Justice Must Be Done.
The prior election must be acknowledged as fraudulent, and steps must be taken to prosecute the fraudsters and restore integrity to the system.
Nothing else matters at this point. Talking about trying again in 2022 or 2024 is hopeless otherwise. Which is not to say one must never talk about this, but rather that one must account for this in ones planning; if fixing the fraud is not part of the plan, you have no plan.
Kamala Harris has a new nickname since she finally went west from DC to El Paso Texas: Westward Hoe.
Lawyer Appeasement Section
OK now for the fine print.
This is the WQTH Daily Thread. You know the drill. There’s no Poltical correctness, but civility is a requirement. There are Important Guidelines, here, with an addendum on 20191110.
We have a new board – called The U Tree – where people can take each other to the woodshed without fear of censorship or moderation.
And remember Wheatie’s Rules:
1. No food fights
2. No running with scissors.
3. If you bring snacks, bring enough for everyone.
4. Zeroth rule of gun safety: Don’t let the government get your guns.
5. Rule one of gun safety: The gun is always loaded.
5a. If you actually want the gun to be loaded, like because you’re checking out a bump in the night, then it’s empty.
6. Rule two of gun safety: Never point the gun at anything you’re not willing to destroy.
7. Rule three: Keep your finger off the trigger until ready to fire.
8. Rule the fourth: Be sure of your target and what is behind it.
(Hmm a few extras seem to have crept in.)
Spot Prices
All prices are Kitco Ask, 3PM MT Friday (at that time the markets close for the weekend).
Last week:
Gold $1763.10
Silver $25.90
Platinum $1040.00
Palladium $2550.00
Rhodium $20,000.00
This week, markets closed for the weekend at 3:00 PM Mountain Time
Gold $1782.30
Silver $26.20
Platinum $1114.00
Palladium $2724.00
Rhodium $19,200.00
The net result of the last week is mostly stability. Rhodium did dip below $19K but has blipped back up over it. (EDITED: these numbers were wrong until about 0330 Saturday, because I forgot to edit them.)
The Endgame for Classical Physics
Introduction
Physics before 1900 is known as “Classical Physics.” It still works as well as we used to think it did–provided you are working on every day scales. Go very small, very fast, or very massive and you discover that classical physics is an approximation very close to the truth for things that aren’t very small, very fast, or very massive, so very close that one can practically ignore the difference.
What happened around 1900? We started investigating things outside that zone.
Our daily lives exist in that zone though some of our tech goes into places where non-classical physics must be accounted for. In fact no semiconductor would work if modern physics weren’t true, and you wouldn’t be reading these words because no dead-tree leftist gatekeeper publisher would have me.
Yes. This is the day. This is the day we don’t stop at 1895.
But first…
Go Back: Avogadro’s Number
I made the claim that no one had any idea as to the size of Avogadro’s number before 1895. As a reminder, this is the number of molecules of some compound with an “atomic weight” of X, in an X gram sample of it, or almost equivalently, the number of atomic mass units in a gram. Taking oxygen as an example, it forms a two-atom molecule, O2 whose atomic mass by definition was 32 atomic mass units. How many O2 molecules in 32 grams? The answer to that, whatever it is, is Avogadro’s number.
It turns out that in 1865 Josef Loschmidt was able to make an argument about at least the approximate size of atoms and how far apart they had to be in comparison for a gas to behave as a gas and was somehow able to figure out how many molecules of a gas were in one liter of it, at standard temperature and pressure.
But since we already know that 22.4 liters of gas under the same conditions is one mole, so simply multiplying Loschmidt’s number by 22.4 gives you Avogadro’s number, the number of molecules of anything in a mole of that thing.
The modern value of Loschmidt’s number is 2.6867811(15)×1025 per cubic meter, but there are a thousand liters in a cubic meter, so 2.687×1022 works well enough for our purposes.
Apparently Loschmidt himself didn’t go all the way through the reasoning to get the number, it was left to people like James Clerk Maxwell (whose name had better sound familiar by now) to cite a figure of “about 19 million million million” per cm3, or 1.9×1025 m-3. Which is a bit over 29% too low, but really, given that we had no way of directly measuring it before then, was pretty good.
So contrary to what I said, we did have some idea what Avogadro’s number was.
Today, the mole is one of the seven fundamental units of the SI (metric system) and is defined to be precisely 6.02214076×1023 particles (i.e., roughly 22.4 times Loschmidt’s number. If it should turn out that that many atoms of (say) atomic mass 12 doesn’t quite mass 12 grams, tough. (We know it’s pretty doggone close though, so high school chemistry lab can ignore the difference. Besides the proportions will be right even if NA isn’t quite where it “should” be.)
OK, that’s out of the way.
The Crookes Tube
Today there are actually four…well, three and a half…stories to be told, and the Crookes tube is tied up in two of them.
What is a Crookes tube? Well, it’s sort of like the “cathode ray tube” no one uses for televisions and computer monitors any more. Or any of a host of other vacuum tubes.
It was first created sometime before 1869, and it was a geek toy par excellence. Take a large, oblong glass tube, run two conductors into it, one near each end. Seal it off and pump almost all of the air out. I’d guess they pumped as much air as they could out of it, but couldn’t get the last millionth out of it. As it turns out it won’t work if it’s a total vacuum.
Now put a ten thousand volt potential between the two conductors. The one hooked up to positive is called the anode, the negative one is the cathode.
You get an eerie green glow on the end of the tube that’s behind the anode.
Now that was interesting, what was going on here? There wasn’t any current flowing. So physicists started tinkering. By putting a cutout of a Maltese cross near the anode (done by Julius Pluecker in 1869) and noting that the green glowing part of the glass had Maltese cross-shaped “shadow” in it, it was proved that whatever it was was traveling in a straight line away from the anode and past the cathode, and some of the “rays” were being blocked by the Maltese cross. (And nowadays any time someone draws a diagram of a Crookes tube in action, they always show it with a Maltese cross; and even the one demoed in the picture below uses it; it’s some sort of cliche now.)

The straight line travel and the fact that glows like this (“fluorescence”) were thus far known to only be caused by ultraviolet light, led some to conclude that these rays were electromagnetic in nature. Others thought that the rays might just be charged atoms.
In 1876 Eugen Goldstein experimented with different shapes of cathode, (a point, a flat surface he could tilt, and so on) and was able to prove that the rays behaved like charged particles. Some sort of electromagnetism would leave every point on the surface going in every direction (which is why you see the whole light bulb not just the point that faces you full on), but if the cathode were a flat surface the rays would all come out perpendicular to the surface, as if they were particles repelled by that surface. Of course, it’s a negatively charged surface, so the particles repelled by it would be negatively charged.
Goldstein gave them the name “cathode rays.”
Heinrich Hertz, who, you’ll remember, discovered radio and though it was useless, decided to try another experiment to see whether the “rays” were particles, or some sort of electromagnetism. He put two other plates inside the tube on either side of the beam and put an electrical potential across the plates. The electrical field between the plates should bend the beam, if it were particles. He didn’t see it happen, but it turned out his apparatus just wasn’t good enough. Later Arthur Shuster repeated the experiment with a better vacuum in the tube and did see the bending. So the “particle” side of the argument was looking better and better.
Crookes himself put magnets on either side of the tube and got the beam to deflect in accordance with:
F = v ⨉ B
(You know, the right hand rule and all that.)
That just made the “particle” theory even better.
So there that stood at the end of 1894. But this time we aren’t going to stop there!
Recap
We had, up to this time, identified at least the following conservation laws that applied to any closed system (one where nothing could get in or out).
- Conservation of mass
- Conservation of momentum
- Conservation of energy
- Conservation of electric charge
- Conservation of angular momentum
The following mysteries were unanswered at the end of 1894.
- Why was the long axis of Mercury’s orbit precessing more than expected, by 43 arcseconds every century? Was it, indeed, a planet even closer to the sun? If so, it’d have been nice to actually see it.
- Why was Michelson unable to measure any difference in speed of light despite the fact we, being on planet Earth that is orbiting the sun, had to be moving through the medium in which it propagates?
- What makes the sun (and other stars) shine (beyond the obvious “they shine because they’re hot” answer). What keeps the sun hot, what energy is it harnessing?
- How did the solar system form? Any answer to this must account for how the planets, only a tiny fraction of the mass of the solar system, ended up with the vast majority of the angular momentum in the system.
- What is the electrical “fluid” that moves around when there is an electric current, and that somehow seems imbalanced when we perceive that an object has a charge? Were there both negative and positive fluids, or just one fluid that had a natural neutral level; below it was negative (deficit), above it was positive (excess)?
- Why are there so many different kinds of atoms? How did electrical charges relate to chemistry? How is it that 94 thousand coulombs of charge are needed to bust apart certain molecules (though it often had to be delivered at different voltages depending on the molecule)?
- Why were the atomic weights almost always a multiple of hydrogen’s? Why was it never quite a perfect multiple? Why was it sometimes nowhere near to being a multiple?
- (and the 8 disappears?) Why does the photoelectric effect work the way it does, where it depends on the frequency of the light hitting the object, not the intensity?
- Why does black body radiation have a “hump” in its frequency graph?
And remember we left the story of Ramsay hanging at the end of 1894, as he decided to look for other elements in the new “Group 0” of the periodic table, now represented by argon, whose mass was between that of chlorine and potassium.
And with that recap out of the way…
WE
ARE
READY
FOR
1895
1895
In November of this year, Wilhelm Röntgen was investigating all sorts of different “tubes,” the Crookes tube among them. He noticed that he could fashion an aluminum sheet with a rectangular window in it and block the cathode rays, but if he placed a board painted with barium platinocyanide ([Pt(CN)4]2−) near the window, the chemical would glow. Something was getting out of the tube and causing fluorescence.
On November 8th (a Friday) he decided to investigate further. He covered a Crookes tube with cardboard to block the light it was emitting (so he thought!), fashioned a similar window, ensured that the cardboard cover was completely covered by darkening his lab and looking for leaks. There were none, but he noticed an odd shimmering out of the corner of his eye. Striking a match, he realized it was his barium platinocyanide.
Over the next couple of days, he discovered that if the tube was firing while a piece of lead were in front of the barium platinocyanide, the lead cast a shadow (even though the cardboard around the tube clearly didn’t block the radiation, the lead did.) He also noticed quite by accident he could see the bones in his own hand by interjecting it between the tube and the barium platinocyanide. That was spooky, spooky enough to get Roentgen to conduct his experiments in secrecy until he was sure of what he had.
Since he didn’t know what these things were he called them X-rays; X often stood for “unknown.” Now that we know what they are, we still call them X-rays, though some also call them Roentgen waves.
The first ever X ray photograph was of his wife’s hand; when she saw her own skeleton she exclaimed that she had seen her death.
As it turned out X rays are also electromagnetic waves, they are of an even shorter wavelength/higher frequency than ultraviolet rays. “Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30 petahertz to 30 exahertz (30×1015 Hz to 30×1018 Hz)” (Wikipedia. A picometer is 1/1000th of a nanometer or one trillionth of a meter, a petahertz is a quadrillion cycles per second. Remember that visible light runs from 400,000 to 700,000 picometers.)
The Crookes tube was no longer a nerd toy. It was now a piece of medical equipment, and it was in practical use within months, probably the fastest a basic physics discovery has ever been exploited. On January 11th, barely three months after Roentgen started investigating the Crookes tube, it was used by someone else (John Hall Edwards of Birmingham, England) to find a needle embedded in a patient’s hand.
This wasn’t some obscure thing; it made the newspapers and this was before they were all fake news.
X rays were going to become a very useful research tool outside of medicine.
Roentgen received the very first Nobel Prize for Physics in 1901, because of this discovery that had turned out to be so very useful right away.
And let us pick up our story of Group 0. Ramsay was preparing to look for more of these gases in the air, but during this year two Swedish chemists, Per Teodor Cleve and Nils Abraham Langlet, discovered a gas emanating from cleveite, an ore of uranium. On further investigation, this gas was totally non-reactive, and had an atomic weight of about 4. Well, that is perfectly midway between hydrogen (1) and lithium (7), and logically the top of that “Group 0” should be an element in between these two, so, very cool, the top of the column was in place. There was a gap between it and argon, and nothing below argon in the column was filled in yet. Ramsay would find neon, krypton and xenon in very, very tiny percentages in our atmosphere, and there’s even a bit of this top gas.
But we didn’t have to figure out a name for this gas. Because when we put some of it in a tube and got it to glow, and took the spectrograph…it turned out to be helium, the mystery element known from the solar spectrum!!! It wasn’t a metal after all, but a gas; logically it should be named “helion” (to match the -on ending of the other gases in that column), but…too late!
1896
Our main character here is Henri Becquerel (1852-1908), who was fascinated by phosphorescence. This was the way an object could absorb one wavelength of light for some period of time, then glow in a different wavelength for a time afterwards, as if the light “pumps up” the chemical which then gets rid of the energy later. He heard about X rays (who could hear anything else in the din) early in 1896 and thought, perhaps some chemicals might phosphoresce in X-rays after being pumped up in ultraviolet light, like from the sun.
Well this was easy to test. Wrap photographic plates up in paper, so that ordinary light and even UV cannot get in. Pump up a candidate mineral, then put it next to the photographic plate. If it is emitting X rays, those should fog the plate even through the paper.
Becquerel’s candidate material was uranium salts, which phosphoresced very nicely in visible light. He just needed a bright day to perform the experiment.
At which point, in Paris, it got cloudy. No bright sunlight to run the experiment in! He put the plates and the uranium salts in a drawer, and waited for good weather, smoke no doubt pouring from his ears.
After a days of this crap, he decided, “Aw, what the hell!” and developed the photographic plate.
This has to be the most consequential “Aw, what the hell!” moment in history.
The plate had blackened right next to his sample. It even bore the outline of a Maltese cross (OK, someone come up with a new idea), which was there to prove the fogging came from the sample, not some other cause–though the cross didn’t block the rays completely.
Something had blasted through the paper and reacted with the chemicals on the photographic plate. Something that didn’t need to be pumped up–or something that stayed pumped up.
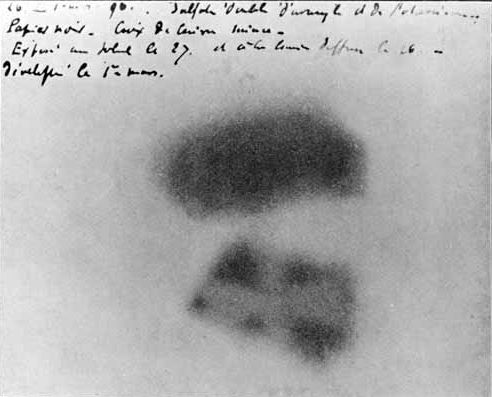
This had actually been noticed back in 1857, by a friend of Becquerel’s family…who didn’t pursue it very far.
Becquerel did. And the world was never the same again.
Further experiments established it didn’t have to be a phosphorescent compound of uranium; any compound of uranium would do this. Pure uranium would do this. This had to do with uranium, and uranium would do this no matter what. By May of 1896 Becquerel had realized this, and published his results, and now the world knew of “Becquerel rays” which we now call “radioactivity.”
His own words from the 2nd of March
I will insist particularly upon the following fact, which seems to me quite important and beyond the phenomena which one could expect to observe: The same crystalline crusts [of potassium uranyl sulfate], arranged the same way with respect to the photographic plates, in the same conditions and through the same screens, but sheltered from the excitation of incident rays and kept in darkness, still produce the same photographic images. Here is how I was led to make this observation: among the preceding experiments, some had been prepared on Wednesday the 26th and Thursday the 27th of February, and since the sun was out only intermittently on these days, I kept the apparatuses prepared and returned the cases to the darkness of a bureau drawer, leaving in place the crusts of the uranium salt. Since the sun did not come out in the following days, I developed the photographic plates on the 1st of March, expecting to find the images very weak. Instead the silhouettes appeared with great intensity … One hypothesis which presents itself to the mind naturally enough would be to suppose that these rays, whose effects have a great similarity to the effects produced by the rays studied by M. Lenard and M. Röntgen, are invisible rays emitted by phosphorescence and persisting infinitely longer than the duration of the luminous rays emitted by these bodies. However, the present experiments, without being contrary to this hypothesis, do not warrant this conclusion. I hope that the experiments which I am pursuing at the moment will be able to bring some clarification to this new class of phenomena.
Henri Becquerel
Marie Sklodowska Curie (who is definitely not the subject of any “Polish joke”) was looking for a doctoral thesis topic and decided to investigate. Her husband Pierre Curie had invented an electrometer, a very sensitive device for studying electrical charge. She discovered that the air around a uranium sample was able to carry a current–the radiation was somehow making the air charged. Careful measurements revealed that the amount of radiation was directly proportional to the uranium in the compound. But this was a compound created in the lab.
And by 1898 she had noticed that thorium, too, was radioactive, though in this case she was scooped two months before by Gerhard Carl Schmidt.
Going to an ore that had had uranium in it for a long time, however, turned out different. The ore was four times as radioactive as the uranium that was in it. There was something else in the ore, something unknown, that was radioactive.
Her husband, Pierre, became so intrigued he dropped his own work on crystals to pursue this. They started with a carefully-weighted 100 grams of pitchblende.
They ended up going through tons of pitchblende. There was so little of what they were searching for that they needed to process that much pitchblende to find enough to actually experiment with.
In July 1898, they announced the existence of the element polonium. On December 26th, they announced radium. These were both elements of atomic weight higher than lead and bismuth, but below uranium (and there had been a big empty gap there in the weight sequence).
It took Marie Curie until 1910 to get a pure radium sample, and she never did manage to drag enough polonium together to constitute a “sample.”
Becquerel and the two Curies got the 1903 Nobel Prize for Physics for their work on these phenomena.
Pierre was killed in a traffic accident in 1906; Marie ultimately received the 1911 Nobel Prize for Chemistry. she was the first to get a second Nobel, and is still one of just two people who have received two different categories of Nobel prize.
And this was just the beginning of what came of Becquerel’s What the Hell moment. We owe the nuclear bomb and the nuclear power plant and our entire knowledge of nuclear physics (and physics even smaller than that) to this moment.
What would have happened had he not done this? How long would it have taken?
Not all that long. Another scientist, it turned out was mere weeks behind him; if Becquerel had tarried publishing, he’d have been scooped. And if that hadn’t been the case, well someone would have noticed eventually, and pursued it.
This was rather surprising to me. It seemed as if this discovery were a freak accident that could have waited another couple of centuries to happen. It’s often used as the perfect example of serendipity; something turning up unexpectedly, but in front of a man with the imagination and intelligence to pursue it. (Read, sometime, the story of the discovery of teflon. Or penicillin.) Instead of that, it’s actually a discovery whose time had come.
1897
The man of this chapter was J. J. Thomson (1856-1840), and he too liked to play with Crookes tubes. He worked at the Cavendish laboratory (we’ve heard that before, haven’t we?).
He did some very careful, exacting work with both magnetic and electric deflection of the cathode rays. He used an electromagnet so he could vary the strength of the magnetic field (rather than simply bringing in a big permanent magnet). He then varied the electric field and the magnetic field until he got both deflections to be the same angle. His goal was to determine the charge-to-mass ratio of whatever particles were in the cathode rays.
The formula for the electric deflection is:
Θ = E e ℓ / mv2
While the magnetic deflection is:
Θ = B e ℓ / mv
Where Θ was the angle of the deflection, E the strength of the electric field, B the strength of the magnetic field, ℓ the length of the plates applying the deflection, m the mass of the particle, and v its velocity, and e the electric charge of the particle.
If the two angles are equal then we can write:
E e ℓ / mv2 = B e ℓ / mv
A lot of simplification and substitutions can reduce this down to:
m/e = B2ℓ / E Θ
The magnetic and electric field strengths were known, as was the length of the plates, and the angle of the deflection, so we now had the mass to charge (or charge to mass) ratio of these cathode rays.
It’s generally quoted as charge to mass, and its modern best value is −1.75882001076(53)×1011 C/kg.
Which means that a single gram of cathode ray “stuff” has a charge of −1.75882001076(53)×108 coulombs.
So that was pretty definitive, cathode rays are made of negatively charged particles.
Now if we step back to Faraday, who figured out that 96,485.3 colombs would break up one mole of electrical bonds, or as by now was realized, was the charge of one mole (one gram) of hydrogen, when the hydrogen had a positive charge, i.e., was a positive hydrogen ion.
The same charge on a cathode ray stuff would be carried by 0.000548 grams of cathode ray stuff.
The conclusion was that the cathode rays consisted of particles that weighed 0.05 percent as much as a hydrogen atom, or (taking the reciprocal) that a hydrogen ion had about 1823 times the mass of a cathode ray particle. (The actual value is closer to 1836, but that’s because a mole of hydrogen does not weigh exactly one gram.) Thomson actually got a value closer to a thousand, but it was still a striking figure.
Now up to this point, atoms were regarded as simple, indivisible things, that could get an electric charge to be sure, but were nevertheless indivisible. Now there had been scientists who had speculated that atoms might be made up of smaller things (William Prout and Norman Lockyer, for instance) but in the absence of any real evidence that’s all it was, speculation. Prout and Lockyer had figured the smaller building block was probably about the mass of a hydrogen atom, so basically hydrogen contained one of these items, helium four, lithium seven, and so on. (Of course they couldn’t explain why the ratios weren’t perfect clean integers and couldn’t explain the elements with large fractions (like a half) in their atomic weights.)
It appeared that the Crookes tube operated by knocking a very, very small chip off of an atom, a chip bearing a huge negative charge. The remainder of the atom had to bear a positive charge that matched. Certainly nothing as substantial as an entire hydrogen atom!
(However, Prout and Lockyer weren’t completely wrong, as we will eventually see.)
JJ Thomson in his own words (extracted from Wikipoo):
As the cathode rays carry a charge of negative electricity, are deflected by an electrostatic force as if they were negatively electrified, and are acted on by a magnetic force in just the way in which this force would act on a negatively electrified body moving along the path of these rays, I can see no escape from the conclusion that they are charges of negative electricity carried by particles of matter.
J J Thomsen
As to the source of these particles, Thomson believed they emerged from the molecules of gas in the vicinity of the cathode.
If, in the very intense electric field in the neighbourhood of the cathode, the molecules of the gas are dissociated and are split up, not into the ordinary chemical atoms, but into these primordial atoms, which we shall for brevity call corpuscles; and if these corpuscles are charged with electricity and projected from the cathode by the electric field, they would behave exactly like the cathode rays.
J J Thomson
The Electron
The new particle came to be known as the electron (not a “corpuscle”) and it soon became clear that this was our “electrical fluid.” You may very well use this word or its derivatives every day (it’s part of electronic, and that’s not a coincidental pun, the word electronic was derived from the word electron), but 125 years ago the word did not exist.
Any positively charged particle that could be ginned up in a tube (and there were indeed some even in Crookes tubes, these were known as canal rays) were basically atoms or even whole molecules with a positive charge. The measurement precision was too poor then to tell for sure, but these positively charged particles were simply a little light, because they were minus an electron. Or two, or perhaps even three.
Thomson concluded that an atom consisted of some number of electrons, embedded in a larger mass that had a positive charge; it’s called the “plum pudding” model to this day (actually, raisins but you know…the Brits).
Now we’ve wiped out one of our mysteries, number 5 above, and we now had an inkling of what was going on between atoms. A sodium atom, for example, could give up one electron and become a positively charged ion, a chlorine atom would scoop it up, and become a negatively charged ion. If enough of these transactions happened, you could crystallize all of these ions into a cube of table salt, or you could dissolve it and there’d be slightly light, positively charge sodium atoms and slightly heavy, negatively charged chlorine atoms floating around in the water. Since the total charge still came out to zero, drinking salt water wouldn’t electrocute you (but could kill you in other ways).
Generally, in an electrical circuit, it’s the negative electrons that move around, not the positive ions. So Franklin was right, there was one fluid. But he guessed wrong, too, in assigning the “negative” label to what turned out to be the fluid. But then Du Fay wasn’t completely wrong either; because there was clearly a positively charged thing out there, the bulk of the atom.
If you remember when I was talking about electricity, with the movement of the fluid, I implicitly assumed the fluid was positively charged, and moved from the positive terminal of the battery (or other power source) through the circuit to the negative terminal. So basically, I should go back and trash all those diagrams, right?
Nope. It turns out it’s mathematically equivalent to the real situation, with a negatively charged bunch of electrons flowing from the negative terminal to the positive terminal. Electrical engineers still simply pretend a positively charged current flows from positive to negative, because the math is exactly the same as running a negatively charged current from negative to positive.(Ask me how I know this.) It makes no actual difference. There are situations where it really does matter that the stuff that is moving is negatively charged, but that’s getting into semiconductors, and you’re moving from the realm of electrical circuits to electron-ic circuits.
Thomson showed in 1906 that the uncharged hydrogen atom contained exactly one electron. No more, no less. There was no way to knock two electrons out of it, as some had thought.
In 1905 he showed that natural potassium was radioactive.
He won the Nobel Prize for Physics in 1906.
Alpha, Beta Gamma
OK, let’s return to radioactivity.
In 1899, right after the Curies found polonium and radium (unlike Vladimir Putin, Marie Curie killed herself with the stuff, eventually dying of cancer in the 1930s), Ernest Rutherford, a Kiwi working at McGill university in Canada, and Paul Villard in Paris, were able to determine that radioactivity, the actual stuff coming out of polonium, radium, thorium and uranium, consisted of three different types of thing. They did this by applying a magnetic field and seeing what got deflected, and also by noting how strongly penetrating the things were.
Alpha particles were massive, had a double positive charge, and would be stopped by a few centimeters of air or your skin or a sheet of paper. Rutherford, in fact, was able to measure the charge to mass ratio and by 1907 proved that an alpha particle was essentially a helium atom, with two electrons removed from it. So it had twice the charge of a positive hydrogen ion, but four times the mass (the charge ratio originally measured would be half that of a hydrogen ion). Their typical speed turned out to be four percent of the velocity of light (which is still 12,000,000 meters per second).
Beta particles were a hundred times as penetrating. Becquerel in 1900 had a comeback, he was able to measure the charge to mass ratio of beta particles…and they turned out to be electrons.
Gamma rays were found by Villard in 1900; Rutherford worked with them and fit them into his naming scheme in 1903. These turned out to be electromagnetic radiation, of even higher frequency (and lower wavelength) than x rays. They were very penetrating, indeed. Today, we know of some forms of radioactivity that produce relatively long-wave gamma rays, longer than some X rays, so the dividing line between X-ray and gamma ray is fuzzy. For EM radiation in this overlapping range of wavelengths, it’s considered a gamma ray if it came from radioactivity, an X-ray otherwise.
It was fairly easy to measure how much energy these particles had in them. It turns out that if you can wait for a kilogram of uranium to decay (which takes billions of years) you get 1.72 trillion joules of energy out of it. And that ignores the fact that what the uranium decays into decays again, and again, and again, adds more energy to a grand total of a whopping 22.8 terajoules.
Compare this to a kilogram of coal: rougly 24 megajoules. The uranium, just in decaying, contains almost exactly a million times as much energy as the coal, though it has to release it very slowly.
A lot of energy, released over a very long time….what does that remind you of?
Stars, I hope. Could this be part of the answer to what powers the stars? At first look, it doesn’t seem promising, and that’s mainly because we now had ways of knowing what was in stars, and there was very, very little uranium in them. But now we had a suggestion as to what the answer might look like, as opposed to just a shoulder shrug.
For now, let’s chew on this alpha particle a bit. It turns out to be a double-ionized helium atom. And it comes out of a block of pure uranium. Somehow, pure uranium seems to consist, at least in part, of He2+ ions.
Maybe Prout and Lockyer were onto something after all, if one atom could be built up, somehow, out of other atoms without it being a molecule, and only breaking apart through radioactivity. Of course the basic piece seemed to be a helium atom, not a hydrogen atom, but who was to say the helium atom couldn’t actually be a group of four hydrogen building blocks?
But regardless of the details, this explains a couple of things.
Remember those Swedes who found helium coming out of uranium ore? That’s why. The uranium was spitting out alpha particles, which were combining with electrons in the rock or the air, and becoming perfectly normal helium gas.
And when, in 1903, it turned out that natural gas wells in Kansas, Oklahoma, and Texas were producing gas that had a lot of helium in it…that made sense. That helium ended up deep underground because it was created there…by uranium and thorium radioactivity.
Every single helium atom on Earth today was once an alpha particle crashing out of a nucleus at 4 percent of the speed of light. You’re filling your balloons with chilled radioactivity.
And that part of the US is still the major world source of helium gas.
(Everyone say “‘Murica!!!” in a high, squeaky voice!)
Conclusion
What a wild ride in only five years!!!
We went from having no real clue that atoms had pieces, to having seen those pieces; we were starting to understand the structure of atoms. We now had some notion of what was going on in chemistry.
And the wild ride was just getting started.
And Joe Biden didn’t win.
A Personal Announcement
I’m probably going to be nearly buried by other obligations, and soon. At some point, I’m going to have to put the physics posts on “hold” and just do a complete skeleton of a post each week (I’d still include the bullion prices)…and this will last for a couple of months. If that’s not satisfactory, I suppose we will need another author to fill in.
Obligatory PSAs and Reminders
China is Lower than Whale Shit
Remember Hong Kong!!!
中国是个混蛋 !!!
Zhōngguò shì gè hùndàn !!!
China is asshoe !!!
China is in the White House
Since Wednesday, January 20 at Noon EST, the bought-and-paid for His Fraudulency Joseph Biden has been in the White House. It’s as good as having China in the Oval Office.
Joe Biden is Asshoe
China is in the White House, because Joe Biden is in the White House, and Joe Biden is identically equal to China. China is Asshoe. Therefore, Joe Biden is Asshoe.
But of course the much more important thing to realize:
Joe Biden Didn’t Win
乔*拜登没赢 !!!
Qiáo Bài dēng méi yíng !!!
Joe Biden didn’t win !!!

“Westward Hoe”
LMAO!!!
She grew up a Westward Hoe in Oakland and Sacramento, but hopes to hit the Big Time as an Eastward Hoe in DC.
https://www.thegatewaypundit.com/2021/06/two-kamala-harris-staffers-charge-planning-travel-resign-amid-disastrous-border-trip/
Something is very weird about this.
Some RUG got pulled out from under her.
If this was ONLY for optics, there is no reason for it to fail.
The White House is FILLED with professional people who do all this stuff correctly. There are Democrats all over Texas who could make this whole thing a no-brainer.
There is no legitimate reason this should have been a disaster.
There is MORE to this story.
Someone in the OutHouse wants to sabotage Westward Hoe.
I did notice that her travel office was going to have to start scheduling her for travel in support of pushing vaccination. QueMala may have ordered all her own people to get vaccinated – and those two WOMEN may have decided to resign.
And YES THEY LOOK LIKE THEY ARE OF CHILD-BEARING AGE…..

A totally different rationale, but they could be combined.
I mean as long as we’re blatantly speculating about this or phlogiston or Prout’s suggestion about atomic structure.
They may be the ones who sabotaged her, in retribution.
Que Mala Harris – born to be a screenplay generator!
Jill Bye, Done?????
Incipient Hell-the-BEAST????
Various catfights brewing there (if only they’d BOTH lose)….
That would be my guess two, Jill wants to stave off the inevitable, but the simpler guess would be Kamala herself. She’s out of her league lacking the mental capacity to do the job and myopic as a result. In such a condition she’ll strike out against things that don’t make her feel comfortable, being uncapable to nurture the responses she wants, while staff is likely moving a few clicks faster and expecting a better performance level from her. In short she’s paranoid.
That’s what I think. The problem is that she’s unlikable and way out of her league, in addition to pushing policies that make no sense, that are destructive, and that sane people don’t want. No amount of staff preparation can mitigate that.
More shenanigans from the other end of the Mall…Nanzi’s visions of grandeur. MeThinks.
My guess Que Mala was pissed that she had to pass the Trump supporters, on the way to the photo-op. at the border, calling her out on her fraudulent election.
One comment after 25 minutes?!?!?
Are you being boycotted, Steve?
Nah…slim pickins on rhe night crew.
Or is it Slim Pickens?
Id say his speevh works for our current sit yee ation too.
Good to see you!
I went back to read the post and create a sidebar link, so *I* have an excuse! 😉
We worked hard yesterday! Some of us are still stuck on Leo 600 IYKWIM. 😉😉
🥰 🥰 🤗 PGROUP 🤗 🥰 🥰
Ni Ni All 🥱😴😴
OK, how do you know that “the math is exactly the same as running a negatively charged current from negative to positive.”?
The net positive flow is in the same direction either way.
Oh. I know that EEs continue to treat it as a positive current…because I spent four painful years as a EE major.
Painful years, I’ve had a few…..
Back a ways, I was known for my mastery of the “Trust Accumulation Distribution Throwback” — which, IIRC, was calculated on Federal form 1041J. At least EE is minimally affected by the whims of Congress.
Or, to take something fairly simple, you get an itemized deduction for estate taxes previously paid on Income in Respect of a Decedent (IRD). If you are an heir with this (like inheriting a parent’s IRA), you need to have a tax preparer (even if it’s you) who understands this — but if you go around to 20 tax preparers and ask them the significance of Income in Respect of a Decedent, 19 will go stone-cold blank.
The math works out much the same as multiplying two negative numbers result in a positive product. One negative is the polarity of the charge, and the other is the backwards movement.
As SteveInCO mentions, the EE point-of-view is that electrical current is treated as positive charged particles moving in the forward direction. This is a convention different from what physicists might prefer, much like EEs like to use j instead of i for imaginary numbers since we use I and i for currents pretty much everywhere. It also avoids having to worry about additional minus signs everywhere, in situations where energy or power is being considered: Positive resistance implies that energy is being transferred out from the circuit, usually as heat, though it might be as radiation from an antenna or other transducer.
Positive charges moving forward or negative charges moving backwards end up as equivalent. This is most apparent in things like batteries where there are positively charged ions moving in the forward direction as well as negative charged ions moving in the reverse direction, being pushed or pulled by the applied or generated electric field. Most other places, there are only negatively charged electrons moving in the direction opposite, but outside of the wire, transistor, or vacuum tube, the internal details makes no difference. Again, there are electric fields present that cause the particles to move, from applied voltages on the terminals.
Even in P-type semiconductors, the «holes», places where an electron is missing, is considered a postive charged particle that moves around as if it wasn’t just a place where electrons pop out of and fall into.
But the holes tend to move more slowly (if I recall–my strong suit was EM fields) largely because they aren’t a real particle. So in this particular case it does make a minor difference what’s really happening.
Yes, the actual nature of the moving charges can be important, and holes move slower than electrons, and the mobility of all of these also varies with the actual semiconductor material. For germanium and silicon the electrons are about 2 to 3 times as mobile as the holes, while for galllium-arsenide the electrons are about 100 times more mobile.
This does affect the possible operating frequency of the resulting transistors. NPN transistors and N-channel MOSFETs (Metal-Oxide Semiconductor Field Effect Transistor) devices in which the moving charges (the carriers) are mostly electrons, tend to be favored over the PNP transistors and P-channel MOSFETs where the majority carriers are the holes. As the ratio isn’t prohibitively large with silicon, devices in both polarities has found plenty of use in all kinds of CMOS (complementary MOS) integrated circuits.
But no-one bothers to make P-channel GaAs devices since the low mobility of holes makes them much worse than similar silicon devices.
So it isn’t irrelevant when looking at materials science and chemistry aspects when designing a transistor, as well as selecting a transistor for some application (or determining a useful replacement if the original device is no longer available for a repair job), but once going outside the package, by now the transistor is what it is, and the interesting parameters are the voltages and currents of its terminals.
Back in the day, we had two types of available photodetectors in our laser receivers — one was GaAsFET APDs and the other I forget [when looking up the spelling of Fabry-Perot, they’re PIN diodes]. While the GaAsFET APDs were more sensitive, they wouldn’t go “ping ping ping” as you modulated the laser — they’d go “pfft, pfft, pfft”, so we tried to minimize their use. We were doing video in 40 MHz bands, 16 channels per fiber, so the effect was hardly noticeable as we reconstructed the original signal, but the more you maintained a clean signal (in an analog environment), the cleaner the end-to-end performance (that the customer cared about) would be.
Our “cheapie, short-link” lasers were selected Fabry-Perot semiconductor lasers from Philips. We’d get batches of 20 from the factory, use 3-12, and send the rest back for some other schmo to use. As business took off, the local rep thought it would be cute to slow foot the return process…..we had almost a million dollars of lasers that wouldn’t do what we needed them to do that were taking up space in-house when I got there.
One of the joys of Fabry-Perot lasers is that you gain a thorough understanding of chromatic dispersion. The “extended, longer-link” lasers were DFBs.
Again — this is not prioritized in accounting classrooms.
Speaking of things not in classrooms…..
The local rep had been cruising on the fact that we were mostly busy doing other things and were generally too nice. To solve this problem, I brought in the Fiancee’s sister as a temp and told her to get rid of this stuff.
The Fiancee tries to look like a laid-back Californian, though she grew up in NYC burbs. Her sister (who also lives locally) lets her inner NYC shine.
We’d tucked the sister into a spare office that I walked past regularly. About a week into the process, I walked past and overheard her side of a phone call, “look, just give me the address, and we’ll deal with the paperwork while it’s in transit. WHAT DO YOU MEAN YOU DON’T KNOW THE ADDRESS? You don’t KNOW where your OWN fucking factory is????”
She looked up, and saw me, and I continued walking to my office, ’cause I didn’t want to interfere with her rhythm.
She came to my office when she was done with the call. I hadn’t quite finished laughing yet. She was prepared to be apologetic, and I kept saying, “no, no, that was perfect.”
All told, it took about six weeks to clear that worry off my plate, and I can stand proud and say it was a very successful and efficient expenditure of corporate funds, however much it may reek of nepotism. I needed a New Yorker, and had one handy immediately, without recruiting.
And, BTW, that rep was completely straightened-out for all the rest of the time I was there. Amazing how that works….
Having moved south from NY, I can personally attest that the rhythm of business interactions by New Yorkers is unique and highly effective.
Let’s just say it is one of the things I miss most about NY. 😀
I should point out that working through semiconductor diagrams is MUCH EASIER when you figure out that TTL (“transistor-transistor logic”) means you are talking about a nominal “0” and a nominal “+5” — and that 0 is tied to circuit ground.
For some reason, they don’t teach this in accounting classes.
Another interest of Pierre Curie was in finding the “Curie Point” at which a material that is ordinarily attracted to magnets becomes nonmagnetic after heating.
Evidently, everyone and their aunt Sally used to know all about this, and there are a whole bunch of recipes among engine builders with steps like “use a torch to heat your workpiece until a magnet won’t stick to it, then drop it in a brick-lined bucket of waste oil.” (That gets you a lovely hardened black surface, BTW).
You have to drop it, because otherwise the oil at the surface will burn as it enters and (among other things) emit large amounts of smoke; and you have to line your (metal) bucket with bricks on the inside because the part may melt its way through the bucket before the boiling oil sufficiently cools it if it touches the bucket. It doesn’t have to be lined enough to be sealed, just lined enough to keep the heated metal from touching the bucket, even if you do a fumble-fingered drop.
I was thinking about automobile engines there for a while…phrasing like (and I paraphrase) “drop” it into a bucket and “be sure not to burn your fingers” argues otherwise.
Well, you wouldn’t do that for any large pieces of engine because you don’t generally want them hardened. Typically you harden smaller bits — things like camshafts and pivot pins
PGroup2, not certain if you are still on here tonight if you will see this comment, but thatnk you for your update and again, you are in my prayers and I think you have a great attitude.
My secret friend! Nice to “see” you.
You do whats necessary. These posts are a lot of work! Thx Steve.

https://twitter.com/RN_Destiny/status/1407903704179789824
McAfee for President!
I hope he beat it. If anyone could, he’s a prime candidate to have done so.
That Colony Hotel is in South Beach, Miami. Anyone hear anything about him owning a unit in the collapsed building?
Yes. We need the people to use their cajones and speak up. Stop being afraid to say voldemort!
BTW, Monkey Werx found flights into and out of Spain of the same sort as when Epstein disappeared. He seems to think McAfee was extracted in a black hat op. I doubt that. But, at this point, all of it is just speculation. I doubt those flights would have been visible on Monkey’s software.
Joe Biden didn’t win!
Raven Constantine
@Artraven
2m
·
Difis hubby and the CA grift too.
Catturd ™ (@catturd2) Tweeted:
$164,000 a year.
The math just isn’t adding up is it?
ChiNA
ChiNA
ChiNA
ChiNA……….
Did you say China?
.
The PDJT faces kill me.
Dead.
The media. TRAITORS.
That place will need to have a spiritual cleansing.
Posh Lake Tahoe compound…all that’s missing is a posh boat house. The whole thing is right out of Godfather ll.
GOOGLE FUNDED THE DEVELOPMENT OF COVID
.
.
.
Great clip!
The ChiCom saying that we have to genetically engineer humans to all get ill when we eat meat – that was a real doozy.
And to think that General Purplehair is going along with this shit!
Sad!
Cernovich (@Cernovich) Tweeted:
A think tank libertarian who I always had pleasant interactions with blocked me when I asked
– How does big tech have the resources to censor conservatives but not sex trafficking?
That’s the direct hit, that’s what they don’t want anyone asking.
Jack Posobiec (@JackPosobiec) Tweeted:
Landmark ruling
Three victims of child sex trafficking sued Facebook in Texas for allowing reciters to operate on their platform
FB argued 230 gave them immunity
TX Supreme Court ruled 230 does not https://t.co/WrT2YsOyhp https://twitter.com/JackPosobiec/status/1408629307681935365?s=20
A repudiation of their “230” immunity is big.
You think that’s bad, twitter has #incest filth and porn videos all over the place. And they banned President Trump🤬‼‼‼‼🤬
If you haven’t done so already, I highly recommend adding this website to the list of your daily “must” visits….
https://www.revolver.news/
It’s a lot like what Drudge used to be.
.
Darren Beattie is an Excellent Journalist!!
Looks like Bitchute is going to start paying attention to British and European control of speech.
LONG MARCH COMMIE COLLAPSE BEGINS.
https://support.bitchute.com/policy-changes/2021-06-01/
I have resisted joining anything after the Parler debacle. I did try Frank Speech but for some reason it didn’t take. Oy.
Anyway, I hope PDJT has a mega MAGA platform coming.
Telegram, Bitchute, Vimeo, etc. They are all falling.
Platforms have to tell foreign countries to SHOVE IT. Gab does this all the time. If it’s legal US speech, it’s allowed.
BTW, I put this science episode in the sidebar as number 9 in the series.
Steve – feel free to take breaks whenever you want. No rush.
Supreme Court limits California union recruiting in favor of property rights
https://calmatters.org/california-divide/2021/06/supreme-court-california-farmworker-unions-property-rights/
For the Left, it’s all about the collective, not individual rights and property rights as set forth in our founding documents. This is a good win.
Hmmmm.
Looks like I wasn’t the only one who saw this earlier this week…
.
https://twitter.com/SharylAttkisson/status/1408173991852969989
.
.
Very shocking read. This guy was no more guilty than any of tens of thousands of Trump supporters who were outside the Capitol Building. And yet FIB did their best to destroy him.
In my opinion, his mistake was cooperating in any way. Treat them as Enemies of America, because they are.
Name, rank, serial number, “talk to my lawyer”.
Original coverage:
https://nypost.com/2021/06/23/fbi-tears-new-yorkers-life-into-shreds-devine/
Picked up by American Thinker:
The FBI has destroyed an innocent man’s life over January 6
By Andrea Widburg
https://www.americanthinker.com/blog/2021/06/the_fbi_has_destroyed_an_innocent_mans_life_over_january_6.html
The FBI has become the DemonRAT STASI or KGB!
SPIT on the FBI and Wray!!!!!!
“This week, markets closed for the weekend at 3:00 PM Mountain Time
Gold $1880.70
Silver $27.63”
_____________
Nearly gaslighted me into a triple-take… 🤣 😂 🤣
I have the continuous silver contract displayed on one of my monitors pretty much always, since February.
Looks like silver closed the week at $26.087
https://www.marketwatch.com/investing/future/silver?mod=over_search
And gold closed the week at $1,777.80
https://www.marketwatch.com/investing/future/gc00/charts?mod=mw_quote_advanced
Dangit, I forgot to update that. Will fix it.
Fixed, though apparently you and I use slightly different services.
Inv 95L

10% chance of development …but subject to change.
(current) Intensity Guide
It’s got a healthy wind circulation:
https://www.windy.com/?11.932,-39.419,5,m:dQ3afqh
But doesn’t look all that organized right now:
https://www.windy.com/-Satellite-satellite?satellite,11.932,-39.419,5,m:dQ3afqh
And the wind currents right now would seem to push it south….to skim South America and later hit LA (again) or MS.
The Classics…
👉 one-upmanship
The 4 Yorkshiremen Sketch, 1967
pre-Monty Python
We Were So Poor
Then again, it’s sometimes difficult to get what you want….
no one leaves empty-handed
The Communist Quiz
(Monty Python)
💥🎵you shake my nerves and you rattle my brain💥🎶
https://www.youtube.com/watch?v=BQa7wOu_I_A
1957
🙌
Love it!
(parallel universe)
…and speaking of Che Guevara..
there’s this…
article…
The UN, Which Che Guevara Almost NUKED, Honors Che Guevara On His Birthday
link..
https://townhall.com/columnists/humbertofontova/2021/06/26/the-un-which-che-guevara-almost-nuked-honors-che-guevara-on-his-birthday-n2591580
posted June 26
birthday was on June 16 🙌
FTA..
“On this day {June 16} was born Ernesto Guevara De La Serna, known as “Che”, in Rosario, Argentina. Let us remember this person {and quite fondly, the Tweet seems to imply} by watching his historic speech at the UN General Assembly, December 1964.”
UNESCO Tweet en Espanol from June 16, 2021 in the article + more.
👉 FYI : not satire
Doesn’t look like a white nationalist to me!
.
https://twitter.com/disclosetv/status/1408735572919738371
Othal
?
what kind of a name is that ?
They like to be different i guess.
Did the tree house give it away? 😉
So are the feds goingbto take over their compound and burn it down ir give them a pass?
https://twitter.com/cathdad4/status/1408249365026643969
😡🤬
This is beyond sick. They got away with it for the little school kids so now they have free rein to demoralize the military.
It’s so strange to me. The kids..has been obvious grooming and indoctrination. With grown men and women with preferences already ingrained…why start the drag queen stuff? I still have a hard time believing these things are really happening..but they are
I remembered learning about decadence prior to ww2. So happens a book was written on it. Look at this and compare!
https://www.dailymail.co.uk/news/article-2847643/Berlin-liberal-hotbed-homosexuality-mecca-cross-dressers-transsexuals-male-female-surgery-performed-Nazis-came-power-new-book-reveals.html
Wow. I knew it was prevalent but not to that extent.
Yep. And thats just what they put in the article. Did you see that it was a whos who of artists, royalty, and political class? If we are having a resurgence of the political thought akin then the decadence is as well from the same types.
“We are no longer accepting comments on this article.”
Here is the archive of the article, just in case…..
https://archive.fo/8VCTG
It’s so awful and disgusting and jarring. God, make it stop, please.
Because we would have been so much more effective and ready during WWII if only…
These people are insane.
Rally thread scheduled for noon EDT.
https://twitter.com/disclosetv/status/1408754636501180417
Bite Me’s handlers WISH they could get any interest at all let alone this.
Thank you for the history lesson. Exciting, and wish every kid had access to history and science entwined like that. Makes learning easier.
Verse of the Day for Saturday, June 26, 2021
✟
“Let your conversation be without covetousness; and be content with such things as ye have: for he hath said, I will never leave thee, nor forsake thee.”
Hebrews 13:5 (KJV)
Thank You, Jesus, for blessings received and prayers answered!!!
BE MY VOICE
PRAYING ON THE ARMOR OF GOD
Father God, I now follow your command to put on the full armor of God, because my battle is not against flesh and blood but against rulers, authorities, the powers of this dark world and against spiritual forces of evil in the unseen world.
I first pray on the Belt of Truth that it may be buckled around my waist, may I be centered and encircled by your truth dear Lord. Hem me inside all that is true and right, and may I be protected and held up by the truth of your living word, in my Lord Jesus name.
I pray on the Breastplate of righteousness, please protect my vital organs and my inner man, cover my integrity, my spirit, and my soul. Guard my heart for it is the wellspring of life, please strengthen and guard the most vulnerable places in my life with that which is right, good, and noble that I might not receive a fatal blow from the enemy, in my Lord Jesus name.
I pray on the Gospel Shoes of Peace. I choose to stand in the shoes of your good news, and on the firm foundation of my Lord and Savior Jesus Christ, the solid eternal rock. All other ground is sinking sand, I pray that I will not slip or fall, but that my feet would be firmly fitted on your lordship, my Lord Jesus. I choose to stand on you, so that the peace of God, which transcends all understanding will guard my heart and mind in Christ Jesus, the eternal Rock of Ages. I receive your holy peace now my Lord, from the sole of my feet to the crown of my head, in my Lord Jesus name.
I pray the Shield of Faith into my hand now. As I take up the shield of faith, I ask that you might extinguish every dart and arrow, that is launched from the enemy to take me down spiritually, physically, mentally, emotionally, and every attempt of the enemy to destroy my joy. I ask that my faith in you would make it flame out. Extinguish every flaming arrow that would come against me, my life, my family, my home, or my ministry. May my faith always be out in front of me like a shield. Give me the courage to “faith my fears” by choosing to walk by faith and not by sight, in my Lord Jesus name.
I pray on the Helmet of Salvation, that you might protect my mind from the thoughts that can lead me astray. I choose to take every thought captive, and arrest all intentioned ideas and motives that would harm others, or distract me from your holy will for me. I submit every captured thought to the Lordship of my Lord Jesus Christ, and ask that you would imprison those thoughts that are not of you my Lord. Transform my mind and renew my thinking that I may think God thoughts, and have a sober mind that is focused on your glory. Please protect me from being double minded that I may allow my mind, I reject to live an earthly life, because I choose to live a holy one, governed by you My Lord Jesus, the prince of peace, please have my mind to be saturated with the holy mind of Christ, in my Lord Jesus name.
Finally, I take up the Sword of the Spirit which is the holy word of God, I pray this powerful offensive weapon into my hand, and ask that your holy word would be fitting for every encounter I face. As the enemy gets close to me, please give me the insight, wisdom, and skill to wield the word of God to drive away the enemy, in my Lord Jesus name.
May the enemy and his team flee from me, upon hearing the word of God spoken by the power and direction of the Holy Spirit. Give me the sword of the spirit to cut through the wiles of the devil, so that I may discern the schemes of the enemy when he is near.
With all kinds of prayers, supplication, and intercession I pray to you my Lord God as the one who fights my battles. Now that I’m in your holy powerful armor, I walk away covered and ready to face my day as you go before me, and please protect me in the midst of the spiritual warfare in this unseen world, in my Lord Jesus name.
Thank you my Lord, for the spiritual weapons of armor and prayer that you have given me. It is written no weapon formed against me shall prosper, and you will refute every tongue that accuses me.
Thank you Father God, my Lord Jesus and the Holy Spirit, that I am more than a conqueror in my Lord Jesus. I pray all of this in the mighty name of my Lord God and Savior Jesus Christ, amen.
Will you be making a quick rally thread? This is the stuff for it.
Or, are we going to stick with just the daily?
Nevermind i see the note.
It’s scheduled to drop at noon eastern.
EXPLOSIVE REPORT! WE CAUGHT THEM AGAIN: New Findings Show Coordination and Collusion Between PA and GA in 2020 Election Steal (thegatewaypundit.com)
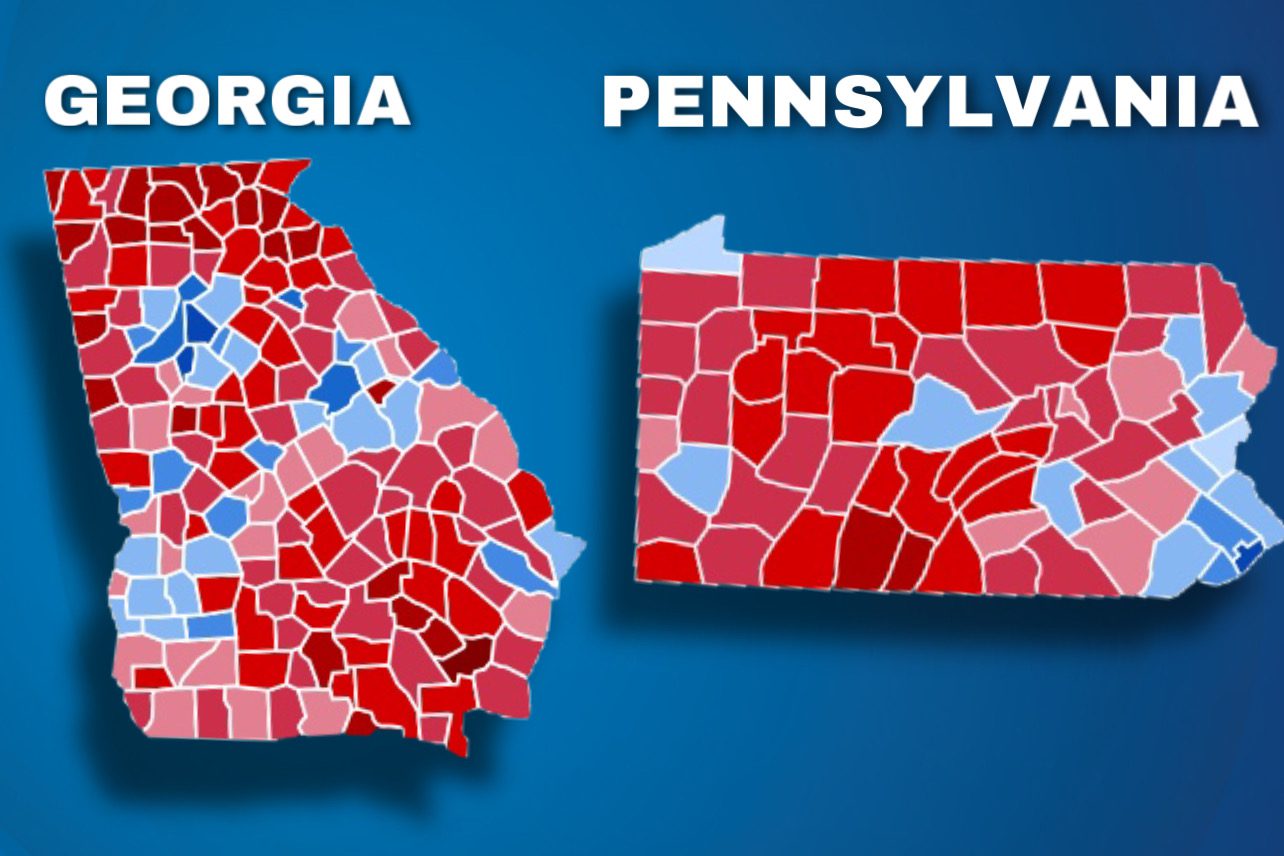
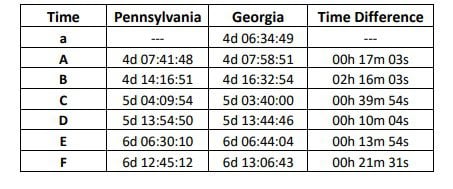
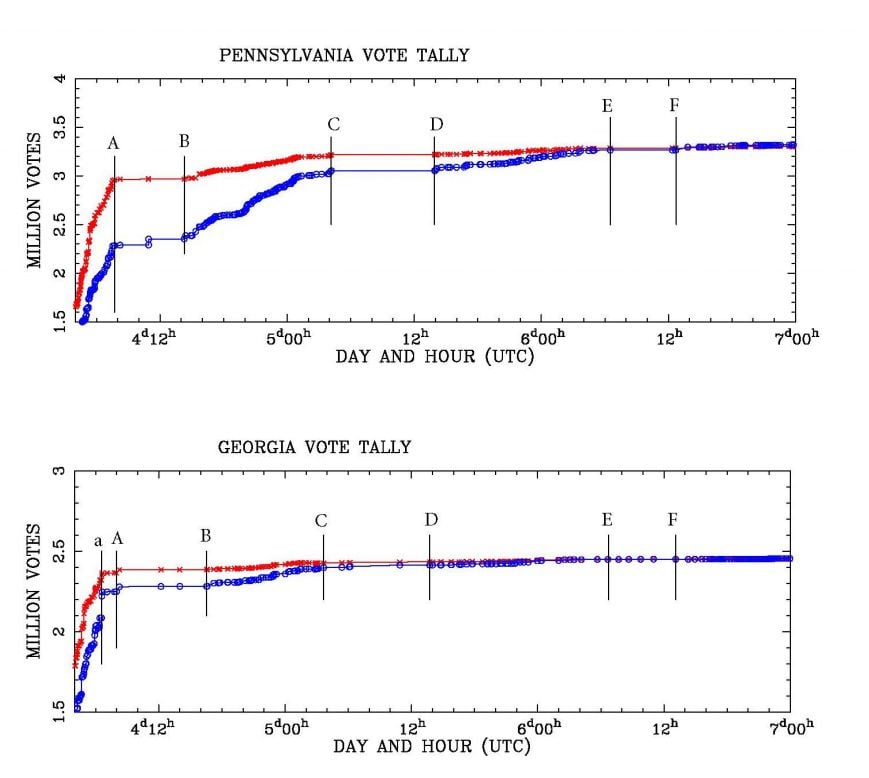
I look at those results and I just refuse to die until Mark Zuckerberg is behind bars.
So to keep you around, we have to slow-roll Zark Muckerberg’s arrest?
Well, no matter what, I hope they do it before the parts just start dropping off! 😉
BREAKING UPDATE: HISTORY MADE AS FINAL ARIZONA BALLOT INSPECTIONS COMPLETED – Huge Preliminary Report Expected Monday When True Number of Ballot Totals Announced (thegatewaypundit.com)
KATIE HOBBS FOR PRISON!!!
Woke, Inc.: Inside Corporate America’s Social Justice Scam Hardcover – August 17, 2021
by Vivek Ramaswamy
A young entrepreneur makes the case that politics has no place in business, and sets out a new vision for the future of American capitalism.
There’s a new invisible force at work in our economic and cultural lives. It affects every advertisement we see and every product we buy, from our morning coffee to a new pair of shoes. “Stakeholder capitalism” makes rosy promises of a better, more diverse, environmentally-friendly world, but in reality this ideology championed by America’s business and political leaders robs us of our money, our voice, and our identity.
Vivek Ramaswamy is a traitor to his class. He’s founded multibillion-dollar enterprises, led a biotech company as CEO, he became a hedge fund partner in his 20s, trained as a scientist at Harvard and a lawyer at Yale, and grew up the child of immigrants in a small town in Ohio. Now he takes us behind the scenes into corporate boardrooms and five-star conferences, into Ivy League classrooms and secretive nonprofits, to reveal the defining scam of our century.
The modern woke-industrial complex divides us as a people. By mixing morality with consumerism, America’s elites prey on our innermost insecurities about who we really are. They sell us cheap social causes and skin-deep identities to satisfy our hunger for a cause and our search for meaning, at a moment when we as Americans lack both.
This book not only rips back the curtain on the new corporatist agenda, it offers a better way forward. America’s elites may want to sort us into demographic boxes, but we don’t have to stay there. Woke, Inc. begins as a critique of stakeholder capitalism and ends with an exploration of what it means to be an American in 2021—a journey that begins with cynicism and ends with hope.
Sounds very interesting. I have I on my list.
Corporate America loves controlling their people through woke corporatism. It’s a win-win for them, while employees suffer with unnecessary and unwanted woke training and constant pushing of sjw BS.
Dudes a little annoying but the line is picking up. I sure hope the place gets full. I dont want it to be like Oklahoma.
I hope it fills up as well.
People – and by people I mean our side, 2016 & 2020 – are in a very strange place right now. And I include myself in this. In any free time that I have I monitor the situation, all day every day. And have been for 6 years running. I pay attention constantly, looking for that nugget that will allow me to actually see the end of the tunnel. I certainly can’t let it go – but I’m weary of paying attention, of searching, of the confusion, and the disappointment.
People are simply shell-shocked to the point of paralysis. Like-minded people don’t know what to say to each other about the situation, because there is no path to follow. The conversation lags, then ends almost immediately. And I’m speaking only of those on OUR side. I don’t engage the other side at all – family, friends or acquaintances.
And I realize that a big part of this is that so few people pay attention to the level that I do. My husband, and I and everyone here.
So as I said, I hope there is a big crowd. And I hope it isn’t only “remember the good old days” and “what a disaster everything is now.” The shift has got to come, because our people are in a holding pattern looking to survive.
Arizona preliminary report Monday.
Good post. I will just say that I don’t expect anything more than “remember the good old days” and “what a disaster everything is now,” for now. The waiting is excruciating, but I think we are still in waiting mode. PDJT might mention something happening next week, which would be great, but what if it’s “just” a lawsuit being filed or something ike that? I don’t think any hammers are going to drop tonight, unfortunately.
I just pray he doesn’t bring up the vaccine again. Getting the shot vs. taking meds for prevention is something that people ARE actually able to see their way forward on – and have done so by this point. No reason to bring it up, and you best bet that crowd will be gritting their collective teeth hoping he doesn’t.
Thanks for the thumbs up. 🙂
I agree with you. People are not done as much as they havent made a decision to move forward.
I’m not looking to move forward. There is nowhere to move forward to. I just need the shift to begin, in plain sight.
Keep in mind this data is from VAERS only, no way of telling the REAL numbers.
NOT MAKING HEADLINES: CDC Officials Admit More Hospitalizations of Young People from Vaccine than From the Actual COVID Virus – Including HUGE Number of Heart Problems Reported (thegatewaypundit.com)
Earlier this week the CDC admitted to finding more cases of rare heart inflammation in young adults than was expected after receiving COVID vaccine shots.
The Wednesday CDC presentation shows a surge in myocarditis/pericarditis cases in young adults after their second dose of the COVID vaccine.
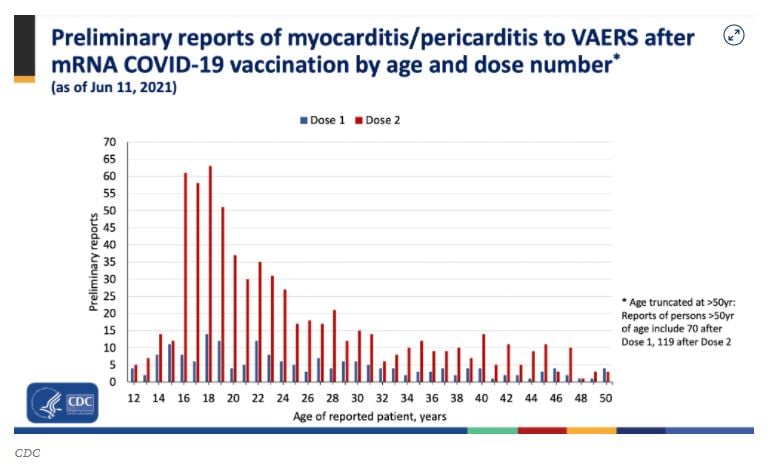
Former New York Times reporter and author Alex Berenson watched the CDC presentation was shocked with their results and dangerous advice to American parents and their young children.
The CDC’s own voluntary vaccine side effect reporting system received about 48,000 reports for ALL vaccines. This year that number is at least seven times that many for Covid vaccine cases alone.
Berenson then notes that from the CDC’s own data they are seeing more children and young adults hospitalized from the COVID vaccine than they ever saw from the COVID virus.
Via AlexBerenson:
This is shocking news.
So why are kids being vaccinated again?
And why are the CDC leaders and their cohorts and enablers like Dr. Fauci still walking the streets?
This is medical malpractice on a national scale!
God help us!
Read the entire report here.
They have gone ALL IN on the plan to control and manage humankind through bio-engineering.
Crime against humanity on a global level.
This needs to be banner headline news. The whole MSM is just as in the tank as the CDC is.
Major Patriot
@MajorPatriot https://gab.com/MajorPatriot/posts/106477340658805133
Tell me again why anyone should get vaccinated?
The Bias News
@thebias_news https://gab.com/thebias_news/posts/106477118823889114
WSJ: About half of adults infected in an outbreak of the Delta variant of COVID-19 in Israel were fully inoculated with the Pfizer vaccine.
A COLD has been turned into a weapon against Humanity, thanks to…..
CHINA AND DEMOCRATS.
Oh Boy! 🙄🙄
Crazy Biden Admin Issues Order Mandating Race Training at Every Level of the Federal Government (thegatewaypundit.com)
How long until this illegal regime gets terminated?
Hopefully Arizona’s Audit preliminary report on Monday starts the timeline.
I’m waiting. Impatiently.
August, September, October, November gonna be lit as more states jump on the audit train. Arizona actually had the LEAST amount of ballot shenanigans of the 6 swing states and the results are gonna be YUGE!!
I’m not holding my breath on anything being released Monday. If it is, great, but too often *things happen*.
Communist pieces of SHIIIIT.
Communist pieces of SHIIIIT.
Communist pieces of
Communist pieces of
Communist pieces of SHIIIIIIIIIIIIT.
I gather you’re unhappy.
😃
You gather correctly!!! 😃
Is that some kind of Limerick?
(In any case things are worse than St. Ives)…
And they’re doing their cr@p the world over,
WITH OUR MONEY!!!
How soon is monsoon… bend that damn dam till it shows the “quality” of ChiCom comstruction…..
Now here’s a Communist tale
That is really beyond the pale:
They made a pandemic
That was only a gimmick.
They should all be rotting in jail!
They always play with the years of combat etc. Same with Vindman.
A crook is a crook whether in the military or anywhere else.
The mockery and criticism are really very dangerous to their authority.
I love it. Pantywaisted pukes. Spit.
And it’s HIGH TIME.
The Cabal’s FIFTH COLUMN in the US Military is a DISGRACE.
“Protect yourself from name calling and mockery – become a public official.”
CLOWN MIL IS CLOWN MIL – working for everybody except America.
Maybe even for “foreign” forces. 😎
…which sounds like treason.
BARRY R. McCHINA SAY WHUUUUUT?
Thoroughly “modern” Milly
Is nothing other than thilly
With rainbows and dykes
His “heroes” on trikes
They still cannot find their own willy….
McCaffrey has been an anti Trump media stooge during the entirety of PT’s presidency. This and the fact that he’s a Council on Foreign Relations minion (and lots of other globalist organizations affiliations) is enough to spell out his bona fides for shooting his mouth off.
FLCCC Weekly Update —Authors of Peer-reviewed Paper Discuss the Irrefutable Science for ivermectinhttps://youtu.be/CauMLm7t_mk
I tried to convince people for decades about Fake Science – and CHINA did it in a year! 😉
Wow.
DP…Thanks for this.
I was getting a little bit worried by the First-In-Line crowd: Not many folks…but no time mentioned.
Gosh, I hope the turnout is Epic!!!
LOL..Her fashion sense is horrible





Didn’t she steal a tablecloth for some other horrid outfit she wore?
Here’s hoping it’s CURTAINS for the Bye,Dones and Kamel-Huh and that THE DIAMONDS ARE RETURNED POSTHASTE (or sooner!)…
Aaaaannnndddd,
Epstein didn’t kill himself,
McAfee didn’t kill himself,
And Bye,Done didn’t win…
Do you know I saw a tablecloth with that lemon print, or pretty close to it, at Homegoods in the last week? I almost bought it just for the laughs.
This is how to do floral. Effortless, and the posture. Always effortless. Just tossing it off.
Gollum’s sister???
It’s live.
https://www.theqtree.com/2021/06/26/rally-thread-save-america-wellington-ohio-june-26-2021/
🤸♀️ 🤸♀️ 🤸♀️ 🤸♀️ 🤸♀️ 🤸♀️
TYSM!!! 😍 😍 😍
Nice catch, Glenn Greenwald.
McCaffrey should be well aware that libel and slander laws with regard to public officials are FAR different than those protecting us normies….
The bar is set MUCH higher for said offenses to have occurred in the case of public officials… and he’s probably had training about that somewhere along the line, and especially working in the media….
First Amendment. Communists should read it.
They come not to tell the truth, but to Barry it…
Only 20 points?
Rumble Link
LIVE: President Donald J Trump in Wellington, OH (rumble.com)
PDJT’s Rumble account picked up 45,000 new followers in an hour.
https://www.thegatewaypundit.com/2021/06/president-trump-opens-verified-account-rumble/
I wonder if Torba at Gab is ticked (even though they are different kinds of platforms).
OK folks….I am thrilled, thrilled, thrilled…Canada’s own citizen owned newspaper is growing like crazy:
https://druthers.net/
Here are some of the articles in it:
https://druthers.net/absurdity-observer-june-2021/
https://druthers.net/vaccine-casualties-is-the-cdc-hiding-the-real-numbers/
https://druthers.net/hacking-of-the-human-mind-they-vs-us/
The True North, strong and free is rising up….Amen! Amen! and Amen!
Excellent! I cannot wait for the day that Tru-dope has to flee to CHINA.
Trump-appointed judges obstruct Biden ‘pen and phone’ policies just months into term
.
Trump judges stymied Biden administration on parts of COVID relief bill, oil and gas leases and more
.
https://www.foxnews.com/politics/trump-appointed-judges-block-biden-policies
Florida man wanted for shooting cop in the head apprehended at black nationalist paramilitary encampment following multi-agency manhunt
https://www.theblaze.com/news/daytona-beach-police-shooting-othal-wallace
A black nationalist paramilitary encampment?
THANK YOU, CHRIS WRAY!!!
Christopher Wray is evil.
Wrong Way Wray…
Or maybe Wong Way Wray…
Both!
Small comfort….Daytona Beach Police Chief Jakari Young said that officers used (Officer)Raynor’s handcuffs to apprehend Wallace.
Strange that the military camp wasn’t surrounded by armed FBI and every nook and cranny tossed…or their other method…killing people or burning it down….oh well..it’s good to be a black anti govt militant in 2021, USA
^^^ EVERYTHING about this guy says terrorist extremist. DOJ and FBI gives him a pass. Behavior OK based on he’s NOT whitey.
FIB is Soviet American political police, with a staff deluded into thinking they’re regular LE. It’s an abused agency.
Hmmm. Haven’t heard much from Louis Farrakhan’s “Nation of Islam” lately…..
“A black nationalist paramilitary encampment?”
____________
No, no, no!!! This is all wrong!
Narrative violation!
We’re supposed to be paying attention to the TWO most dangerous threats to our nation, Globull Warming and White Supremacists — numbering in the tens, maybe even the tens of tens!
[also Unidentified Flying White Supremacists and Globull White Supremacists who are Warm but not Warming!]
Quick article about a lady who got out just in time
https://nypost.com/2021/06/26/florida-survivor-recalls-escape-from-collapsing-condo/?utm_medium=SocialFlow&utm_source=NYPTwitter&utm_campaign=SocialFlow
Florida survivor recalls escape from collapsing condo: ‘You need to run’
I don’t believe it.
You think it was demolition and her story is disinfo ?
No, I just can’t see enough time to assess the situation and then act. Too many people watch too many movies.
Good to see you pgroup! 👍
Damn happy to be here!
👍😁
A good point. If I was reading it right..she also ran down 6 floors of steps, didn’t attempt an elevator
I wouldn’t have attempted the elevator either. Could be the elevator was at the far end of a hallway and the stairs were closer, but regardless, you could probably already be down 4+ flights of stairs before the elevator even arrives at your floor.
A younger person might make it down all six flights of stairs before the elevator arrives at the sixth floor.
And you don’t want to be stuck in the elevator if the power goes out.
In case of any emergency that would require evacuating a building, the stairs are probably your best chance.
Yeah, there are some of us who know how to react in a situation.
I ran down 10 flights when a tornado appeared near a building once – and stopped on the ground floor to exit and watch it, before proceeding to the basement.
You were extremely lucky. Back when I had just graduated college, I got into a suit and had a portfolio full of paper resumes and intended to hand them in at every accounting office in a particular office building in Century City. I took the elevator to the sixth floor, did all the offices on that floor, then figured it would be quicker and easier to go down the stairs to the 5th floor.
All the doors all the way to the ground floor were locked.
At the ground floor, it let me out of the staircase — onto a narrow curb next to a driveway into their parking structure, with cars zipping by…..and it relocked behind me.
Trust me – that door never shut behind me! 😉 I swapped door duty with two others who wanted to see the funnel.
I was too startled to find there was no designated walkway from the exit on the open side of the door, so I let it shut a bit to see if there was a walkway that I couldn’t see on the other side of the door.
Y’know how some door dampers hold and slow, then just go “swoosh, snick”? At which point, I gave it a vocabulary enhancement. There were no walkways in either direction. The edge of the door literally reached the traffic edge of the curb, and that was all the space there was.
If the same sort of thing happened today (when I’m older and
wiserslightly less stupid), I might not have gotten caught — and I might anonymously call it in as an OSHA violation. I can just imagine if a fire alarm went off in the tower and a dozen people were injured/killed by autos outside that door.The driveway, BTW, was for the parking lot of the Sherman Oaks Galleria. Connoisseurs of the form might recognize it as the mall in Fast Times at Ridgemont High, Back to the Future Part II, and Terminator 2. It’s part of the LA experience, where you see something on film and go, “I bought a pair of shoes in that store.”
I think I read somewhere that that mall had been torn down.
The main part of it was substantially remodeled as an open-air mall — which, given that it was a multi-story air-conditioned enclosed mall, is much the same thing.
Not when it hits a hundred it isn’t.
Headed to Frogstar B /HHGTTG) 😉
Yep. Situational awareness is crucial, especially in times like these… seems LEO, etc., are minutes/hours away, when seconds matter…
And it’s not always (or even usually) their fault. Over here, LEOs are being reduced/workforce-managed/etc. and “unempowered” like never before. Local PD is about 10 miles away, HQ about 50 miles away… yet we still pay as much for “security”… Yeah, right, Angie-potatokopf…
There he is. Pictures or it didn’t happen😂
New Political Moonshine article/video:
VIDEO: Surfside Condo Collapse – Controlled Demolition?June 26, 2021
https://politicalmoonshine.com/2021/06/26/video-surfside-condo-collapse-controlled-demolition/
Intro: “VIDEO: Does close examination of the Surfside condo collapse suggest controlled demolition? It absolutely does.
Why is the front end video edited out in every available version of surveillance footage that otherwise should be widely available for examination?
Is it because it would depict the obvious detonation of CD charges? Yes, that’s precisely why.
Watch and decide for yourself knowing that there is a reason the aftermath so closely resembles that of 9/11.”
OK.
But the building was occupied by property owners and folks leasing.
Part of why I bring all of this up, besides it FITS SEASIDE in FL.
Thoughts?
MeThinks both buildings taken down by controlled demolition.
—
Besides FF with AZ audit finished. ut this doesn’t really fit. Seaside to be FF should have come down Monday morning. early. A few hours ahead of AZ audit release.
Have read McAfee rumored to have “dead man’s switch” info in seaside building. Not really buying this. This total novice and poor strategic planner, would have multiple locations to ensure “dead man” switch(es) succeeded. Redundancy would help ensure success, if McAfee met his demise.
Paraguay or some country politico in the building? Respectfully, MeThinks he is NOT important enough for such an operation. PLUS, lots of logistics issues to say the least.
Spit balling here.
Wonder if Daughn has thoughts on Seaside. She had some great threads on FL, including a few comments that smelled of cartel operations. Money laundering that she walked away from.
Just a quick note to your causes. I’ve not been following closely, but initial reports claimed renovation was occurring on the roof portion of the Tower that did not collapse. Gives reason for work men to be in the area. Those were initial reports and I’m not aware if they’ve been modified, ruled out or expounded upon.
Yep. There was a picture of that up yesterday, and it looked REALLY grotty as compared to the other buildings around. Also read elsewhere that there’s a fire in the rubble, which sounds oddly familiar. And sadly familiar, as it’s hampering search and rescue operations…
Wonder, though, if there was something else, perhaps related to election or other fraud, which was about to be discovered, or “needed to be” removed before it was seized???
Exactly. This is the SAME THING as 9/11 – they had all kinds of workmen in there with “ostensible reasons” because of asbestos.
It’s an interesting video.
I noticed the flashes immediately, but I don’t know how to tell the difference between an explosive charge flash and a random flash that could come from an exploding circuit breaker (or back up power generator, or similar).
Against a pitch black background, even a camera flash would stand out. As the building fractures and begins to collapse, and electrical wiring throughout the building is being torn, could random power surges cause light bulbs to momentarily flash ‘on’ as they explode?
It seems that explosive demolition charges would be better placed near the foundation of the building, but it’s possible they were, and those flashes are not visible.
There were multiple flashes on the roof of the section that did not fall. What was the source of those flashes, and could they also explain the other flashes that appear to be coming from various condos throughout the building?
The woman in the article, who woke up to a loud noise and thought her balcony door had come loose, tried to close it and couldn’t, later guessing that the building had already shifted, making the door frame no longer square / level.
Then she noticed large cracks spreading across the wall, which is when she grabbed a couple personal items and ran for her life.
The article, citing Zillow, says the condo sold for $600K. According to the woman who escaped, there were known structural issues that were not disclosed when she bought the condo, and each condo owner was being charged $1K for structural repair.
If I remember correctly from a previous article, there were at least 55 condos in that building. Assuming $600K is the average value, with 55 units, that’s a $33 million building.
We don’t know how many condos are owned vs. leased/rented.
Political Moonshine’s author is a former insurance fraud investigator.
I would want to know if a significant number of condos in that building were owned and operated by a property management company, REIT, individual investor, etc., someone who stood to lose a lot of money if they found out that the structural damage was severe enough that it might be cost-prohibitive to repair or would have led to the building being condemned.
What was the insurance policy on the building, was it paid up at the time of the collapse, how long had the insurance policy been in place, who is the beneficiary, is the insurance company doing their own investigation or settling quickly, etc.
That’s probably a dead end, but I would not rule anything out until I was sure.
Have read comments about the possibility of short circuits and the like as the building collapsed and live circuits shorted out.
Likely many insurance adjusters are looking quite closely at the collapse.
Have not looked into fake news for information. I do hope ATF, DOJ and FBI stay the hell away from seaside.
“I do hope ATF, DOJ and FBI stay the hell away from seaside.”
___________
Decent chance we’ll find people from all three agencies already there.
Buried in the rubble… 😁
To paraphrase Briscoe Darling, from “Mountain Wedding”, on the “Andy Griffith Show”, “gotta watch out for those hot flashes”……….. 😆
“It also FITS HAMAS HQ being taken out by the Israeli’s. MeThinks Mosad. NOT the Israeli AF.”
Yesterday while out walking, the revelation slapped me in the face, above is totally out to lunch.
Back when the Hamas HQ destruction was being discussed in various posts, I posted a pic of a likely LGB – Laser Guided Bomb. Likely a penetrater LGB. It was dropping literally was dropping directly down towards Hamas HQ. It was not a controlled demo.
Surely wish my memory recall was more timely. 😉
Seems ODD we are only seeing video from one distinct location. Really? No other CCTV in the “high rent” area that could have caught something?
Maybe. But I am not buying this either.
Something IS really OFF about this whole Seaside collapse.
Agreed. That building coming down just like Building 7 is totally suspicious.
Also we’ve only been shown a video at the precise moment the building began to fall. Not even seconds earlier. Where’s the earlier?
Read elsewhere that is a flag of sorts. Where’s the videom from before the collapse? One minute? Five, ten….? Should ALL be available.
Even my crappy eight channel CCTV security has ~22 days or so of data. Then it over writes oldest data on the HD.
Note the discussion of “recertification” of buildings greater than 40 years old.
This potentially leads to pulling certificates of occupancy for buildings en mass, and in this location, mass condemnation of waterfront property. Conveniently fits the agenda, and leverages a crisis.
Build back better.
I’m bringing this comment over from something shared yesterday, in reply to this original comment:
https://www.theqtree.com/2021/06/25/dear-kag-20210625/comment-page-2/#comment-757021
By the way, it is possible that 2 different people posted about the building collapse using the different timed twitter video so that the discussion I’d previously read exists elsewhere in yesterday’s comments…
Gingersmom posted the reply above to which my copied comment below replies…
https://secure.gravatar.com/avatar/708c239d356f60bb11c40b8c2e6c65e0?s=64&d=identicon&r=g
Valerie CurrenOnline
Coyote
Reply to Gingersmom2009
June 26, 2021 17:34
OK, this is strange to me because I’ve had to re-load the page a few times & I was re-reading the comments to this reply (w/ the different speed videos of the FL collapse) & a bunch of previous replies are now missing, including several claiming to think this was all just a natural occurrence (which I thought when 1st reading them was strange & an opinion I didn’t share even though it seemed to have been started by Wolfmoon)…
So, is someone messing w/ the Q-Tree site or are we now no longer leaving All replies as “evidence” & for historical accuracy. If WM started the “natural collapse” viewpoint & later changed his mind wouldn’t he normally leave up his previous opinion AND spell out the steps that led him to changing his mind?
What’s really going on here???
Valerie, the discussion you are referring to is still there. It happened on page 1 of the comments, then Gingersmom brought it up again on page 2. I made the original post about it here:
https://www.theqtree.com/2021/06/25/dear-kag-20210625/comment-page-1/#comment-756485
The discussion below it included Wolf’s thoughts on it.
Thanks so much Linda!
Just for the record, I don’t delete posts. If posts ever go missing, it’s a PROBLEM.
The insurance company for the building will get to the bottom of it. They will be looking to exclude coverage as usual. Excluding all the conspiracy stuff for a second, the most likely cause is a foundation failure, sink hole. Most policies exclude ground movement, ie earthquake, sink holes unless there is specific language or an endorsement to the policy to add the coverage.
As to the controlled explosion you would have to explain that one.
Bomber: Ding dong. Hi, I need to come in to attach these explosives to the structural column in your apartment and run these wires. I will need to rip off the dry wall while I am at it. Please excuse my mess.
Renter: Sure, come on in.
All the flashes can easily be explained as electrical arcing or if they had natural gas flashes from that igniting in spots.
For the sake of argument, the bldg super/security would know which rooms were currently unoccupied by either vacation status or not leased/sold.
The bldg has a basement and work crews were involved in roof work on portion not collapsed. They could of had business/easy access to basement.
Is there a history of sink holes taking out towers on Miami Beach properties? Or anywhere else?
Pretty sure this is the first.
Wires are so too 2000. In 2021 we just ask Alexa.
While sink hole and structural problems with bldg sinking a bit looks to be the right answer but the timing sucks. Biden meeting with the mayor of Miami (why did so called Republican Mayor get access?) the very day before is suspect, as is that info not being known until Kamala had to remind Biden to to mention the towers or else he wouldn’t of said anything and when he does he mentions the mayor meeting and proceeds to give a discombobulated response to the incident, there’s distraction for McAfee or AZ being a possibility, Dog comms were bandied about well prior to this as being suspect and then Champ (as in Champlain Towers) died what, two day’s before? One could call the Champ thing a coincidence but then one wouldn’t know how long this has been in the works (kinda like pulling the available disaster of the day/week/month off the shelf and scheduling it). Then there’s the Ivanka one block away thing with the need for Trump to stand down.
Concerning Sink Holes. Miami Beach has a very very poor history of sink holes. Other areas that do in Florida don’t seem to take down towers.
Can’t recall this being a problem with towers in the rest of the world either.
http://www.luckysci.com/wp-content/uploads/2015/09/world-sinkholes.jpg
Does it have to be demo? What about sound amplification to weaken structure or loosen soil?
Anyway, plenty of room for conspiracy theory here, and little room for trust, right or wrong.
Thanks, RF, good points to ponder…
For a controlled demolition, wouldn’t you need to blow just a couple of the top floors to go down before their weight would collapse the next floor, and so on, all the way down?
Would you need to wire the entire building to make it collapse like that, or just the top 2-3-4 floors?
I would have no idea. My mind would say the opposite, if you collapsed the bottom few floors then the cascade effect would be uncontrollable. But regardless, collapsing into the building’s footprint seems evidence of professional planning to my way of thinking.
Way back in the OKC Murrah Building era a friend in OK said that there had been early reports of rescue personnel seeing unexploded ordinance attached to support pillars in the remaining portion of the building. Also there were reports of “seismic” activity showing multiple “events” reflective of perhaps multiple charges going off…
Good grief! It just hit 107 degrees here, and tomorrow it’s supposed to hit 111! That has to be an all time record.
In better news, our ditzy governor Kate Brown is rescinding all COVID restrictions on July 1st. I guess she was waiting until everyone else did.
Kate Brown is the man Mike DeWine hopes to be!
“Kate Brown is the man Mike DeWine hopes to be!”
____________
And Mike DeWine always hoped to be a woman, so it’s all very confusing! 😂
Heck, they just renewed our local temp to 111, on Monday. We are on a peninsula surrounded by the Puget Sound. It should be impossible yet here it comes.
Well, it appears that ** whoever ** planned and executed the controlled demolition of the Surfside buildings is making quite sure that all the evidence, and possibly any survivors, are destroyed:
http://www.msn.com/
1 hour ago
“Miami-Area Collapse Search is Hampered by Fire”
Down and Dirty Summary: There is now a large fire that is raging inside the rubble of the two collapsed buildings of the Champlain Towers at Surfside, Florida. There is heavy smoke and noxious smells everywhere. More than 130 firefighters are on the scene.
At least 159 people remain unaccounted for. Search dogs, mobile cameras, and sonar devices are being used to try to locate survivors and/or to recover the dead.
The first lawsuit against the condo’s HOA was filed yesterday, charging that the HOA failed to properly maintain the buildings.
Numerous officials, from Gov. DeSantis down through the Miami-Dade Police Department, are promising a thorough investigation “once rescue and recovery operations are complete.”
The department of buildings inspection stated that an audit of all buildings 40 years or age and older will begin, to determine potential safety issues in these structures.
Me: Here’s a tip for you, Gov. DeSantis. Find any det cord before it’s burned away. Have the other Champlain Towers complex buildings searched with a fine-tooth comb for any evidence of det cord and explosives Get a copy of the slow-mo video of the demolitions from @swaffarforcongress and have it analyzed frame by frame in the highest resolution mode possible by several independent professionals. Get any CCTV footage of the entire area going back for the last month and have this analyzed frame by frame. There’s a start for you.
Lead Witness in US Case Against Julian Assange Admits To Fabricating Evidence For FBI In Exchange For Plea Deal
And THAT’s just the tip of this disgusting iceberg…the details are stunning!
.
https://stundin.is/grein/13627/key-witness-in-assange-case-admits-to-lies-in-indictment/
.
We need a new federal law that makes double or triple the normal sentencing guidelines in a criminal case against FEDERAL law enforcement agents and/or employees of the DOJ mandatory with no possibility of parole until AT LEAST 75% of the sentence has been served. This law should be RETROACTIVE.
Time to up the stakes for malfeasance of ANY kind.
As the joke says, its a good start. Your suggestion should become federal law.
Any abuse of power. That includes making sure your cousin gets the contract for the motor vehicle department and other small-time shenanigans. Government power is force and shouldn’t be there so you can feather your buddies’ nests….much less so you can persecute people.
The problem is always ENFORCEMENT.
If there was consistent and strict enforcement, we wouldn’t need 90% of the laws already existing.
Things just keep getting worse, so we add new laws, and things get worse, so we add new laws…
Enforcement is the missing key.
And the reason it’s missing because FibDoj is a 100% criminal enterprise.
well, yes, that too. good point.
That bldg collapse in FL.
It’s got everybody’s attention. Spending so much time analyzing what happened.
Hmmmmmm.
Makes me wonder what else may be happening that no one is paying any attention to.
Can’t help it. It’s the times we live in. \_ 🤨 _/
Georgia voting law threatened by Chicago dirty judge and now head of DODGE Merrick Garland.
New Political Moonshine (#2 today):
Surfside Condo Collapse Threads Directly to China, the CCP and The Swamp
June 26, 2021
https://politicalmoonshine.com/2021/06/26/surfside-condo-collapse-threads-directly-to-china-the-ccp-and-the-swamp/
Intro: “Understand that we’re picking-up in the middle of the biggest global conspiracy in history – the generational political overthrow continuum with 9/11 and COVID-19 situated on it as sequential steps. As I have explained before, it’s akin to this,
Today’s coiled rope has us seeing with vision as clear as mud and with the task of coming to understand how the Surfside Champlain South Tower collapse threads directly through to China and the CCP. Your vision will become crystal clear shortly so bear with me.
The work lead me to ask whether this plagued building with documented structural integrity concerns dating back to the 1990s was hand selected to fall at a precise and needed time; and whether there may be a line of connections and nexuses from one end of our rope to the other and thus supportive such a notion.
I’m writing this article because I found exactly that.
By the time you’re done reading this, you’ll likely agree with me in that with a modicum of understanding about basic Chinese doctrine and the CCP’s tactics, there are no coincidences.
You’ll also come to understand how there is no other rational explanation comporting with sinking buildings and the universal laws of physics than Surfside being controlled demolition (CD). I’ve spent a couple of decades studying CD post-9/11 and it took all of about 3 seconds to sniff out this constructed event and begin piecing evidence together.”
Can I get a summary of what the real President said today?
The 2020 election was stolen, we won, and it has to be fixed before the 2022 election.
And that got the crowd on its feet, and my wife clapping and cheering.
And there are no diamonds in the mine (Leonard Cohen says so).
So the diamonds must be returned… 👍😁
BOOM. Too much TRUTH there!!!
Disco https://gab.com/emoji/271d.svg
@disco
1d
·
Schrödinger’s gun.
201 likes
18 comments
42 reposts
FIB. They earn their name every day.

Trump impersonator Shawn Farash: Trump talks Klondike bars. 😂
Love it!
Courtesy of Citizen Free Press, here is an interview where you can hear his real voice. He is a patriot who has founded a group that is fighting critical race theory, among other things.
EDIT: I didn’t realize the video would start where I left off. Go back to the beginning to see more.
That’s epic. And to top it off, the interviewer is imitating the interviewee…..
MAGA!!!!!!!!!!!!!!!
KAG!!!!!!!!!!!!!!!!!
KAG MAGA!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!!
(Help, Marica, I need a refill 🙂 )……
He should endorse “Eskimo Pie” bars/etc. That would REALLY light up the snowflakes…
Seems that killing off the Land O’ Lakes miss wasn’t enough for them. Now they’re after almost anything Alaska…
Klondikes are great, but messy. As are “It’s It” from the SF Bay Area. Such a great dessert, from a cruddy area…
Guy’s got real talent. What’s funny, too, is you look at him, and then hear the voice. Not at all what you’d expect.
Wonder if anyone’s trying to imitate Bye, Done or Heels-Up. OK, on the last one, there are plenty on Macarthur Blvd. in Oakland and San Pablo, no doubt. (Sad, too, that those for whom those streets/cities were named are stained by that association)…
(Those two streets had the first Doggie Diners, in better days…)…..