron absorption, the experimenters could see that the signal matched the profile of the reaction and conclude a neutrino had hit a proton.
Even though ten trillion neutrinos passed through every square centimeter of the detector every second, only about three neutrino events per hour were detected. And just to prove that it was neutrinos from the reactor, they shut the reactor off and continued monitoring the detectors, and noticed a drop in the number of events.
But the actual detection of neutrinos (well, actually, antineutrinos) is getting ahead of our story.
Next time we’ll tackle Mystery Number 3.
Obligatory PSAs and Reminders
China is Lower than Whale Shit
To conclude: My standard Public Service Announcement. We don’t want to forget this!!!
Remember Hong Kong!!!
中国是个混蛋 !!!
Zhōngguò shì gè hùndàn !!!
China is asshoe !!!
China is in the White House
Since Wednesday, January 20 at Noon EST, the bought-and-paid for Joseph Biden has been in the White House. It’s as good as having China in the Oval Office.
Joe Biden is Asshoe
China is in the White House, because Joe Biden is in the White House, and Joe Biden is identically equal to China. China is Asshoe. Therefore, Joe Biden is Asshoe.
But of course the much more important thing to realize:
Joe Biden Didn’t Win
乔*拜登没赢 !!!
Qiáo Bài dēng méi yíng !!!
Joe Biden didn’t win !!!









Maybe someone will notice my comment now?
Since I appear to be first today.
How are you doing? Any residual symptoms?
Corona 1st…
Sciatica next….
Numb Knee (just getting over that)
Sore Heel (Probably from favoring the leg with Sciatica)
High Medical Bills from all of the above 🙂
Y’know, I’d bet that numb knee and sore heel would feel markedly better after kicking the ever-lovin’ cr*p out of some bureaucratic medical executive…..
lol…. I saw that they were LIARS when I was in THEIR offices.
Evidenced by the “Nurses” that had a clue and steered me towards Ivermectin
Wow, good for those nurses! Too many are simply following the narrative.
I think you have a GREAT point here 🙂
Good on the recoveries..bad on lingering probs.
Sometimes I feel like we’re the only ones that still a) have bills or b) care that we have bills. It seems like so many have free everything.
Hi Rayzor,
😍 👋
Sorry you’ve been having to deal with all that misery!
😧
Hope you continue on the road to recovery and get better real soon!
Thanks…. Things ARE looking up… I’m getting better by the day.
You should be nuce and healthy ti fight off flu season…😉
Yikes.
Yeah, you should have pretended to not speak any English. Everything would have been free.
lol… seems so. I have the tan for it 🙂
👋 👋 Hey Ray 👋👋
rayzorback
Good to hear from you! Thank heavens the ‘rona is gone.
Sorry about the Sciatica and the other issues.
Hang in there.
Thanks, American Patriots can … and do…. whatever it takes to get through.
Or we don’t and go to a better place 🙂
…. 😃🤚‼️❤️‼️ … 😉
Well, … would you like a nice fed Notice to Appear? In a faraway city?
Count your blessings, boy. 😂
More uncalled for Bullshit!
Expected. Boy?
There Ain’t No Justice!!!!
–little fish
Any word about bakocarl???
just posted update
TY!!!
so very welcome…
With all the Sturm and Drang in the markets, I am seeing a whole lot of “Meh.” from metals.
Carl has an update after his procedure today:
—————————————-
bakocarl
October 2, 2021 at 12:03 am
Update 4 –
We got our Kleen Kovid Klearances in the nick of time. Evelyn finally got hers at 0610 this morning and we left at 0615 for the hospital in Las Vegas.
We found out that the deliberate blockage of the left internal iliac artery (IIA) wasn’t to facilitate the operation, but would be a permanent blockage because blood flow through the left IIA endangered the integrity of the stent sheath repair to the aneurysm. However, the surgeon assured me that blood flow from a healthy right internal iliac artery would provide ample blood flow to the effected pelvic and buttocks areas. So, with a big gulp, we proceeded.
The two-hour procedure only required a local anesthetic at the site of the femoral artery and a light sedative for me . . . they wanted me awake for some reason.
All went well with the procedure and the light sedative was absolutely wonderful, making me feel all warm, comfy and pain free throughout the procedure.
The worst thing was the utter boredom for 12 hours, minus the 2 hours of warm and comfy during the procedure. I was thrust a bit further down the road to completely nutso.
In about two weeks, after my kidneys have removed the iodine contrast from my system, I’ll get the endovascular abdominal aneurysm stent graft repair . . . under a general anesthesia (yippee), but will have to remain in the hospital at least overnight (Boooo! Hissss!) . . . but that hospital has full Wi-fi (Yaaay!).
So, onward and upward . . . time will tell.
Let God’s will be done!
YAY!!!!
Praise God! 🙏 So glad to hear your doing well Carl. ❤❤❤
Thank God! That’s great news.
WOW! Hallelujah!
bakocarl
So good to hear this update!
God keep you perfectly through the next procedure and may rapid healing commence!
Amen.
Great news Brother!
Yay‼️Yay‼️Yay‼️ ….. ❤️‼️ ..
Ha!
Someone tell me the power of prayer and the Lord Almighty isn’t real.
Thank you, Holy Father. Thine is the glory!
Good news! So happy to hear it. Blessings, Carl.
Side note for Steve:
arth
So if you branded cattle with ℏh, that would be the “h bar h ranch”?
Send in the neutrines!
Those are both great!
She certainly is! 👍😁
Many a man in love with a dimple makes the mistake of marrying the
whole girl.
— Stephen Leacock
Dimple? Oh now I see it! 😉
Fixated on the projecting, FJB.
Lemme go back for the dimples and…
A question for anyone who had the virus with the no taste/smell symptom.
What was that like ? Simply no smell or taste or did anyone have a strange taste in mouth while drinking or eating ?
I have had some headache, fatigue…generally feeling kind of odd. I upped the supplement regime and felt normal but today I had the most awful taste while eating or drinking. Even water tasted punk. Thought it was old ice, got new. Only used water from the purified cooler thing…………still icky. Orange juice..ick. Iced tea..urp.
Does that sound familiar to anybody ?
I will Testify about my experience….
YES!
Things smelled AND tasted Putrid for a couple of weeks.
It was towards the end of my experience with the “Rona”.
Hope this helps.
I credit Ivermectin, Zinc and Vitamins C and D for my recovery.
I now have antibodies and am STILL a “Pureblood” 🙂
Ready to star in a “Horry Patter” book: “Rayzorback and the Pureblood Curmudgeon.”
Wait…I thought he WAS the Pureblood Curmudgeon.
“Get off my lawn, you commie millennials!!!”
😀
Thanks a lot for telling me about your experience.
I never got any ivermectin so…will have to hope the OTC stuff helps. Might not even be the Rona but it’s weird.
Molly, there are still a few places you can get livestock Ivermectin fairly quickly. Upco.com is one. They have the injectible, that you just add to liquid. Amazon still has the paste. Either will work just as well as the tablets and you can have them in a few days.
Gulp !! I’ll order the paste , maybe it ‘ll come quickly.
Should’ve done it a few weeks ago when I was thinking about it
Yes, please do. I had COVID before the big hoopla about it and before anyone knew how to treat it. So I basically didn’t know about quercetin or green tea with zinc or anything else, though I always take plenty of C and D3. Those things plus ivermectin would have made the whole ordeal a lot easier. I was sick a good 3 weeks and lost my sense of taste/smell for well over a month.
Oh…that’s a rough one. Glad you finally got over it.
I thought that I had it already too…but don’t know.
The antigen tests ($25 for a 2-pack at CVS) are really accurate, and if you’re tasting and smelling funkiness, then the test will show positive if you’ve got COVID-19.
Some other coronas affect taste and smell, too, but the test won’t flag them.
Antigens will fade however. I believe after 8 weeks pretty much gone. They say the T cell test will work if you had covid a long time ago. It is expensive but I am trying it out. Will update when I get results.
You’re thinking ANTIBODY tests, and YES, the antibodies will fade, but that is a different test.
ANTIGEN means COVID proteins – and specifically the ones that can be picked up in the cells, mucosa, saliva, etc.
BinaxNOW test uses the nucleocapsid protein. The test looks for levels of the protein that are HIGHER than a certain amount – and thus consistent with active infection – NOT lower levels like in people who are simply inhaling COVID dead virus and dust, or people who are recovering.
Do you need the swab up to the brain or does spit work?
It’s actually in between.
With BinaxNow and tests like it, you stick a shorter swab roughly 1/2 to 3/4 inch up the nose, so it’s not even as far as the bridge.
QUOTE:
https://www.fda.gov/media/141570/download
The antibiotic Ciprofloxacin has loss of smell as a rare side-effect (along with some VERY gnarly other side-effects). I’ll take losing smell over popped ligaments (one of those side-effects) any day, though…
I had it also before everyone knew anything. I took lots of Vitamin C and in my eye meds I have Zinc and drank green tea. The cough hung on a long time. Something I did right that I am still kicking 🙂
“Gulp !! I’ll order the paste , maybe it ‘ll come quickly.”
_____________
Hopefully next day delivery option is available. I’m sure that will cost extra, but worth it 👍
Tractor Supply still has it around here at times. It works fine. Been using it as a prophylactic, but I’ve noticed a little improvement in respiratory function even from fall allergy problems.
Thanks for the info Linda. 👍
My other opportunities dried up in shipping 🤔
Check your local feed stores and co-op’s
“I now have antibodies and am STILL a “Pureblood””
_____________
100% pure old fashioned homegrown human, born free, right here in the real world 👍😁
… As in the days of Noah….
Some believe that at that time, mankind was so genetically corrupted that Noah and his family were the only “Purebloods” left on earth.
Looks like Coudenhove-Kalergi (what Merde-Kuh’s trying to implement) was “alive” and kicking even back then… reversing GOD’S judgement on the Tower of Babel (or, rather, attempting to…)…..
You got HCQ or ivermectin? If so, it’s time.
Don’t have either of them. Hoping for the best * if* I do have it.
Load up on C, D, Zinc. Take Quercitin, Selenium, Melatonin, Green Tea, Antihistamine, Turmeric
My 17yr old nephew said he lost taste/smell, took nothing.
have all of those except the antihistamine, will get that tomorrow.
I also read another good thing to take is kefir. Duh, can’t remember what it supposedly does but going to get that tomorrow too
The antihistamine should be generic zyrtec or claritin, and the protocol looked like your first dose should be 2x the dose on the box (but verify!).
Why generic? Any reason?
OK Thanks 🙂
Kefir or yogurt and Israeli study shows beneficial.
I also took one aspirin.
I do the regiment you described. I ordered from Dr Zelenko.
Antihistamine works with the vitamins for me. I never took Antihistamines before. Something is floating around had for 3 days sinus problems headache strange taste. Took vitamins doubled D3 bone broth, green tea and slept a lot. Feel way better today.
It is good to be in tune with the body.
IIRC, you’re essentially in the Jefferson area of CA. You may be able to get a script from an online doctor and get IVM from a local pharmacy by tomorrow afternoon. The window for HCQ is probably closing.
What’s your regimen looking like? And which decade are you in?
If I had that symptom, I’d be at 250mcg D, 2000mg C, regular Zn, regular (for me) Se, 2x500mg Quercetin….and IVM.
Anyone nearby? I can see it now — meet at a city park, on a park bench, wrapped up in bogus PPE and disguises. “Life is short.” “But the years are long.” “Not while the evil days come not.” “*polishing fingerprints* Here’s the tube — I was never here.” “Found it on this park bench.” “Good luck.”
Hahah you crack me up sometimes.
No, I’m down in So Cal . I could check the local feed place (never actually been in there but know where it is)…the next burg over is rural/horsey
Hopefully, you have someone to run errands for you. If you get the right stuff, you can bounce out of this in a few days. If it gets bad enough that you have to go into a hospital, the bureaucrats get to decide whether you live or die and how unpleasant it will be either way.
I just remembered that I had a home test unit here. In 15 minutes it’s supposed to detect it or not.
At 11:30 pacific time…I’ll know !
I’m not sure if I’d believe any test any more than a coin flip. Elon Musk famously took four tests in one day administered by nurses and got 2+ / 2-.
Those were PCR. The antigen tests are solid!
They were solid. A politicized FDA and CDC corrupt anything they touch. The first ten on the gallows will be a down payment on my trusting ANY American medicine again.
This is all-or-nothing thinking – you’re hanging out with Scott too much. 😉
CDC fucked up PCR even more than it was already fucked up as a technology. Antigen is solid and it’s the WAY OUT OF THE MESS.
It was ALWAYS the way out of the mess.
They SHOULD have used antigen tests from the beginning. THAT is exactly how CDC and China fooled us all. And THAT is why they killed Kary Mullis, I’m positive.
He could have instantly explained why we needed antigen tests right away, and why PCR was bullshit. But PCR wins elections – antigen does NOT.
Don’t equate the two – if you do, you only help the other side.
BinaxNOW and tests like it are generally hard to game. They are NOT overly sensitive, because they have a good design, IMO.
And that’s why they realized in Canada that antigen tests were giving THE PEOPLE too much information, so they’re restricting them.
EVIL.
Don’t help the other side. ANTIGEN TESTS will help us see clearly.
Great Scott!!!!! 🙂 I always trusted my Auntie Jen… (anything other than those tickle-the-brain tests…)….

LOL!!!
I may have to borrow this – for a completely different reason!!!
yahoo…according to this home jobber I don’t have it.
Still doesn’t explain my strange symptoms so I’ll go ahead and still take all my OTC things.
Thank you very much everybody for riding along on the Molly hypochondria train !
It’s not hypochondria if you really have it…..and I wouldn’t trust a test more than a coin flip.
Right but you know the old mind over matter adage….but I will proceed as if I do have it…but for sure not googling what other conditions or diseases can produce the symptoms…lol…that’s where the hypochondriac paranoia can really set in !
Other coronas can affect taste/smell.
They have their own spike proteins.
But, so far as I know, all are vulnerable to the same protocols.
Very likely so, although if a mutation affects an actual active site (e.g., where the Pfizer inhibitor binds), then there may be a difference in susceptibility.
The mutations are inevitable with the “vaccines” and even herd immunity. mutations from the original would likely be good as the mutates in each generation are likely weaker, less transmissible than the original.
Unless of course new vaccines produce new versions resistant to immunities. Versions created and ready to be introduced by the very “vaccines” that are meant to STOP the virus. They would not do that would they? OF COURSE they would there is SOME reason to PUSH those vaccines SO hard. IE MONEY. creating a never ending supply of viruses AND dependability on “vaccines”
Yup. Pfizer is an ENEMY OF THE PEOPLE.
Makes me wonder just what Mercks play is here. If as I suspect it is to cash in on the success of Ivermectin, it will gut the Pfizer, J&J and Moderna “vaccine” monopolies, while “repurposing” an OLD product into new more expensive packaging. Making Merck a MINT while crushing the rest.
The questions are will the others allow this?
Will the others produce like generics?
Will the others use lobbys and Fauci to CRUSH Merck?
IF Merck is allowed to bring this to market, it is my guess the JIG is up on the “vaccines”, and they cannot hide the truth from the world much longer. BUT, they ALL want to cash in on Covid some more.
An old friend once said “There is NO money in the cure, the money is in the DISEASE,”
I never believed him. I always thought there would be a clamor to find the cure, that there were STILL going to be REAL Dr.s, researchers, etc that would “do the right thing” He was right, Covid proved it. What ELSE have we the cure for that has been held back?
I guess Saulk was the LAST real researcher who cared more about saving lives than MONEY. I am ashamed to work in the place I do anymore.
Yes on chemical therapeutics, NO on many antibodies, and most definitely NO on vaccines – and yet THAT is what we pursued with such determination, and yet so wrongly.
Curious what happened to all those flu case from 2019 isn’t it. 39 million to 2176 in 2020. LOL coincidentally there are a reported 39 million or so Covid 19 cases in 2020-21.
The PCR test is a SHAM. Covid 19 is and was nothing more than the regular flu weaponized to make it more easily transmissible.
The hype machine and BAD actors with BAD protocols and suppressed therapeutics and BAD polices like ignoring herd immunity and doing EVERYTHING opposite of modern medical practices did the rest…OH and giving the hyped scam its own high dollar money billing code was the grease that moved this skid throughout the healthcare system.
Covid is a cash COW for people that need the money.
Question. IF Covid is SO bad and overwhelming the hospitals SO much, WHY introduce a huge amount of illegals and refugees into the system that come from places with little to NO healthcare and rampant old world diseases with NO mandates for masks or vaccines for them, while at the same time FIRING thousands of healthcare workers for NOT taking the death jab leaving a huge void? Perhaps trying to maker a self fulfilling prophecy of “overwhelmed” hospitals.
What happens when even THAT dose not produce the desired result?
If symptoms continue, give yourself another antigen test in a day or two.
Antigen tests are solid, but sometimes the PEAK viral load lags by a day or two.
The bad beauty of the spike is that if your viral load INCREASES now, you will have more symptoms, and you will also have correpondingly more nucleocapsid, and you will test positive.
If you DON’T have symptoms, then you don’t need to waste another test.
And I will say right here that anybody who says antigen is a coin flip is WRONG. I was an early advocate for antigen tests and backed the doctors who were promoting them. The MATH behind them is SOLID, unlike PCR.
Something to consider.
https://rumble.com/embed/vjgie1/?pub=4
Stew Peters and Judy Michovits (sic) She talked about shedding and how it was just the spike proteins that were passed and how it wasn’t picked up on tests? Short segment. She gave solutions, but talked very fast.
If your going for the 1.87% paste you don’t want the Gold or Plus. Just the basic DuraMectin or other durvet. with Antheiminitc and Boticide, nothing else.
I never lost the sense of smell completely, but for a long time, many things had a kind of burnt smell instead of the original. Even things like eggs, vinegar, windshield-fluid, alcohol, and lemon. More this generic burnt smell than the characteristic smell that they actually had. It has slowly corrected itself. I still have occasional «bad days» where the distortion has returned, briefly, but these are further and further apart.
Very strange…very.
Evil alliance setting this virus off into the public to usher in the new communism brand.
I think the smell/taste thing kind of changes over time. For the longest time it was hard to taste/smell almost anything. Then some tastebuds/scent-receptors seemed to partially come back “online”. Nothing is completely restored now nearly a year since we were sick. My husband still hates the scent of his favorite cologne. Unfortunately our ability to smell bathroom scents has mostly come back 🙁 The lack of that capacity made for slightly more harmonious domestic tranquility 😉
LOL!!!
Yeah there was the day at my husband’s work where there were sewer issues & all his co-workers were bumming but he & our son were entirely oblivious in their post-covid states. Like my poor color-blind co-worker that we used to tease about having mismatched blue & black socks, which were actually matched, but it was just too much fun to yank his chain & he was such a good sport. We worked in a federal program & he shared his terms for various types of welfare, these I still remember:
Gravy for Granny
Bucks for *****
A colorblind friend of mine got married once, and part of his outfit was a deep green, so I said to him, “why are you wearing red” and he laughed and said his fiancee had picked the colors so he felt safe.
Nice!
Everything was awful with the second one.
My first time through, just smell disappeared.
Yes that is what it feels like.
I truly believe she’s right
IMO, we passed the point of reconciliation with the Left many years ago. You can’t reconcile with the enemy; you must defeat it.
I understand what Candace is saying, but we can’t cede certain states to them. This is the United States of America, with a Constitution and founding documents that are antithetical to what the Left believes and wants. They don’t get to take parts of the USA. They need to be defeated and/or leave if they want to be socialists/communists/globalists.
We have some mechanisms in place to stop this, like the oaths of office that people take to protect and defend the Constitution. When they violate that, no one does anything. We could start right there and clean up a lot of the mess.
I think we should look into ceding certain counties, rather than states.
We could just get rid of all the major metro areas and let them figure out how to get food. They think it grows in grocery stores.
 So true !
So true !
[*Ahem* — pretty much the same thing.]
Agree, but my only quibble with this map is how many of the blue areas are actually stolen blue areas based on the machines?
Askin’ for a friend … 😉
Is this the fake one from last year?
It’s not.
That one was simply the fifty six largest counties in the US (or some such number), showing that half of the US lived in those counties.
My own county is one of the fifty six but voted for Trump, and that’s how I smelled it out.
I even found the blog that showed the map with both the correct label and the fake one, but wasn’t allowed to kneecap the asshole.
Food “grows in grocery stores” or some may ever expand that to warehouses & “fulfillment centers.” 🤪
For those who remember the SF Bay Area in the 60s,
“Farms? In Berkeley? MOOOOOOOOOO!!! ”
(Old Berkeley Farms radio ad, on KSFO and other bay area radio stations, KNBR, KCBS, KNEW, KKHI, KDFC, KIBE, KYA, KEEN, and others)… (not sure if KABL or KFAT had the ad 🙂 )…..
Prisons
First, we have to get the power back AND punish the crimes. ENFORCE THE LAW, what a concept!
Had the power for four years.
Didn’t punish anyone.
Not even a little.
Except patriots.
Punished us all the day long.
We didn’t have the power for 4 years, the SWAMP did.
Thank you!
I agree. Even PDT knew his limitations of power within the DS. Which is the point. He and We the People only appeared to have the power for a short time. We now see up close and personal that he and We did not. Which is PDT’s point and why they hate him and us so much. We the People are awake. The core MAGA’s know the truth now. Our children and children’s children are being taught to never believe what the government says and does as well as the schools teach them without evidence. So what do we do with the knowledge?
You cannot effect lasting change if what you are led to believe to be true is just a huge deception like 45’s term ended up being. The NWO can basically overturn everything he accomplished. For now, I will celebrate the battle victory of my beloved East TN landing the headquarters of Smith & Wesson as they vacate the crap political state of Massachusetts, which no longer reps the Constitution and America that its founders fought and died to build. It will be 20 minutes away from me, so pass the ammo.
Saw that.
https://www.thegatewaypundit.com/2021/10/leading-gun-manufacturer-smith-wesson-moving-headquarters-massachusetts-tennessee/
Congratulations!
Springfield MA. no longer deserves them even if they are my hometown.
>>”the crap political state of Massachusetts, which no longer reps the Constitution and America that its founders fought and died to build.”<<
Like too many other States, “Commie-chusetts” has lost the plot, and its power resides way too much in the cities – Boston especially.
For decades, any “Republican” politico has been a de facto Dem – “RINO” isn’t a strong enough term.
Bill Weld? Mitt Romney??? Puh-lease!
It’s a disgrace.
The only ray of hope I’ve seen in decades is Dr. Shiva, who has had a series of wins with his lawsuit. The AG, Maura Healy, allowed for the DELETION the election records without consequence, and I saw a video of her walking away from Dr. Shiva without answering any of his questions; and the SoS, Bill Galvin, has been MIA w.r.t. this, and other, voting “irregularities” (a euphemism if ever there was one!).
Not to mention the Guvna, on Pompeo’s “friendly” list with the Shy-Knee. All the “bio-tech” contributed, no doubt.
The kicker, of course, is that SO many residents here have the same quintessential ARROGANCE as the self-entitled “elites”.
The ignorance is stunning. At the first practicable opportunity, I shall leave. Too many self-serving, self-important. self-aggrandizing Mass-Hoes here.
And again – this attitude has prevailed for decades. Worst drivers ever, too!
None so blind as those who WILL NOT SEE.
*I* feel like an alien here.
I wonder whether Nor’easter and Yucki agree …
If we gave them certain states, WE might be satisfied because they’d no longer be among us bothering us, but THEY would never be happy because they MUST bother us because they have the crusader mentality.
Yes. They want to force everyone to do their bidding and to live in the h3llholes of their making.
they need conservatives
Yes, but then we could have an armed perimeter.
“I wonder if we’re moving past the point of reconciliation with the Left.”
______________
😂🤣😂🤣😂
SEVERE battered conservative syndrome there…
We’ve been past the point of reconciliation with the Left since the Clinton administration.
Besides all that, the Left wouldn’t accept ‘reconciliation’ even if your terms were unconditional surrender to them.
Samuel Adams is too good for them, but sometimes it’s a helpful substitute for what I really think about them.
……………………………
“Contemplate the mangled bodies of your countrymen, and then ask yourself, ‘What should be the reward of such sacrifices? Are we just to do nothing? To allow the men who have let loose on us the dogs of war to riot in our blood and hunt us from the face of the Earth?’
I detest any submission to a people who have either ceased to be human, or have not virtue enough to feel their own wretchedness. If ye love comfort better than liberty, the tranquility of servitude better than the animating contest of freedom, [then] go from us in peace.
We ask not your counsels or arms. Crouch down and lick the hands of your master. May your chains sit lightly upon you, and may posterity forget that ye were our countrymen!” — Samuel Adams, August 1, 1776…Faneuil Hall — Boston, Massachusetts
…………………………..
>>”reconciliation with the Left”<<
The order is backwards.
It is THEY who must seek reconciliation with US.
Full stop.
Send them to some island, maybe with the name GITMO, in it.
Send them to some island, maybe with the name “Atlantis” on it — so long as the ships are full when sent out and empty when returning…..
Letting the left have their own states to destroy would not work given that destroying entire countries has only forced to move on to other countries…while blaming others for their failures.
Yeah Hitler was satisfied with Czechoslovakia & Poland AND…NOT!
All we have to do is stop feeding them for a while. They will come begging on their knees.
Remember the Great Success of the Chaz/Chop? That pitiful garden?
If we all just quit at once, it’s over in a month.
👍👍👍👏
I haven’t read any of yesterday’s thread and don’t know if this has been discussed. I find Kristi Noem’s denial of an affair with Cory Lewandowski to be a non-denial. She does not directly state “I did not have an affair with CL” or “I am not having an affair with CL.” Instead, she tries to dismiss the charges with this:
https://www.foxnews.com/politics/gov-kristi-noem-denies-allegations-of-affair-with-corey-lewandowski-total-garbage
She refers to “rumors,” not her own conduct. Then she invokes generic attacks about conservative women needing a man’s help, which doesn’t ring true with me. I don’t remember seeing those kinds of attacks on conservative women. The criticisms are usually about the woman’s ideology (e.g., being against abortion, favoring home schooling, being pro 2A, etc.). She says she loves her husband, as if that would preclude an affair (it wouldn’t). Then she invokes religion, which could be seen as virtue signalling. Finally, she tries to dismiss it all by “getting back to work.”
I don’t know if the allegations are true or not. I just don’t think her denial is an effective one.
The thing is, it isn’t a political problem if she feels compelled to screw two random strangers in the street before every day of governmental duties — such things used to have a strong set of societal guardrails and were handled on a personal, local level — despite the political impulse to make personal behavior a political issue since almost the inception of the Republic.
I find it disgusting that there are no such societal safeguards and that politics has descended to a 24×7 full time s**tshow unconnected with actual governing.
Back in the day, Andrew Jackson had an admirable method for dealing with those who impugned the honor of his wife. He shot them. The venue was known and witnesses were welcome — and the other party could shoot back at that time or profusely apologize in advance, withdrawing any charges, and avoid the moment. Frankly, it was a more civilized time.
And, somehow, this method of dealing with personal scandal didn’t stop him from being a US Representative and US Senator from Tennessee, Governor of Florida, and 7th President of the United States for eight years.
“The thing is, it isn’t a political problem if she feels compelled to screw two random strangers in the street before every day of governmental duties —”
_____________
It’s a character problem.
The devilrats tried to claim that it wasn’t, or that it wasn’t relevant to leadership, and taught the world what ‘compartmentalism’ was after Bubba stained an intern’s blue dress, and then lied under oath about it.
And he was rewarded for it.
That was the end of any notion (for me) that America wasn’t sick to the core.
If you don’t have strong mechanisms for character problems, you end up with character problems bleeding into political issues.
Jackson left a string of corpses of those who disputed his character. This was a distinct advantage when keeping policy disputes in context.
And if anyone believes that this resulted in “watered-down” policy disputes, I can point to one of the greatest political speeches in US History — https://millercenter.org/the-presidency/presidential-speeches/july-10-1832-bank-veto
If you have studied every detail of the squabble and its history, the entire speech is a rich feast. But even if you know nothing of the context, the final paragraphs just pound the essence of America into stone:
“It is to be regretted that the rich and powerful too often bend the acts of government to their selfish purposes. Distinctions in society will always exist under every just government. Equality of talents, of education, or of wealth can not be produced by human institutions. In the full enjoyment of the gifts of Heaven and the fruits of superior industry, economy, and virtue, every man is equally entitled to protection by law; but when the laws undertake to add to these natural and just advantages artificial distinctions, to grant titles, gratuities, and exclusive privileges, to make the rich richer and the potent more powerful, the humble members of society–the farmers, mechanics, and laborers–who have neither the time nor the means of securing like favors to themselves, have a right to complain of the injustice of their Government. There are no necessary evils in government. Its evils exist only in its abuses. If it would confine itself to equal protection, and, as Heaven does its rains, shower its favors alike on the high and the low, the rich and the poor, it would be an unqualified blessing. In the act before me there seems to be a wide and unnecessary departure from these just principles.
Nor is our Government to be maintained or our Union preserved by invasions of the rights and powers of the several States. In thus attempting to make our General Government strong we make it weak. Its true strength consists in leaving individuals and States as much as possible to themselves–in making itself felt, not in its power, but in its beneficence; not in its control, but in its protection; not in binding the States more closely to the center, but leaving each to move unobstructed in its proper orbit.
Experience should teach us wisdom. Most of the difficulties our Government now encounters and most of the dangers which impend over our Union have sprung from an abandonment of the legitimate objects of Government by our national legislation, and the adoption of such principles as are embodied in this act. Many of our rich men have not been content with equal protection and equal benefits, but have besought us to make them richer by act of Congress. By attempting to gratify their desires we have in the results of our legislation arrayed section against section, interest against interest, and man against man, in a fearful commotion which threatens to shake the foundations of our Union. It is time to pause in our career to review our principles, and if possible revive that devoted patriotism and spirit of compromise which distinguished the sages of the Revolution and the fathers of our Union. If we can not at once, in justice to interests vested under improvident legislation, make our Government what it ought to be, we can at least take a stand against all new grants of monopolies and exclusive privileges, against any prostitution of our Government to the advancement of the few at the expense of the many, and in favor of compromise and gradual reform in our code of laws and system of political economy.
I have now done my duty to my country. If sustained by my fellow-citizens, I shall be grateful and happy; if not, I shall find in the motives which impel me ample grounds for contentment and peace. In the difficulties which surround us and the dangers which threaten our institutions there is cause for neither dismay nor alarm. For relief and deliverance let us firmly rely on that kind Providence which I am sure watches with peculiar care over the destinies of our Republic, and on the intelligence and wisdom of our countrymen. Through His abundant goodness and their patriotic devotion our liberty and Union will be preserved.”
Thank you for sharing this excellence!!!
So true is is a character problem. From my own exsperience I always distand myself from a man if they did double talk coming on to me. Normally that was enough and if not I spoke clear so they understood 100% I was not available.
I have no understanding with a married man/ woman having an affair unless there is already a problem in marriage. Maybe people should first end one commitment they made before God before entering in another.
She’s a snake woman.
Snakes are sneaky, just like RINOs.
She’s being tossed aside by the machine, IMO. Sex scandals, whether they happen or not, is/are a safe way to do that without revealing too much.
As far as I’m concerned, this is a non-story being dredged up to get rid of both Noem and Lewandowski. They both did something to piss someone off, and this is the cover to get them off the stage.
>>”Sex scandals”<< >>”the cover”<<
It’s the preferred “go-to” to get rid of traitors from either side.
I’d wager that the REAL reason is something much more nefarious.
It’s nigh on impossible to achieve such a lofty position without “costs”.
Some have done it; the vast majority of others haven’t succeeded on their own merits.
Noem is flaming out, quickly.
I actually think it’s Lewandowski, and Noem is collateral damage. She is a RINO, and expendable. But Lewandowski has been dropped by Trump. Something happening there.
If it is not true it will not work. Truth wins.
Per our host, Mr. Wolf, I’m bringing this post from last night to today’s thread:
Whoa! This article has some really good data showing that the vaccines are definitely causing ADE.
BREAKING: AI-powered DoD data analysis program named “Project Salus” SHATTERS official vaccine narrative, shows A.D.E. accelerating in the fully vaccinated with each passing week
https://www.naturalnews.com/2021-10-01-ai-powered-dod-data-analysis-program-project-salus-shows-ade-accelerating-fully-vaccinated.html
This is an interesting finding:
“Finally, a horrifying slide in the data set reveals that one of the highest risk factors for being hospitalized after being vaccinated is simply being of Native American descent. According to the data in this slide, Native Americans face around 50% higher odds of being hospitalized after being vaccinated, compared to other ethnic groups such as Whites.
“Hispanics face a slightly lower risk which appears to be around 40% higher odds. Blacks face around 25% higher odds.
“Why is this the case? The gain-of-function properties which were engineered into the SARS-CoV-2 biological weapon — via Fauci, Daszak and the NIH — target ACE2 receptors which exist in higher densities in targeted organ systems of many minority groups such as Native Americans, Hispanics and Blacks. This has led many observers to conclude that the covid spike protein — which is generated in the bodies of those who take mRNA vaccines — is a race-specific bioweapon designed to achieve depopulation of minority groups.”
Thanks!
I think Mike Adams did a great job – he made some very pertinent observations about the document that were missed by others.
The one thing (so far) I don’t agree with, is the idea that the results show DEFINITE ADE. It’s currently my opinion that the results MAY show ADE, and if they do, then it MAY be significant, but we can’t really be sure yet. There is some possibility that the vaccines are simply losing effectiveness, as far as this data goes, and the “worsening” is really a return to a no-protection state. Also, even if there is ADE, it may not be as bad as the “it killed all the ferrets, and two kids” kind.
However, that does NOT rule out TERRIBLE ADE in the future, if we keep playing vaccine whack-a-mole! If there is a vaccine-variant pair with the right differences, there could be a really bad reaction with inappropriate antibodies.
Mass vaccination MUST be stopped, IMO.
But wouldn’t the fact that viruses generally mutate into “more contagious-less deadly” come into play? If it isn’t ADE. then this virus has mutated as more deadly. We have more deaths in highly vaccinated areas than a year ago when we had no clot-shot.
Sounds like ADE to me.
First of all, there is a LOT of play in the numbers, because of CDC games. Sadly, we can’t trust the numbers enough for strong, clear, early warnings, IMO. That is why Malone had an “AHA” / GOTCHA moment when he heard the whistleblower leak that serum virus titers were higher in the vaccinated. That is a number that can’t really be messed with easily. But we are NOT seeing really strong and obvious deaths of the vaccinated due to the disease – YET. And because we have a really good range of vaccine loss of effect, it’s pretty clear to me that we have NOT encountered what I would call “killer ADE”. But that could come, easily, with a future variant.
Next, we have NOT had a normal virus evolution – BECAUSE OF THE VACCINE. Malone explains this REALLY WELL here:
https://youtu.be/qP31cfD3YOY
It’s long and technical, but he tries to explain how the leaky and very specific “spike” vaccines – named rather ironically – create a sharp “spike” of evolutionary pressure, which the virus then evades by highly directed evolution. Thus, we’re TRAINING the virus to improve by FIGHTING, rather than be becoming copacetic with humanity. This will likely lead to a Marek’s situation.
It is WRONG to force humanity into a Marek’s disease dilemma.
I agree. We are in a world of hurt if things don’t change SOON.
What “they” are doing is creating false virtue signals to the deluded COVIDIANS so that they think they are protecting “the chilluns” and “grandma” and all these other things, when logic says they’re actually condemning all of us to a controlling slavery.
You know, I used to feel sorry for the deluded, but not anymore.
Nothing I or you know about any of this is obscure or unknowable. We don’t have special powers. We just look at things, read, and pay attention.
Anyone HAS to have seen enough by now about the controversy about covid and vaccines to have their curiosity piqued at least, if they have ANY interest in being anything other than a government sheep. If they haven’t looked by now, they are the walking dead. They will just go along to the gas chamber without a peep. It is just stunning to me that there are so many of them.
Some people are so addicted to the “high” of the virtue signal that they are willingly trading their lives for it. It’s no different than drug or alcohol addiction. In AA they say “some die so others may live.”
Wow. That makes sense, actually.
Lol, don’t sound TOO surprised! I make sense occasionally. But don’t get used to it!
LOL 😉
Be great if a Sunday talk show highlighted this. Yea. Not holding my breath. Maybe Tucker will highlight this next week.
The only Sunday host who might go near this is Maria B.
Yea. She has the integrity. Short of videos posted QTree, have not watched Maria or any talking head in a long time.
Seems to me, this is NOT a likely topic for Maria. Hope I am wrong.
I know that I’ve been sporadic of late, and I have taxes coming up besides, but late at night I sometimes research stuff I want to use with Robert Baker when he gets his Linux going.
And I just happened to trigger something out of older, stranger Linux….
“Q: How do you save a drowning lawyer?
A: Throw him a rock.”
It’s all fine to say that Linux can run browsers or word-processors or email or spreadsheets like everyone else…..but that’s not always where the fun is.
The Fruit Loop is operational. It is connected to the net but I have not yet logged on because you suggested you had some info to pass on before I did so. Awaiting further instruction.
OK, if you changed the password during startup (I forget when they started asking for this), then it’s simple — open a terminal (it’s something like the fourth icon across the top — looks like like a black rectangle with a >_ in it).
At the prompt, type “sudo apt update”, then return. If prompted for a password, enter the pi’s password, then return.
Then type “sudo apt install ufw”, then return.
Then type “sudo ufw enable”, then return.
If it comes back with something like “Firewall is active and enabled on system startup”, you should be fine.
If you did NOT change the password during startup, it’s a little more convoluted. Let me know.
From here, it’s like a “choose your own adventure” — if you’d like to know what we just did, let me know. If you want to know about browsing, that’s another good choice. Talking about items on the menu is another good option. If anything else strikes your fancy, just ask.
Let’s get that firewall turned on ASAP, though.
It did not ask for a password. When I entered sudo apt update it returned:
Get: 1 http://archive.raspberrypi.org/debian buster InRelease [32.0kb]
Get: 2 http://raspbian.raspberrypi.org/raspbian buster InRelease [15.0kb]
E: Respository ‘http://raspbian.raspberrypi.org/raspbian buster InRelease’ changed its ‘Suite’ value from ‘stable’ to ‘oldstabe’
N: This must be accepted explicitly before updates for this repository can be applied. See apt-secure(8) manpage for details.
Do you want to accept these changes and continue updating from this repository? [y/n]
While I was typing the above my monitor screen went black and I hit the RTN key to refresh and the following text appeared
0%[working] 78B/s 1h 23min 53s
Get: 3 http://archive.raspberrypi.org/debian buster/main armhf Packages [393 kB]
Fetched 440 kB in 10min 10s (721 B/s)
Reading package list…Done
E: Failed to fetch http://raspbian.raspberrypi.org/raspbian/dists/buster/InRelease
E: Some index files failed to download. They have been ignored, or old ones used instead.
I think they’re having trouble keeping track of what versions they’re running. Let’s press on.
You are doing well, btw — you’re clearly in a terminal window and you have apt’s attention.
Add to your available options: “Tell me about apt”.
OK, still in the valley twixt sally and response….
It has become a “thing” for modern Linux variants to make everything into “packages”. Any of us old-timers who dealt with UNIX, BSD, Xenix, SCO UNIX, or any of the other old-style systems will remember how much of a pain-in-the-ass it was to painstakingly assemble a functional operating system back before this. Naturally, this lead to the creation of software to manage the packages.
But when I say, “make everything into packages”, I should maybe say, “make EVERYTHING into packages.” The list of lists of acceptable packages is, itself, a package. And this is what got RB crossways when I had him say “sudo apt update” — that command tells apt to access the online package lists based on its stored package of acceptable online package lists.
Clearly, it needs to upgrade its package of acceptable online package lists and try again….but we were pursuing a different path.
This is annoying, and it is a QA issue, but it is ultimately of little import if we had a faster and more responsive “chat link” and “download link” to get software right.
I posted a longer response.
If you did not change the password during startup, and plugged it into the internet, you have already taken on a chunk of risk you didn’t have to. It’s still about 0.001% of the risk someone has when walking around with an Android phone, but it’s risk you never needed to have. Let’s change the password.
Simply open a terminal and type “passwd” (since it is your own password you are changing, you needn’t use sudo). It prompts you through the process.
Again, this is VERY COMMON — you get the same thing if you buy a new router and plug it into the internet before configuring it and changing accounts/passwords. Or plugging in a new internet “security camera”, or anything else that ships with a built-in user and a built-in password.
If you are exceptionally paranoid, and would like to take further steps, let me know. Otherwise just continue and enable the firewall.
FYI, the further steps would begin with creating a new user with all the powers and priveleges of “pi”, then logging in as that new user and nuking “pi”. No biggie, but a bit of a detour.
When I try to change the password it ask for my current password. When I begin to type it does not register on the monitor. The cursor does not register any keyboard input. As to the internet all I have done is to connect the computer to the router. I have not opened the browser.
When you type in the password, it does not show. You have to type precisely, but blind. It’ll let you know if you get it wrong.
Several weeks from now, we might decide to talk about routers and NAT.
Many corporations with dedicated IT departments don’t pay enough attention to the basics.
Not opening the browser is a very good move. When you do open the browser (AFTER THE FIREWALL), use chromium instead of the default (unless the default is Chromium).
To find out, hit the raspberry, go into internet, and see if there is any other browser listed besides chromium. If there is, just use chromium — if there isn’t, you can use the globe up next to the raspberry.
So far I think the password I set was for the router and not an overall password for pi. I have a chromium browser and two others at present. I still have not tried to go on the internet because I am not certain I have an appropriate firewall other than the one on the router. I am mostly blind. I can change the password from the config tab but I have not yet done that. My immediate question is whether or not I have updated to any new raspberry system files before I do any internet activity. This forum approach is a little like postal chess. It takes time between messages.
Exactly — as I noted before, Wolf has instituted a 3 minute delay before a post that I make can be seen by you — but even then, you have to update before you can see it. If you immediately respond, it won’t be visible to me for 3 minutes, and only then after I update.
Before anything else, how are you doing and what TZ are you in? I am up and rarin’ to go, but if you’re on the downside of Nod, we’d be better off to find a time to reconvene.
Second, should we maybe try zaggin’ to the utree, which (AFAIK) doesn’t have the 3/6 minute lag?
As a general note, I haven’t been talking about any of your other computers or your router, I’ve been addressing only what you can see on your pi. If you somehow accessed passwd for your router in a browser, it’s because your router also runs Linux. But we’re trying to work on getting your Pi 400 up and running safely.
I don’t know what issues are happening, but I’m calling it for tonight.
The destination for Robert is a login/password that isn’t generally known on the internet, and a firewall that drops unwanted attention.
I know that it’s easy to get things booted without addressing these two issues, but I consider them a bare minimum for internet security.
— Further communication on Sunday’s thread.
Looks like some of your conversation is taking place on Sunday (Eastern time) so you guys might consider copying the whole conversation to Sunday’s thread & continuing there to get the greatest exposure from people reading along, like me.
(1) Bakocarl has beautiful posts of quiet meditation. I don’t like to be the person that bustles in with random overnight business. (Though I’ve often been the second person….)
(2) There is not much to be learned from this early stage — it is so dependent on whether a box is shipped on the 14th or 20th of a month, and stuff changes daily. Once we get Robert’s firewall up, things should relax and be of more general interest.
OK, thanks CT. At some point I might ask my new son-in-law to help me in setting up a Pi/Linux system. He was overlooking me as I was reading on the Q-Tree one time & got fascinated by a tech discussion unfolding. We’re hoping to red-pill him in increasing measures as time goes by 😉
It may be quicker to talk you through on the utree….
Thanks, CT. I’ll keep it in mind when/if the time comes–Blessings!
While I’m waiting to help Robert (which, BTW, it’s not your imagination — if you put in a comment that you can see, it takes 3 minutes and a reload before I can see it — per Wolf. A “round trip” from your thought will take a minimum of six minutes and two well-timed reloads. It ain’t me.), I’m going to try doing a bit of exposition — after I check what’s new.
OK. A firewall is merely a programmable gate in-between your computer and the network (anything plugged into its network jack, not necessarily the internet that is behind a router).
You will notice that Windows complains if its firewall is not turned on.
Linux has Uncomplicated Fire Wall — ufw — as a quick-and-easy firewall. On Ubuntu, it is shipped installed — BUT NOT TURNED ON. On Debian, including Raspbian and Raspberry Pi OS, it is not installed.
In a perfect world, you would have an operating Pi, plug in the internet cable, install ufw, then turn it on within a matter of seconds.
Once ufw is operational, its default behavior is to ALLOW anything INSIDE the computer to do what it likes to the internet, to allow continuing conversations, and to DROP anything from OUTSIDE that is not a continuing conversation.
If a firewall is not in operation, a remote process can try to mess with your computer — often several times per minute — until it can break in. After some weeks, we can swap your microSD card for one loaded with Kali and show you how this works — we can play with breaking into our own routers, our own computers, and the like — and maybe arrange demonstrations with our neighbors.
*** WARNING. BREAKING INTO STRANGERS’ COMPUTERS IS ILLEGAL AND MAY BE DANGEROUS IN WAYS THAT LAWYERS CANNOT HELP YOU. ***
Things that didn’t happen on January 6th, 2021 —
*On this date [February 28, 1890], Charles Kincaid, a newspaper correspondent, fatally shot former Congressman William Taulbee of Kentucky. The two men had a volatile history which began in 1887 when Kincaid wrote a story implicating Taulbee in an extramarital affair. Taulbee opted not to run for a third term, however, his work as a lobbyist kept him in close proximity to Kincaid who covered Congress for the Louisville Times. During the next few years Taulbee and Kincaid traded verbal insults. On the day of the shooting, House doorkeepers had to separate the two men. The “tall and sinewy” Taulbee allegedly warned the slight reporter—described in a contemporary account as “a little pint-of-cider fellow”—to arm himself. Kincaid later met up with Taulbee and shot the former Representative on the east staircase of the House Wing of the Capitol. The media widely covered the shocking event. “For the first time in the memory of man a gunshot was heard in the National Capitol today, and the marble steps of the staircase leading from the House floor to the restaurant below were stained with human blood.” Taulbee eventually succumbed to the gunshot wound and died on March 11, 1890. A jury later acquitted Kincaid on the grounds of self defense.*
https://history.house.gov/Historical-Highlights/1851-1900/The-death-of-Congressman-William-Taulbee-on-the-steps-of-the-U-S–Capitol/
Dirty RICE !

White House Deploys Susan Rice For Negotiations As Biden’s Agenda Stalls In Congress
link…
https://www.breitbart.com/politics/2021/10/01/white-house-deploys-susan-rice-for-negotiations-as-joe-bidens-agenda-stalls-in-congress/
10/1
the puppetmaster ( hi obama )…director of the WH domestic policy council. 👎
more, at the link.
the liar. 👺
Is one visiting the other in prison??? 😉
we can HOPE..
but I think it’s from the Benghazi daze.
🙂
Also something that did not happen on January 6, 2021 —
*On May 22, 1856, the “world’s greatest deliberative body” became a combat zone. In one of the most dramatic and deeply ominous moments in the Senate’s entire history, a member of the House of Representatives entered the Senate Chamber and savagely beat a senator into unconsciousness.
The inspiration for this clash came three days earlier when Senator Charles Sumner, a Massachusetts antislavery Republican, addressed the Senate on the explosive issue of whether Kansas should be admitted to the Union as a slave state or a free state. In his “Crime Against Kansas” speech, Sumner identified two Democratic senators as the principal culprits in this crime—Stephen Douglas of Illinois and Andrew Butler of South Carolina. He characterized Douglas to his face as a “noise-some, squat, and nameless animal . . . not a proper model for an American senator.” Andrew Butler, who was not present, received more elaborate treatment. Mocking the South Carolina senator’s stance as a man of chivalry, the Massachusetts senator charged him with taking “a mistress . . . who, though ugly to others, is always lovely to him; though polluted in the sight of the world, is chaste in his sight—I mean,” added Sumner, “the harlot, Slavery.”
Representative Preston Brooks was Butler’s South Carolina kinsman. If he had believed Sumner to be a gentleman, he might have challenged him to a duel. Instead, he chose a light cane of the type used to discipline unruly dogs. Shortly after the Senate had adjourned for the day, Brooks entered the old chamber, where he found Sumner busily attaching his postal frank to copies of his “Crime Against Kansas” speech.
Moving quickly, Brooks slammed his metal-topped cane onto the unsuspecting Sumner’s head. As Brooks struck again and again, Sumner rose and lurched blindly about the chamber, futilely attempting to protect himself. After a very long minute, it ended.
Bleeding profusely, Sumner was carried away. Brooks walked calmly out of the chamber without being detained by the stunned onlookers. Overnight, both men became heroes in their respective regions.
Surviving a House censure resolution, Brooks resigned, was immediately reelected, and soon thereafter died at age 37. Sumner recovered slowly and returned to the Senate, where he remained for another 18 years. The nation, suffering from the breakdown of reasoned discourse that this event symbolized, tumbled onward toward the catastrophe of civil war.*
https://www.cop.senate.gov/artandhistory/history/minute/The_Caning_of_Senator_Charles_Sumner.htm
“Senator Charles Sumner”
misread (briefly & hopefully) as Charles Schumer
spare the rod & spoil the child!
I noticed the hairy arm a few days ago when they were doing this Fake-Potus-gets-Fake-Jab- on- Fake-WH set fiasco.
Glad to see that I wasn’t the only one.
Other pics posted in the replies:




……….From the replies:
JeremiahG — “That’s about 12 ml too much of (product x) being injected btw. Perhaps at least get the sizing of the needle correct before a failed publicity stunt is launched.”
Russel4America — “That needle is much too long for the recommended intramuscular injection of 5/8”, 1”, or 1-1/2” depending on body type.
I call “False Flag”
MAGA-wake — Not only too dang long but too thick of a needle! I had to get yearly flu shots from my employer….no way did the needles look like that!
Don’t Gulag Me Bro! — Weird needle placement too…headed right for the acromio-clavicular joint (NOT the deltoid muscle, as is usual)!
Fleek — Plus…if you have ever had on a long sleeve shirt like that there is no way you can roll it up to your shoulder!
biden’s legs ???

or are those really Hillary’s as a stand-in ???
so hard to tell anymore. 
Wookie walkers 😉
👌
The eyes tell the whole story.
Window to the soul, ’tis been said.
Biden’s eyes have (had) life – a recognizable spark (someone IS home!).
Biden’s eyes look dead – no light at all (no one’s home).
IMO, different individuals.
Emerald, its not just the eyes…look at the ears!!
Something is fishy here, but I don’t believe any of the narratives as presented.
The left-handed vs. right-handed issue has been debunked because someone reversed the photo.
The photo below has to have been photoshopped. It is from Yahoo News. There is no way the arm was that hairy.
https://www.msn.com/en-us/news/us/biden-gets-pfizer-booster-shot/ar-AAOSB3z
https://img-s-msn-com.akamaized.net/tenant/amp/entityid/AAOSOVE.img
Likewise this one from CBS News:
https://www.cbsnews.com/live-updates/president-biden-covid-19-vaccine-booster-shot-watch-live-stream-today-2021-09-27/
In this Reuters video clip, the arm does not look like that.
https://www.reuters.com/world/us/biden-get-covid-19-booster-shot-monday-white-house-2021-09-27/
The video shows him speaking, then going over to get the shot. It is definitely him speaking. No one can impersonate someone that well. The arm does not look hairy-ape-like in this video. Someone is definitely off with some of the pics.
I do not think this is evidence of a body double, but it is definitely weird that a set was used for this event.
I also do not find anything significant about all the headshots of him. I noticed years ago when he got hair plugs. Likewise, years ago when he got some kind of facial work done — botox or whatever. I thought it stretched his skin and changed his appearance for the worse. He probably continues to get things done.
As for the needle, since it is from the hairy-ape-man pic, I don’t trust that it is accurate either.
From Sydney Powell’s deposition of Eric Coomer–
.
https://www.warroomforum.com/attachments/powell_deposition_25_sept-png.9474/
School Boards Ask Biden Admin to Treat Parents’ Protests as ‘Domestic Terrorism’
.
https://xyz.frankspeech.com/article/school-boards-ask-biden-admin-treat-parents-protests-domestic-terrorism
Sniveling weasels!
*insert string of cusswords right here*
Ht OT
(with video)
https://www.lifesitenews.com/news/if-you-get-the-pfizer-vax-youre-more-likely-to-get-covid-insider-leaks-fda-study/
Note the date on this one, not like they didn’t see it coming.
https://www.fda.gov/media/89036/download
White House Deploys Susan Rice For Negotiations As Biden’s Agenda Stalls In Congress
.
https://www.breitbart.com/politics/2021/10/01/white-house-deploys-susan-rice-for-negotiations-as-joe-bidens-agenda-stalls-in-congress/
Cindyseestruth
@AllTogetherNows
10h
·
**WARNING**
This is hard to watch. But adults need to understand what is CURRENTLY in your child’s school. This in Virginia, but this parent had absolutely no idea a book like this existed in her school system. I would be willing to bet similar books exist inside your child’s school system as well. This is pure filth.
You have been warned.
[video src="https://media.gab.com/system/media_attachments/files/086/550/531/original/bc87697820b379b2.mp4" /]
can’t even listen beyond 30 seconds.
absolute filth !
sodom & gemorrah
nauseating.
hard to believe we have devolved into such depravity….willingly.
IKR?
😳 😕
It’s sickening.
God Bless that courageous mom for speaking out!
This is probably why the School Board Association wants to have these protesting parents declared “domestic terrorists”.
😠
I can’t even imagine what it must be like to try to raise decent kids…and educate them properly in today’s perverted world.
and finding appropriate peers for them.
the influences are toxic and pervasive everywhere we look.
I feel sorry for the kids today…
…esp if they have Progressives and the radical Left for “parents”.
We need to start calling them “DOMESTIC COMMUNISTS”.
OMG. I got to the illustrations and had to stop.
If I found this in a school library they would be picking pieces of the librarian off the walls.
This is too much. It is too damned much.
It’s time for these vaxxed-up perverts to start dying off. The rest of humanity deserves it.
OMG.
A right and proper response.
I love how the parents are reading the filth out loud, putting it right in their faces. Toward the end she shows the pics to the board and says, “Can’t take what you’re” showing the kids? The crowd shouted, “Shame!” She also cited a statute and says the board should be charged with child pornography. Yes!
And Apples wants to search OUR phones while “ALLIES” of Tim Cook and his PEDO PATROL put this stuff in school libraries?
OUST TIM COOK!
OUST TIM COOK!
OUST TIM COOK!
OUST TIM COOK!
OUST TIM COOK!
I prefer the General Strike. The French have it down to an art. The Aussies are working on it (despite the news blackout)…. or whiteout…
jennifer aniston = 



….and none of that “turkey bacon” crap, neither!
They have beef bacon (yuck) over here, to keep our “Fachkräfte” (refauxgees) happy…. Funny thing, they’ve never done anything like that for the Jews, who seem to be able to fend for themselves quite nicely…
Oddly enough, they don’t buy it anyway… as well as most of the other junk the food companies/processors are vomiting out in the name of “diversity and equality and tolerance”…..
Does beef bacon taste like bacon AT ALL?
If it did, I might be interested just for “light-cooking” it, which is a bit dangerous with pork.
I’ve never tried it; the thought of it is nauseating… They also have beef salami and turkey+beef salami, again to appease our “guests”. The rest of us, I guess, will eventually have to endure “Like Meat” or ground-bug salami or some such.
I know they have Steak Tartare here, which is basically raw (Beefaholic’s answer to Sushi, I guess).
The T-B salami is HORRID. I bought a frozen pizza with it (by mistake)…. haste makes waste… Ended up peeling the T-Bcrapsalami off, adding some cheese on top of what was left of the toppings, and pepperoniwurst on top of that. Ten more minutes in the over (broiler) and all was well in Covfefeville again 🙂
I can’t figure out why they’re bending over backwards here to placate and/or please our invaders. They can neither be placated nor pleased, as they’re hell-bent on conquering and subjugating (Islam) us… We’re getting sold up the river by a bunch of crooks who can’t swim…
The invaders are very adept at human psychology.
They’ll start by making some mildly inconvenient (or even not inconvenient at all) demand, and be totally inflexible about it. It’s only a mild inconvenience, so it’s easier to just give in. Repeated iterations of this teach the victim that the best thing to do is just appease these people.
So now you have bogus food products like this. And even that isn’t a big deal…unless you can no longer get the real ones. But you can see the handwriting on the wall even then.
Yep. Trouble is, eliminating the REAL product is their goal. And they pursue the “Salamitaktik”, even over years, decades, or generations, if needed.
In an historic town not too far from here, they (ditib, a Moslem association run by Airheadman, though not openly) built a Mosque right next to a beautiful Greek Orthodox church.
Zoning laws be damned, in Ditib’s opinion. The Mosque was five meters or so (18 feet-ish) too tall, so building had to stop, if not reversed. Meanwhile the Greek Orthodox Church, built following all codes, etc., and its community well integrated in the German city and country, had to put up with the disruption of traffic, noise, dirt and debris, etc., from the mozzie construction site.
The city said “STOP”.
So the mozzies did.
And waited.
And waited.
And waited.
All the while their neighbors suffered under the disaster of a building site next to them; a building that looked like a portal into Hell, half-finished, and completely disfigured and disgusting.
And they waited some more. Ten years or so.
Finally someone in the city manglement said, can’t we get this problem to just go away?????
Around the same time the greens took over the presidency of Baden-Wüerttemberg, and Merde-Kuh opened to floodgates to the invaders…
Fearing problems, the city gave in, and the carbunkle (to paraphrase Chuckles the Klown) was completed… At least they weren’t permitted to have their minarets, which would have been even higher… nonetheless, the conquest which minarets signify was completed…
Give ’em an inch, and they’ll take a mile. If not our lives…..
Agreed.
Refusing To Fight The Culture War Will Mean Pain For Those You Love.
The left isn’t going to nicely make compromises and exceptions for you, no matter how far the bench you’re sitting on is from the field.
.
https://thefederalist.com/2021/06/21/refusing-to-fight-the-culture-war-will-mean-pain-for-those-you-love/
Agreed. If we don’t push peaceful resistance to the limit, it will be war or slavery.
More fear mongering.
Advocacy disguised as ‘science’: ‘Intergenerational inequities in climate extremes’ – Watts Up With That?
No…just…no
https://twitter.com/hardhatintellec/status/1444147109373124613
BRAIN BLEACH, POR FAVOR!!!
That’s too true.
They’ll call you uncultured because you don’t think a painted color gradient is art. I’ve seen that argument on the internet, and it boils down to “if you don’t see it you don’t understand.” Which is an argument through intimidation.
This is FAR in advance of the others here, actually…because it does actually say something.
Whether it’s anything worth saying is another story entirely.
but artistically it sucks.
I have always hated it.
Yeah, it’s a technical disaster.
I remember the painting differently.
As in, a painting using small dots / pixelated points of color, wherein one sees the general picture, but one must look DEEPLY to recognize the panic in the girl’s expression.
It was HIDDEN to the viewer, unless one zeroed in on the facial expression.
I found it powerful in its subtlety – but I searched for this rendition (which I thought was the *original*), and I couldn’t find it.
Perhaps the one I remember seeing FIRST was actually a re-do. If so, I found it WAY more powerful than the original.
I guess you’re talking about Pointillism ?
but Munch was definitely not a Pointillist painter…he didn’t do the “dot” style..
he was a German Fauvist painter.
Fauvism is the genre…a form of German Expressionism.
post- Impressionist.
you must have seen a copy.
Fauvism….
Dangerously close to Faucism (and Faux-Xi-ism) 🙂
I’ve seen a number of images of it online, but only in white (or black and white)…
One might call that painting (I don’t like it either) an exercise in pointlessism… (OTOH I like Dali’s dripping clock, as it were, because I’m always a little bit late it seems…
Dali is Surrealism.
Yep…
The Persistence of Memory
Salvador Dali
Never could figure out what that off-white thing is.
Well, if you start by noticing the eyelashes, it appears to be a distorted and incomplete face, with the watch lying across a cheek.
Not only does it seem to be a face, but one that is distorted into a sleeping swan-ISH thing with a wing-ISH thing where the tail would be – as in “time flies” but we sleep through it.
Abstract for sure.
example of classic Pointillism…
by one of the Masters of the genre…French post-Impressionist George Seurat (1859-1891)
I am not a fan of Pointillism.
just giving you an example.
One of the many astonishing things I learned at the Musee d’Orsay traveling show was that Seurat could actually paint. He did about 60 conventional small paintings to prepare for making https://en.wikipedia.org/wiki/A_Sunday_Afternoon_on_the_Island_of_La_Grande_Jatte .
The painting itself is in Chicago, but the studies may have been loaned by the d’Orsay — and they were displayed to show how they were incorporated into the larger work.
Anyone remember the Kenner Girder and Panel (and Highway) sets???
The small post/cap was designed to be painful; LEGO was late to that party… (the sets were GREAT fun, though)…
These HURT bigtime (they were kind of a plum color in my set):

There are, BTW, about four “originals” of this painting.
I’ve heard it said that it expresses the artist’s feelings about his mother’s cerebral hemorrhage.
Onement VI

Barnett Newman, American
color field abstract expressionism
should be titled B-O-R-ing
sold for a whopping $43 million in 2013
wanna see smore doozies that sold for gobs of $$$, go here…
https://www.the-artists.org/10-weird-and-ugly-paintings-worth-millions/
exception…Rothko’s work…very dynamic up close…and some of De Kooning’s abstracts that weren’t figurative (his women are awful).
sometimes it just is what it is.
Looks like (how) a lot of John Adams’s music sounds… minimalist, in this case what a FORTRAN programmer would call NAN – not a number… too low to represent…
One might say the “artist” blue it….
(Ducks and runs from tubes of paint, dried-out brushes, and dog-eared scores of “Nixon In China” 🙂 )…..
Verse of the Day for Saturday, October 2, 2021
✟
“And if I go and prepare a place for you, I will come again, and receive you unto myself; that where I am, there ye may be also.”
John 14:3 (KJV)
Thank You, Jesus, for blessings received and prayers answered!!!
BE MY VOICE
‘You’ve got to be really hurting to do this kind of destruction’: Pro-abortion vandals spray-paint church, topple and trample crosses
.
Mark Evevard — youth director for Sacred Heart of Mary Catholic Church in Boulder, Colorado — described the wave of emotions that hit him after seeing for the first time the pro-abortion destruction that vandals wreaked upon his church, KCNC-TV reported.
.
https://www.theblaze.com/news/pro-abortion-vandals-spray-paint-church
PRAYING ON THE ARMOR OF GOD
Father God, I now follow your command to put on the full armor of God, because my battle is not against flesh and blood but against rulers, authorities, the powers of this dark world and against spiritual forces of evil in the unseen world.
I first pray on the Belt of Truth that it may be buckled around my waist, may I be centered and encircled by your truth dear Lord. Hem me inside all that is true and right, and may I be protected and held up by the truth of your living word, in my Lord Jesus name.
I pray on the Breastplate of righteousness, please protect my vital organs and my inner man, cover my integrity, my spirit, and my soul. Guard my heart for it is the wellspring of life, please strengthen and guard the most vulnerable places in my life with that which is right, good, and noble that I might not receive a fatal blow from the enemy, in my Lord Jesus name.
I pray on the Gospel Shoes of Peace. I choose to stand in the shoes of your good news, and on the firm foundation of my Lord and Savior Jesus Christ, the solid eternal rock. All other ground is sinking sand, I pray that I will not slip or fall, but that my feet would be firmly fitted on your lordship, my Lord Jesus. I choose to stand on you, so that the peace of God, which transcends all understanding will guard my heart and mind in Christ Jesus, the eternal Rock of Ages. I receive your holy peace now my Lord, from the sole of my feet to the crown of my head, in my Lord Jesus name.
I pray the Shield of Faith into my hand now. As I take up the shield of faith, I ask that you might extinguish every dart and arrow, that is launched from the enemy to take me down spiritually, physically, mentally, emotionally, and every attempt of the enemy to destroy my joy. I ask that my faith in you would make it flame out. Extinguish every flaming arrow that would come against me, my life, my family, my home, or my ministry. May my faith always be out in front of me like a shield. Give me the courage to “faith my fears” by choosing to walk by faith and not by sight, in my Lord Jesus name.
I pray on the Helmet of Salvation, that you might protect my mind from the thoughts that can lead me astray. I choose to take every thought captive, and arrest all intentioned ideas and motives that would harm others, or distract me from your holy will for me. I submit every captured thought to the Lordship of my Lord Jesus Christ, and ask that you would imprison those thoughts that are not of you my Lord. Transform my mind and renew my thinking that I may think God thoughts, and have a sober mind that is focused on your glory. Please protect me from being double minded that I may allow my mind, I reject to live an earthly life, because I choose to live a holy one, governed by you My Lord Jesus, the prince of peace, please have my mind to be saturated with the holy mind of Christ, in my Lord Jesus name.
Finally, I take up the Sword of the Spirit which is the holy word of God, I pray this powerful offensive weapon into my hand, and ask that your holy word would be fitting for every encounter I face. As the enemy gets close to me, please give me the insight, wisdom, and skill to wield the word of God to drive away the enemy, in my Lord Jesus name.
May the enemy and his team flee from me, upon hearing the word of God spoken by the power and direction of the Holy Spirit. Give me the sword of the spirit to cut through the wiles of the devil, so that I may discern the schemes of the enemy when he is near.
With all kinds of prayers, supplication, and intercession I pray to you my Lord God as the one who fights my battles. Now that I’m in your holy powerful armor, I walk away covered and ready to face my day as you go before me, and please protect me in the midst of the spiritual warfare in this unseen world, in my Lord Jesus name.
Thank you my Lord, for the spiritual weapons of armor and prayer that you have given me. It is written no weapon formed against me shall prosper, and you will refute every tongue that accuses me.
Thank you Father God, my Lord Jesus and the Holy Spirit, that I am more than a conqueror in my Lord Jesus. I pray all of this in the mighty name of my Lord God and Savior Jesus Christ, amen.
–a resounding THANK YOU. Always enjoy and welcome your set of posts—find praying these prayers out loud is very restorative to my soul. And today—considering the posts that came just before— these prayers are especially appreciated and most welcome.
I wish you continued frequent and many blessings—L.
Most welcome, L – So good to hear – you just gave me a blessing – and I so appreciate it.
God Bless You Real Good!!! <3
Funny, but sad at the same time because of how true it is.
Yep.
Common sense isn’t…..
Ground Report – My 9/27 Pneumococcal Vaccination (PPSV23 for 65 & older)
Reminder of a post I made earlier in the week about my wife and my Medicare Wellness exams. I indicated we rejected once again the COVID jabs and added the flu jabs to the rejection, much to the dismay of our NP who is obviously rewarded in some manner for pushing jabs. I surprised her by agreeing to take the PPSV23 for its benefits relating to my personal and family bloodline health disorders after having researched it.
For those considering it – it is powerful and was adjusted from the original 2019 version to reduce side effects. It is a true vaccine provided by Merck that is one and done now. I experienced all four of the most common side effects of muscle soreness, fever, fatigue and pain. All were relatively minor, however, I am a relatively healthy, strong guy for my age. The soreness existed at the location of the jab for 3 days and is gone. For two days it hurt to even be touched, which my grandson did not quite understand when he would lay his head against it. Of course I let him anyway. The fever was minor – a half degree above normal for 24-36 hours. I was very fatigued for 48-72 hours, but returned to 100% in the 5th day. Felt a little brain fogged for a few days. The pain was more like random pin pricks in one side of my torso for a few days. Similar to what I felt when I had Shingles a few years ago. Aggravating, but not overly concerning. They were gone in the 4th day.
So, I would do it again barring knowledge that they had injected nRNA into it. That would be another, AW, H–L NO!
For some reason our NP has never followed up on how I am doing since she ordered that jab. Guess she has moved on to her next victims. Doubt I will get a Christmas card from her this winter if I live that long. Come to think of it, maybe that is why she was so happy I agreed after all the previous jab rejections. 😂
Not only that, he fought with William Wilberforce, a few times physically. Brown was pro-slavery and Anti-Christian as well…
Kamel-Hoe would do well to, say, offer some reparations to make up for HER FAMILY’S past…
[Of course I don’t mean that: we shouldn’t have to pay for what our ancestors did or didn’t do. GOD will deal (and has dealt) with that]….
SteveInCO
“antineutrino does condescend to react with a proton”
THIS type of explanation is why I read through the physics openers you post, instead of scrolling past with eyes glazed over.
Thanks!
I know at least three people are reading them!
(Not counting myself when I skim through later and spot all the mistakes. This time I wrote it in a Linux app something like notepad, and when I pasted it into the post editor ALL of my paragraph breaks somehow disappeared. I had to go find them all and put them back in, and probably missed one or two.)
Make it four, Steve.
I’ve known much of what you’ve posted, but there are a few “blanks” which you’ve filled in which I very much appreciate.
Cheers, and TY!
vi and EMACS for the win 😀
I could have written it in my own text editor but it doesn’t display with word wrap (it’s for writing code).
ICYMI – Nancy’s “slip” …
She was notorious for calling Trump “Bush” too (remember?), so I don’t think this necessarily means anything other than that she’s got that Senior Moment thing going on.
Don’t you think she would have walked it back by now if it was a “slip” ?
Can’t fault that logic.
YAY, MUFFINS!! 🤸♀️😋🤸♀️
Periodically *could* mean hourly…so yes.
😂
National School Boards Association Begs Biden To Label Outraged Parents “Domestic Terrorists” and Use The Patriot Act Against Them
.
https://www.thegatewaypundit.com/2021/10/national-school-boards-association-begs-biden-label-outraged-parents-domestic-terrorists-use-patriot-act/
My response:
YOU, ma’am, are DOMESTIC COMMUNISTS.
[video src="https://media.gab.com/system/media_attachments/files/086/550/531/original/bc87697820b379b2.mp4" /]
#FJB – FLUSH JOE BIDEN 😂
200
sheetscheats per roll.brought to you by the same geniuses who want the butthole spy/health camera–puke!
It’s a miracle! I’m in this morning!
Just parking this here.
And apparently, the stock is being shorted.
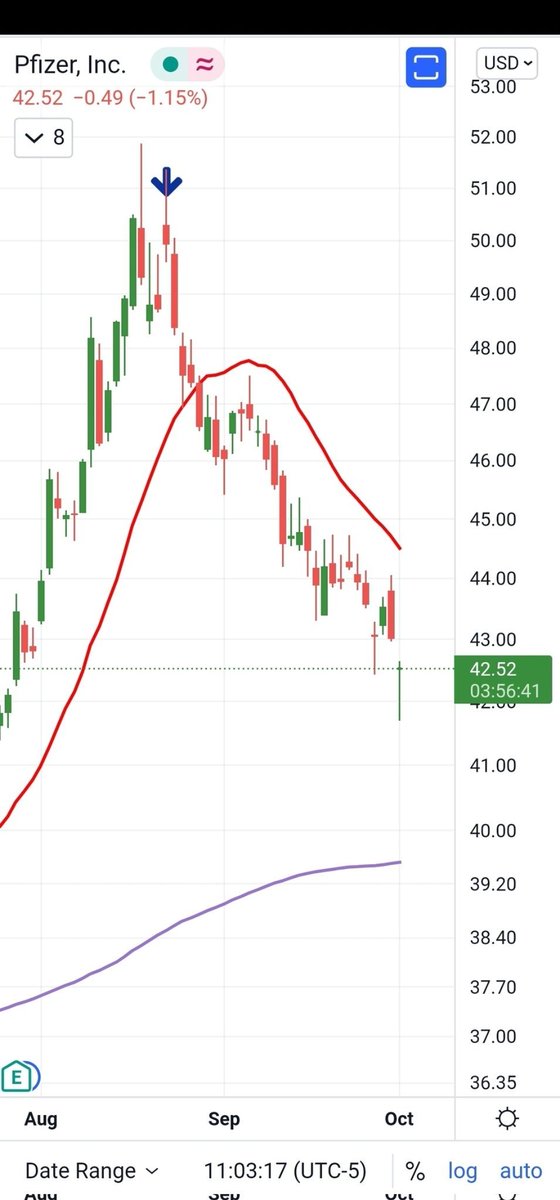
Something like Sauros, Bøzøs, BillyGhoul Gates, the Røthschilds, and the cabal in general would do.
Squeeze out profit every WITCH (not a typo) way they can…..
And they’re probably shooting for killing 212 million per day…
Hello friends, I am back after a long hard hiatus. Here is my new post on my blog if anyone is interested. Hope to talk to you all and converse soon. Prex
http://politicalprognosticator.politics.blog/2021/10/02/back-in-black/
Glad you are back!
Thanks, happy to be here!
Great to see you. Am reading your post that is on yesterday’s daily.
https://www.theqtree.com/2021/10/01/dear-kag-2021-10-01/#comment-809435
I pray that FINALLY, quietly at first, this is the beginning of the END of the Cabal. I suspect a TON of flips and “covidicide” for the lower Cabal members.
👍👍👍👏
👋👋Welcome Home! 🏡🥰
Thanks!
👍
prex
Yay! Great to see you again!
Bringing this over from yesterday’s thread as I would like to respond:
DePat response:
If Sullivan is the next to be indicted that would be interesting. However, I have seen reports that it’s going to be a tech executive, and not one of the names we know. Somebody high up at either Alphabet or Microsoft. It has to do with the software used to pull it all off. Going straight to the White House…not sure that’s the onion peel you want as a next step, not if the next thing on the audit agenda is to examine the routers.
I’m also seeing that it will begin to make sense after the 14th. Now, here’s the thing about that…fixed dates in this game are an anathema. I have no confidence in it, but that tidbit is out there.
In my personal opinion, any number of sources out there claiming insider status and giving “intel” may well be being used to spread dis and misinformation. Too much of it is contradictory, and to be blunt about it, the leaks from all sides – the REAL leaks – essentially stopped in 2018. I’m wondering how many canary traps were laid and how many are in the wings ready to be sprung.
I also doubt seriously the movie we are watching bears any resemblance to reality other than the side effects of the shots and the failing hospital protocols. Okay, the censorship and communism, yes, but I doubt seriously the avatar being called Biden is anywhere close to Washington.
This is quite the time to be alive, that’s for sure. Glad to see you back.
Maybe Eric Schmidt?
I posted (September 19th) about him meeting Julian Assange and Garble’s deep connection with Hell-the-BEAST…
A few years back Julian Assange published an interview of Eric Schmidt “When Google Met Wikileaks” . There’s an article about it, “Google Is Not What It Seems”, https://wikileaks.org/google-is-not-what-it-seems
From the referenced article:
Garble and the DEMONRATS have been in bed, so to speak, for at least a decade, in not more… The article is well worth a read…
No, not Eric Schmidt. It was a name I had never seen before.
Welcome back 🙂
Happy to be back!
Good 🙂
I’ve been wondering about you! Nice to see you!
Likewise
This is Great!! I usually re-post DP’s (and others’) memes to Gab and this one is a frequent flyer which usually gets 25+ likes and reposts minimum every time. Looks like someone has expanded on Wolfie’s original. Way to go Qteam!!
Raven Constantine
@Artraven
6h
·
950 likes
66 comments
691 reposts
EXCELLENT!!!
I highly recommend that one watch the last half-hour of today’s Bannon’s WarRoom.
This final guest (from Peru) gave better intel than I’ve had in a LONG time on the events precipitating in South America.
The guy (couldn’t get the name, sorry! – update – Frank Gaffney?) speaks with aplomb, and with sincerity and purpose, especially about the Shy-Knee capture of so many countries in SA, owing to the commie infiltration of their social and political arenas.
The playbook is EXACTLY the same as is being used here in the States.
It’s a WORLD WAR, in case anyone hasn’t figured that out yet!
Please watch, if one is able and willing to do so. I found him a pleasure to listen to.
Stunning stuff.
We are NOT alone!
They stole the election in Peru too! Another subtle yet for certain Communist takeover. CCP is taking over South America which is driving their populations northward to here. Those Border Haitians had been living in SA for 2-4 years.
Oh, AND…
.
.
.
PETER NAVARRO 2028!!!
This man is a frikkin GENIOUS!!
And Bolivia last year. The party of the ex-president, who fled the country, unexpectedly, won and that criminal dreck (he was Chaves’s pal) returned to the country.
They trying to do the same with Bolsenaro of Brazil, where they trying to get the socialist Lula, who was convicted of corruption, back in.
I can’t decide if Chy-NAH is really doing this on their own or if they are being used. I lean toward being used.
Yep. To the hyper-rich, EVERYONE is a tool… even countries and peoples…
That’s why they can act with such coldhearted impunity, pursuing their soulless goals and shaking their demonic, but mortal fists and immortal and INFINITE GOD HIMSELF…
They will eventually get the Nebuchadnezzar cure…
I think it started out that way, but it’s not where it’s going. IMO the ChiComs are smarter and more patient than the people who think they’re mastering China. The closest thing to a direct confrontation between the Cabal and China was Hollywood. China suckered them all, IMO. When Hollywood starts putting out anti-British, anti-Jewish, and anti-Euro-globalist content, and eventually overtly pro-China content, you’ll know who’s really in control.
I was wondering about that w/ the genetic tendencies of “covid” to target minorities, including Native Americans, who are usually showing up as East Asian in DNA results. China knows how to play a long game too. It would be Amazing if the Chinese as a people, especially their leadership, bowed to Jesus’ Lordship in this life (all will bow in the next/eternity). That would be an Epic plot twist!!!
Reminds me of “Deteriorata” that was popular in the 70s… 🙂
🤣
Looks like the buck(s) stopped there…
(ducks and runs)…
Top down CORRUPTION.
Communism and CORRUPTION always travel the SAME PATH.
See Nancy Pelosi.
See Dianne Feinstein.
See the Cuomo Crime Family.
I was intending to post a link to an article about this terrorist yesterday, but got side-tracked…
Glenn Beck does it much much better, here…
have a listen…
Oct 1
introducing 2 of biden’s recent…and RADICAL… nominees..
Senate, unfortunately, has confirmed Tracy Stone-Manning to lead the BLM (Bureau Land Mgmt) in spite of her close involvements with ECO TERRORISTS…just listen to what she’s about…
I can not stomach this shit anymore !
but wait…next up…
Saule Omarova... biden’s pick to lead the Office of the Comptroller of the Currency…
geez
Omarova (what a hellish name) really really really hates private banks.
in fact, she wants to end the banking system, as we know it.
better NOT call Saule
In nut shell after you peel away all the convoluted language all Banks are to be federalized so that all money is controlled by the government. They then set limits on how you spend your money to include making access to your money dependent on your social credit score, while they also digitize the monetary system, creating the cashless society so there is no choice in the matter, meanwhile they can introduce negative interest rates. I’m sure though if you have something really important you’d like to buy there will be some back door procedure where you can cough up an extra 10% for the big guy and get what you want anyway.
That the Bidenese would make this pick tells you this is what they want regardless if Omarova is gets approved or not.
🤑 💲 🏧 🤑 💲 🤑
Hmmmm…..
Lemme guess.
Could she be a RED ChiCom plant????? And I don’t mean dope (although she could be that, too, merely a puppet)…..
They’re fucking communists. It’s that simple.
America did not vote for this shit.
Wadda ya think…luggage commercial? or freaking hero?
(Guy demolishes three ticket kiosks at the airport as people stand around shocked, apparently they did not take his QR code.)
https://twitter.com/Lenabellalou/status/1444074257412206596?s=20
Is he available for elections ? HIRED.
Those damn things never work for me either.
interested in American gun legislation ?
sharing this link…to NRA- ILA
https://www.nraila.org/articles/20210927/biden-administration-asks-the-us-supreme-court-to-judicially-nullify-the-right-to-bear-arms
article from 9/27
biden’s amicus brief, NY.
lots of other relevant, timely articles on the subject, FYI.
The tsunami of destruction keeps coming at us.
The lawfare ministry cranking out more and more ..
.
https://wiredailynews.com/2021/10/02/gop-ohio-gov-dewine-is-helping-pelosi-keep-her-house-majority-with-this-devious-new-plan/
In case you don’t have time to read this, Dewine is going redistrict Jim Jordan out of his seat along with 3 to 4 more Republican seats in Ohio. WTF!
They’re corrupt and driven by evil. They won’t stop until..I don’t know the answer to that.
Fair and balanced is determined only by them .
YES. This is WEAKNESS on DeWine’s part. Let me explain.
The DEMONCRATS have been running a “gerrymandering” theme very strongly. They run it nationally, but DeWine is a serious target. DeWine is easily manipulated by the Democrats – he is very proactive on political angles, but that bites him in the ass when he responds to nearly EVERY SINGLE Democrat manipulation. He is not good at facing challenges from the left.
DeWine is facing a tough challenge from the MAGA center with Renacci, who is an AUTHENTIC pro-business AND pro-freedom candidate, but not by any means a hard right candidate. Renacci is just a straight-shooter who is a lot like DeWine, in reality, but with a BACKBONE.
DeWine IS actually very pro-business, but by being WEAK on the mask mandate issue, he hurt himself BADLY for re-election – as the Democrats planned. As you can see, he’s a “survivor mentality” RINO – he is NOT a “MAGA hero”.
DeWine’s new campaign theme is that he LED on COVID and the pandemic – that he “took action”. It’s a bit of a farce, but it’s actually smart, because it’s “half-true”, in that he committed to a leftist folly against all advice from the right. What he did was played to the left on masks, but played to the right by exempting businesses to the greatest degree possible – but not all by his own doing – a lot of that was PRESSURE from non-RINO GOP.
I suspect that DeWine is trying to LEVERAGE the GOP for support with this threat. That is NOT SMART for many reasons, not the least of which is that it is “penny wise, pound foolish” on the GOP’s future – even the machine RINOs. There is a better way.
DeWine needs to become what he’s trying to sell, which ironically is true MAGA like Trump (even if he doesn’t want to say it). Moving away from mandates of any kind as a THEME and as a REALITY is how to do it. What would Trump do? Yeah, that’s what he’s selling – “Leaders don’t sit on the fence”. Trump took action on vaccines, even against the odds. Now, nobody wants to say that DeWine fell on his ASS off the fence with lockdowns and the mask mandate, but that’s the truth. He actually HURT business terribly with that. He only kept businesses on LIFE SUPPORT – and Ohio lost a lot of smaller ones. But a LEADER LEARNS – and admits the truth, if only to self.
We all got CONNED by the Dems on COVID, but we don’t have to say that. What we have LEARNED is that MANDATES DON’T WORK – they’re counterproductive – they lead to BAD STUFF – they help the Democrats. Masks have consequences – including elections, where DeWine needs NO MASKS, or the Dems are going to machine back into power once again with fake elections. You think they’re gonna try Philly in Ohio’s big 3 cities in 2022? I DO.
DeWine is free to say that he did preserve business, but that the SCIENCE HAS CHANGED and now we know that we didn’t need masks, and that businesses would have been better with LIGHTER PROTOCOLS like SUGGESTED social distancing, hand washing, etc., but that we still lost a lot of businesses due to lockdowns and masking. This is a FACT.
Renacci knows this – that DeWine was good for Ohio business with one hand, but terrible for them with the other. Renacci would be GOOD for business – authentically.
DeWine had depended on COVER by the GOP (legislature, mostly) to say “my hands are tied” on masks, etc. IMO it would be better to say that he’s changed on the issue – that he helped business during COVID, but that he’s learned that lockdowns and mandates are simply too hard on business, and that any new pandemic will be handled more smartly, because that’s what a leader does. His goal in any future pandemic is to PROTECT ONLY THE VULNERABLE – to stay with the REAL science – to keep the state MORE productive.
I think this will sell, too, because even the COVIDIANS in Ohio now seem to be very non-compliant on masks. Nobody wants them back. Ginther tried to bring them back, and they do nothing but hurt business. Very poor compliance, and where there is compliance, business suffers.
MASKS ARE A THREAT TO BUSINESS. They don’t work, and even if they do, it’s at too great a cost. Gotta get rid of them. “We’re smarter than masks.”
Therapies are the way forward. Like they always have been. Vaccines were an industry GRIFT to get a green light on gene therapy. They’re done. Move on to where the industry is going – therapeutics.
Well Put Wolf!
Thanks!
I really don’t think DeWine is actually trying to help Pelosi – BUT HE’S HELPING PELOSI.
The GOP machine wants to hold Ohio, and I totally get that, and they WILL hold Ohio with either DeWine or Renacci, but not if they make too many stupid mistakes like giving Pelosi a boost due to infighting. The Demoncrats are CUNNING. Prepare for their next THREATS. That preparation needs MAGA strength and MAGA savvy.
You see THAT at the ground level. Ground level GOP is a TIDAL WAVE building across the ocean. We’re DONE with these fucking communists. Part of getting fair elections again is STOPPING THE MASK BULLSHIT.
I hope you get Renacci. Dewine is duplicitous and reminds me of a bully’s toady.
Fingers crossed!!! Renacci would really send a message.
The Flu Shot Stupidity in [Market-Ticker-Nad]
OMG Denninger completely gets covidism. I don’t know why I’ve never stumbled on his website before now so thanks so much for the link!
https://rumble.com/vn78v9-joe-biden-boss-of-the-biden-crime-family-rudy-giuliani-ep-175.html
This might have been up before, but this is from German pathologists who has performed autopsies of people who had died shortly after being jabbed.
https://freewestmedia.com/2021/09/22/lymphocyte-riot-pathologists-investigate-deaths-after-corona-vaccination/
My «incline-o-meter» has gone another order of magnitude more negative….
https://rumble.com/vn76k5-trump-slams-media-for-ignoring-project-veritas-stories.html
Lick it, Joe! It may be a giant malted milk ball!
WOW!
https://beforeitsnews.com/prophecy/2021/09/new-stew-peters-dr-carrie-madej-first-u-s-lab-examines-vaccine-vials-horrific-findings-revealed-2524291.html
Can we imagine if we had an honest media ? The greater public would be shocked and demand answers, demand a halt,demand accountability….but we don’t have that.
Exactly. If we had an FDA that was not controlled by the industry, they would be examining this stuff and PUBLICLY stating what they were finding.
Because there is a LACK of good information from this corrupt regime and its TAINTED, COMPROMISED, and CONTROLLED agencies, we are inundated with weak information, misinformation, and disinformation.
And our stupid media, which actually KNOWS exactly the right people who should be examining vials, does NOTHING.
16 to 3 against boosters at FDA, and simply overruled by Fauci’s butt-girl, Rochelle Alinsky, at CDC.
No we don’t. It IS a shame.
Thanks! I did look at the video. Sadly, this is not terribly convincing at a scientific level. The macroscopic appearance of a complex mixture of nanoparticulates as they warm up and fall apart (but then the pieces do their own thing) is just going to be a mess.
She’s not a specialist in this kind of thing. And even more sadly, there are VERY few people who are capable of doing the work. Almost all of them are in the industry.
True… she couldn’t identify what she saw…..
But what she saw looked pretty bad.
Yes – but lots of things look weird under a microscope, because the laws of biological and physical growth are often similar and follow the same mathematics. Thus, I used to grow crystals that absolutely looked like some kind of alien plant. But they were not – they were just organic molecules crystallizing as best they could. Not looking like salt or snow – looking like wrong-colored FERNS, MOSS, LICHENS, and LEAVES.
Politics is Downstream from Culture: YouTube reaction videos
The following reaction video is a fantastic example of why I think independent rap artist (I stress the word ARTIST) Tom MacDonald is so extremely important to the culture war.
The video stars the Cartier Family featuring 3 young Black men. Immediately, you notice a BLM poster on the wall behind them. The song they are reacting to is Whiteboy by Tom MacDonald.
The fact that these kids were open minded enough to watch the entire video…respect! They did not agree with everything Tom said but did agree with much! And notice how the discussion evolves to the end of the video…Tom says some things that kind of stops them in their tracks.
The point is not that we might not ourselves agree with everything Tom raps about…the point is that his MESSAGE is starting genuine conversations! And Big Tech has noticed evidenced by the suppression tricks and attacks he has received from trolls and jealous fake woke artists who wish that they could compete with his INDEPENDENT success.
Tom’s is a rags to riches story…a REAL one. Not to say he grew up poor…but he fell into a hole and lived in the hood in LA…broke…addiction…alcohol almost killed him about 5 years ago.
Together, with the help of his now girlfriend Nova Rockefeller they were able to turn the corner…now Tom is making millions working his ass off….while still remaining true to who he is. The Left does not want these types of genuine success stories out there undoing their years of brainwashing.
Tom is a pioneer…and the powers that be do not want it to become a trend.
As you may have gathered…I am a Tom MacDonald fan. Bought all of his CDs and he signed the cover of each one.
I think Tom is a great artist with not only his rap…but his message. I love that he takes on the industry face forward. Tom could take a big contract but gives them the finger.
Now this next video may not be everybody’s taste in music…but I post it as another example of how Tom’s message is dangerous to the Left’s agenda.
This one is hosted by the beautiful Asia and BJ. They are reacting to Tom MacDonald—No Response<<<I do not even like rap in general…but Tom shows all of his haters that he is truly a world class artist! He goes off for over 5 minutes of straight hard rap that even metal-heads could appreciate. 😎
Again, why is this important? The message and perhaps more important…the CONVERSATION…we can disagree on the details. Just these two reaction sites alone have over 150k followers together…most new followers from reacting to Tom MacDonald songs! Some reaction channels have MILLIONS of followers…many also review Tom’ work…reaching audiences that need to be reached!
If you know some kids listening to the mainstream rap that Tom says is destroying kids lives and even putting them in the grave…glorifying drug use…sex….money…INDUSTRY POISON! Try to slip some Tom MacDonald in while they are not looking. 😉
Shout out to the HANGOVERGANG! Tom’s loyal fans!
We need variety in all things, music too. Freedom to express ourselves and all that. Any avenue to get that message across is great.
Forgive me…just one more…I promise. 😊
This is Asia and BJ again reacting Tom MacDonald’s masterpiece ‘Don’t Look Down’. If you like Rock and have never liked rap…Don’t Look Down just might turn out to be an exception to that rule for you.
Tom is showing all sides of himself…the vulnerable to the self-glorification (but that is only to trigger his haters…notice the laugh when Tom calls himself a superstar). That said, Tom talks about his addictions…cleaning up and working hard…success…Don’t Look Down to the people that are like crabs in a bucket with their claws pulling you down.
The song ends in a crescendo that…I do not care what genre of music…EPIC! 7 minutes of pure 🔥 . Now remember…Tom gets a LOT OF HATE…so he does drop some F-bombs aimed at those that want to see him fail. BUT he is nice enough to censor them in the video.
Unfortunately, you will most likely never hear this song on MTV or radio…only zombies need apply.
I do not know of any truly independent artists in ANY genre that are as influential in a positive way that are having the impact that Tom has currently…and he is only getting started.
Btw, Tom’s new song America trended #1 on HIP HOP yesterday, and #2 all genres. I do believe that was digital downloads…YouTube, Spotify, Tidal, etc….NO LABEL…
If you do not have the time to watch the video…just check out some of the comments below the video. Tom is reaching a lot of people!
I agree with ya JW. I’ll listen to him and through that found other rappers that are anti woke too. He’s definitely making waves in the industry, the kind they don’t want though. And as you say when he spews out truth the blacks listen IMO. Thing is these rappers dis each other all the time to get respect from others, I didn’t understand it at first but after watching what others said when they did the analysis of each other I got it. His buying the NFT to Eminems Slim Shady and coming out with Dear Slim was a marketing genius move.
It’s a whole different culture for sure.
The attention to detail right down to the car with mud and weeds…water rolling down the windshield as if it had been pulled out of the water. He and Nova are a great team!
Some of his best stuff he only releases on his CDs…YUP he still believes in a good ol’ hard copy…just like me.
General McInerney
BREAKING Trump sues Twitter, seeks account reinstatement ahead of 2022 midterms.
Ground report. Typically, I’d call this good news.
Gas prices have dropped 0.22 over the past two weeks.
True, I think it is normal to drop a bit with the end of summer, school back in session.
BUT, I suspect it is folks out of work, not driving.
Interesting. Course here no drop and plenty of 24hr. Went to wally world. Tons of empty shelves. Not sure if its bc its Saturday m, but also not so many being stocked. Bottled h2o decimated. Pasta, crackers, paper plates, cheaper meats(big steaks and roasts arent selling anywhere), frozen potatoes, anythibg for school like juice boxes and snack size anything, and halloween except candy.
The amount of candy, cookies, and crap they had like that was sickening .
The shelves empty here are sports drinks more than anything else.
Ive seen that too, but they were fully stocked today. Really need juice boxes but luckily ive got him satisfied with ice water mostly.
Have you tried getting juice concentrates at the grocery and mixing with sparkling water for a special treat?
You know apple cider and ginger ale is good.
I havent done what you’ve suggested but i may do it. I like the flavored sugar free carbonated waters and he likes to have the smaller ones with juice. But ill try this too.
Ht P.win
https://www.faa.gov/licenses_certificates/medical_certification/
So basically a 48hr no fly rule after receiving any one shot of any of the vaxxx’s for medical airmen who are certified.
This appears to me at least as if one small group in the military is paying minor lip service to the Ft. Rucker Flt Surgeon. Likely expecting other authorities to follow suit so they can all get on with serving the beast as soon as possible. She must of had some effect to get them to say, “See we heard you, stay home for two days and then shut up and get back to work. End of discussion.”
Sorry if this got posted already. But they are right. And when the USA vetoes. Oh boy.
We shouldn’t veto, but given the regime claiming to be in charge….
Ikr….
This Dr., goes right up to the line but doesn’t step over and get himself banned lol.
It was him that was pushing Vit D3 in early 2020, caused me to buy some.
https://citizenfreepress.com/breaking/dr-john-campbell-deep-dive-on-indias-success-with-ivermectin/
“COVID-19 Enterprise Fraud Construct Timeline: Major Dates, Events, Entities & Legislation”
https://politicalmoonshine.com/2021/09/29/covid-19-enterprise-fraud-construct-timeline-major-dates-events-entities-legislation/
Not a fan of rap, but at least I don’t have to merge into the slow lane to understand this one.
His youtube is “samson”. I enjoy hearing the rebel yell getting louder.
A theory by Lin Wood: https://t.me/s/linwoodspeakstruth
Lin Wood
He might have a theory with teeth. It definitely seems like some get a hot shot and others not
The problem is that it’s not ‘attempted genocide’ if tens of thousands of people have already been killed.
If there are any white hots, WTF are they waiting for now?
If Lin Wood is posing this as a theory, it’s more than likely a fact.
👍 I’m surprised that your comment didn’t get more traction.
Nobody listens to me.
I do
Haven’t posted in a while, but have to tell you about my big accomplishment today. Sometimes you have to fight back locally in small ways. Today I got up the courage to exit a writers’ organization that I belonged to, that I had actually founded 7 years ago. They are a bunch of ultra-liberals, and I was fed up with all of the increasing political discussions at meetings, which has nothing to do with our group or mission. We’re talking Trump-bashing, vaccine fear-mongering, Biden worshiping and the works. I was their guest speaker today (via Zoom because there’s no way I’d present with a **&* mask on), and at the end of my well-received presentation on research, I told them I was done. They were fairly shocked, but I just had to do it. I really can’t be among this group any more; life is too short for that much stress.
Secretly, I told some conservative members (all 2 of them!) about my real reasons for leaving, and they feel the same way. Maybe someday liberals will figure out why people leave their groups, but I can’t be a part of this craziness any more. Perhaps this is just a drop in the bucket in the grand scheme of things today, but it was a big move for me, and very freeing. Thanks for listening!!
Thats a sad way to exit a big accomplishment, but the silver lining is that it was on your terms. You did it once, you may be called to do it again, but perhaps a different mission. Saving our country needs lots of people with lots of skill variety.
Too bad you could not lock the door on the way out. Understands good guys would not do that, alas.
Bravo! This is tough love for your fellow writers – but they need it.
I’m dealing with a similar bunch from my past. They are tolerable – even lovable, in venues which forbid political discussion, but let them loose on Twitter, and they’re rabid communists and useful idiots.
I’ m sending you a huge high five!
It’s hard to finally get to that place in your head that we can’t have relationships that are toxic. It’s one thing to have good natured disagreements, religion, politics, etc but ever since Trump bashing 24/7 on every single show, print, online and the meaner the better…the average Joe has gotten meaner too, imo.
They’re not worth it but I am sorry to hear it’s the end of something that meant a lot to you.
Thank you all for the comments and support. I still can point to the group I founded as an accomplishment and it will live on, but I also think it’s headed in the wrong direction and people may leave. Kind of bittersweet, but it was time for me to leave. I have another creative local “tribe” with my local radio station and that group stays out of politics, so I’ll be happier hanging out with them.
Gotta do what you gotta do. Sometimes, the things you create grow up…..wrong. It’s the nature of creation.
Here is a great “cut” of a long Yuri Bezmenov interview. It’s the classic on ideological subversion, with the usual beginning but a somewhat different ending.
I’m sure Baghdad Blitzer knows!
https://twitter.com/TrumpJew2/status/1444460915802484737
The reporters are natural born liars.
She knows exactly what the crowd was saying.
She didn’t have to comment on it at all.
But she knowingly, willfully misrepresented it as something completely other than it was.
That’s MSM.
Yes – EXACTLY. And she’ll get a BONUS for LYING.
h/t kyblue:
https://twitter.com/Zieleds/status/1444480440438378501?s=20
OMG – this is such a GRIFT.
Blood money
Isn’t that the truth?
All the anti-vaxxers used to be the dirty hippies with the unwashed hair and kids in filthy cloth diapers, burning incense and smoking weed. Now those same people are vaccinating those same kids with an untested poison, and bragging about it on social media. Meanwhile, all us straight-laced religious types with the clean hair and well-dressed kids are running screaming for the exits.
What a world.
Jewish Deplorable (@TrumpJew2) Tweeted:
The reporter’s desperate damage control might be my favorite part
https://twitter.com/TrumpJew2/status/1444459397590224896?s=20
So…how big is the big guy then!!!!!!?????
That’s a twenty-footer if it’s an inch. Whoo-boy, that’s scary.
Y’all need more than a screen door on yer house to keep that mutha out.
“Y’all ready for some fried chicken?”
And its in the guy’s backyard! Om Lird thats scary cause he isnt the only one out there.
Right ?!?
Not gonna lie, gators scare the crap outta me!
The bitty ones are cute but otherwise im good staying out of their space.
They got big heads. I don’t think he’s 20 feet.
I’d guess about 15 feet. Don’t worry, they hardly ever attack humans.
We were walking through a garden area in a plantation in South Carolina. They had some of those little twelve-inch garden fences of bent wire between the path and the flowers. Then we came to a little footbridge, and while we were on the footbridge a 10 foot gator went underneath. I don’t know if the flowers or the bent wire were supposed to stop him.
I just think about all those disnee hotels next to the swamp….ugh.
The ones wearing employee badges are ok.
Lol.
I can see it now: A new restaurant chain opening up in Florida (and most of the deep South): Gators and Taters 😀 🐊
(Maybe a spinoff from Suds and Spuds 🙂 )…..
Much more common than I thought!
https://www.jstor.org/stable/3892690
Cannibalism in the American Alligator on JSTOR
jstor.org
› stable › 3892690
Cannibalism in the American alligator (Alligator mississippiensis) was evaluated in Louisiana by examining 706 alligator stomachs for web tags previously attach…
Crocs do it, too!

https://nypost.com/2020/12/22/13-foot-cannibal-crocodile-eats-unlucky-smaller-beast/
Dinosaurs….no mercy.
Thats very interesting. Control their own population fairly regular.
Humans control each other’s contribution to population using a thing called “society” which makes us all responsible for progeny. An infinitude of CHEATS happen around that.
Crocodilians control each other’s contribution to population by EATING each other’s progeny!
Well, certain caballists must think they are reptiles.
LOL…I wondered if it was in South Carolina..the tweeter is a Gamecock fan so I’d say yeah.
My neighbors there said one day a big one was spraddled on top of a utility box. 3ft off the ground sunning himself !