Okay you knuckledragging ChiComs trying to take us down…here’s a history lesson for you.
For millennia, you had to suffer from this:

Yep. Steppe Nomads. They laid waste to your country, burned, raped and pillaged (but not in that order–they’re smarter than you are) for century after century.
You know who figured out how to take them on and win? The Russians.
Not you, the Russians. And it took them less than two centuries. And Oh By The Way they were among the most backward cultures in Europe at the time.
You couldn’t invent an alphabet, you couldn’t take care of barbarians on horseback, and you think you can take this board down?
HAHAHAHAHAHAHA!!!! We’re laughing at you, you knuckledragging dehumanized communists…worshipers of a mass-murderer who killed sixty million people!
I mean, you still think Communism is a good idea even after having lived through it!
By my reckoning that makes you orders of magnitude more stupid than AOC, and that takes serious effort.
His Fraudulency
Joe Biteme, properly styled His Fraudulency, continues to infest the White House, and hopium is still being dispensed even as our military appears to have joined the political establishment in knuckling under to the fraud.
All realistic hope lies in the audits, and perhaps the Lindell lawsuit (that will depend on how honestly the system responds to the suit).
One can hope that all is not as it seems.
I’d love to feast on that crow.
Justice Must Be Done.
The prior election must be acknowledged as fraudulent, and steps must be taken to prosecute the fraudsters and restore integrity to the system.
Nothing else matters at this point. Talking about trying again in 2022 or 2024 is hopeless otherwise. Which is not to say one must never talk about this, but rather that one must account for this in ones planning; if fixing the fraud is not part of the plan, you have no plan.
Political Science In Summation
It’s really just a matter of people who can’t be happy unless they control others…versus those who want to be left alone. The oldest conflict within mankind. Government is necessary, but government attracts the assholes (a highly technical term for the control freaks).
(A comment I wrote last week that garnered some praise.)
Lawyer Appeasement Section
OK now for the fine print.
This is the WQTH Daily Thread. You know the drill. There’s no Poltical correctness, but civility is a requirement. There are Important Guidelines, here, with an addendum on 20191110.
We have a new board – called The U Tree – where people can take each other to the woodshed without fear of censorship or moderation.
And remember Wheatie’s Rules:
1. No food fights
2. No running with scissors.
3. If you bring snacks, bring enough for everyone.
4. Zeroth rule of gun safety: Don’t let the government get your guns.
5. Rule one of gun safety: The gun is always loaded.
5a. If you actually want the gun to be loaded, like because you’re checking out a bump in the night, then it’s empty.
6. Rule two of gun safety: Never point the gun at anything you’re not willing to destroy.
7. Rule three: Keep your finger off the trigger until ready to fire.
8. Rule the fourth: Be sure of your target and what is behind it.
(Hmm a few extras seem to have crept in.)
(Paper) Spot Prices
Last week:
Gold $1768.40
Silver $23.40
Platinum $1059.00
Palladium $2162.00
Rhodium $15,150.00
This week, 3PM Mountain Time, markets have closed for the weekend.
Gold $1793.00
Silver $24.40
Platinum $1047.00
Palladium $2104
Rhodium $15,250
Nice moves upward for gold and silver, but the platinum group metals are considerably mixed.
XXII Powering Stars
One of the things that was puzzling physicists and astronomers in the late 1800s and even into the early 1900s is how stars could continue to belt out such phenomenal amounts of energy every second, year in, year out for millions and even billions of years.
Our sun, for instance, has been pumping out 3.828 x 1026 watts, continuously, for billions of years. To be sure the current conclusion is that this number is actually increasing slowly so that in the past, say a billion years ago, it might have been ten percent less.
To put that into some sort of context, the best estimate we can make is that the entire human race uses 15 terawatts, that’s 1.5 x 1013 watts. The sun belts out ten trillion times as much power as we consume.
That power goes out in all directions from the sun, and only a tiny fraction of it hits the earth. By my calculations, the earth catches about 1/2 of one billionth of all of that energy, because that’s the fraction of the possible directions for light shining from the sun, that is covered by the disc of the earth as seen from the sun. (I may very well have dropped a decimal somewhere.) If that number is right, the Earth absorbs solar energy at a rate of 176,000 terawatts.
Where does this energy come from?
In the 1800s the only imaginable energy sources were combustion (like burning coal), the sun getting hotter as it shrank, and objects striking the sun. These were all unsatisfactory answers. A sun-sized pile of coal (never mind the oxygen needed to burn it) would have run out in a couple of thousand years [not long enough even to account for history since Caesar, much less all of recorded history]. The other two sources would last less than a million years at most (and there’s simply not enough junk in the solar system to hit the sun and supply the energy that way, or we ourselves would be getting bombarded by it).
We had every reason back then to believe the Earth is tens of millions of years old, though many argued it had to be much older. They were correct. We now have every reason to believe it’s roughly 4.5 billion years old. (Anyone disagreeing today is either simply ignorant of the evidence in favor of this statement and the massive preponderance of evidence in favor of earth being billions of years old (without putting a precise number on it), or is (in rare cases) quite aware of the evidence and is lying.)
So we need a way to power the Sun–and other stars–that can keep them going for billions of years.
And indeed Arthur Eddington–he is the astronomer who measured the deflection of starlight by the sun in 1919, which was strong evidence in favor of Einstein’s theory of General Relativity, which in turn had been published in 1915–well, Arthur Eddington suggested in 1920 that perhaps it was nuclear energy that powered the stars.
Nuclear energy had not been known in the 1800s, but it was now apparent that nuclear energy could supply roughly a million times as much energy as coal, per unit mass.
Fission of uranium would be plentiful, if only the sun were made of uranium, but honestly the biggest yield would come from the fusion of hydrogen into helium. If only the sun were made of hydrogen.
We know today that it is roughly 3/4 hydrogen, but that was not clear in 1920. We had spectroscopic evidence that the Sun contained certain ingredients (most of the elements are in the Sun at some concentration or another) but it wasn’t clear how much of anything there was. The proportions were a mystery. In fact the consensus at the time was that the Sun was pretty much made up of the same sorts of things, in the same proportion, as Earth. There was some reason to believe this, but we didn’t have all the facts.
Enter Cecilia Payne (later Ceclia Payne-Gaposhkin) (1900-1979).
Classifying Stars
But first, let’s go back a bit further to Annie Jump Cannon (1863-1941).
Annie Jump Cannon, along with Edward Pickering, was responsible for the current scheme by which stars are classified. She did most of the grunt work, he got most of the credit (though that is changing). This current scheme is known as the Harvard classification because, well, they were working at Harvard (pronounced HAH-vahd).
How do you classify stars? The same way you classify anything else: on the basis of what you can perceive about the objects. And with stars, that’s very confined. You have the star’s direction in the sky, its brightness, and its color. With telescopes, and some very specialized accessories, you can get the star’s spectrum, which is actually very useful since it can tell us what the star is made of, how fast it’s moving radially (towards or away from us–but this won’t include any sideways motion as seen from Earth), and even how fast it’s rotating in absolute terms. Today we can even use those spectra to detect planets orbiting those stars.
We truly didn’t have a science of astrophysics until we got a good close look at those spectra.
All of those things I mentioned as being able to be determined from spectra depend on absorption lines. These had first been noticed by Joseph von Fraunhofer (1787-1826) in the Sun’s spectrum (and so they are called “Fraunhofer lines”). They are dark bands visible in star spectra.

(Incidentally, astronomers who deal with visible light talk in wavelengths; radio astronomers talk in frequencies; and gamma ray astronomers talk in energies measured in mega electron volts…so when these guys get together at a conference it’s hard for them to relate to each other.)
Later on in the 1800s it was realized that these lines were actually characteristic of different elements in the Sun. Different atoms would either absorb or emit certain wavelengths of light under differing circumstances. For instance if you heat a sample in a Bunsen burner flame, the atoms in the sample will emit only certain frequencies of light, creating an emission spectrum; under other circumstances those atoms will absorb those same frequencies from “white” light, leaving dark bands in the spectrum.
It turns out the Fraunhofer lines were due to the Sun’s atmosphere absorbing some of the light emitted by the photosphere (which is the part of the sun we actually see if we are so foolish as to look directly at it).
And indeed helium was detected in the sun’s atmosphere by this means decades before it was discovered on earth. The name “helium” comes from Helios, the Greek god of the Sun who rode his very bright chariot across the sky every day.
When we turned telescopes to look at (other) stars, they too exhibited absorption lines, but they didn’t all exhibit the same absorption lines.
And those differences gave way to a variety of classification systems.
Annie Jump Cannon looked at hundreds of thousands of spectra and could classify them on sight, according to systems then in use, and eventually according to the system she refined in 1901-1912.
One thing that had been noticed, certainly by her and probably by others before her, was that there was a strong correlation between the color of a star, and which spectral lines were prominent.
And we already knew from studying blackbody radiation that the color of a star was determined by its temperature. Blue stars are hot, at least ten thousand Kelvins. White stars are hotter than our sun, which is a yellow-white and therefore has a temperature of 5,772K–or rather the other way around.
(And this is why you can’t buy a light bulb any more without selecting its “color temperature,” you’re picking the color of the light according to the temperature it simulates. A true tungsten light bulb filament actually did get as hot as its color temperature, and the light it emitted tended to be quite yellowish in color. And of course this is a “thing” in photography since the camera cannot adjust what it sees, but our eyes can, based on ambient color temperature.)
Annie Jump Cannon divided stars into classes with letter names (holdovers from older systems) O, B, A, F, G, K, M. Type O stars were the bluest (and hottest) of stars, down through G (like our sun) to M (reddish color).
Why these particular colors? A “white” star has the peak of its black body emission curve in the middle of the visible part of the spectrum, so the curve is about the same height at both the purple and red ends of the spectrum. It’s fairly uniform across that range, and we perceive that mixture as “white.” A cooler star has its peak somewhere below the red end of the spectrum so what we see contains more red light than yellow or blue light–so we see orange or red. And blue stars are so hot most of their radiation is ultraviolet; the visible light part has much more blue than red in it.
Cannon actually subdivided each of those letters into ten sub-types, numbered from 0-9 with zero being the hottest. Since these plots always put the hot end of the spectrum at the left (which is counterintuitive, but the habit formed, and once formed, stuck, and we are stuck with it today), you’d see a progression from O0 to O9, then B0 through B9, and so on.
Another useful thing to consider is how bright the star is, intrinsically. Not just how bright it looked, but how much light did it actually emit, compared to our sun? But in order to know that, we have to know two things: how bright it appeared to be here on Earth, and how far away it was. The first was easy, the second very hard, and in fact impossible to determine much of the time because the star was too far away for our measuring methods to work.
Nevertheless, when plotting luminosity against temperature, we saw some clear trends, and not entirely what was expected.
Most stars ran along a diagonal line that got named the “Main Sequence.” Other stars were of similar colors but much, much brighter intrinsically. And a few were obviously very hot, but also very dim. In particular, Sirius B was one of the latter (I described it in my second post on stars).
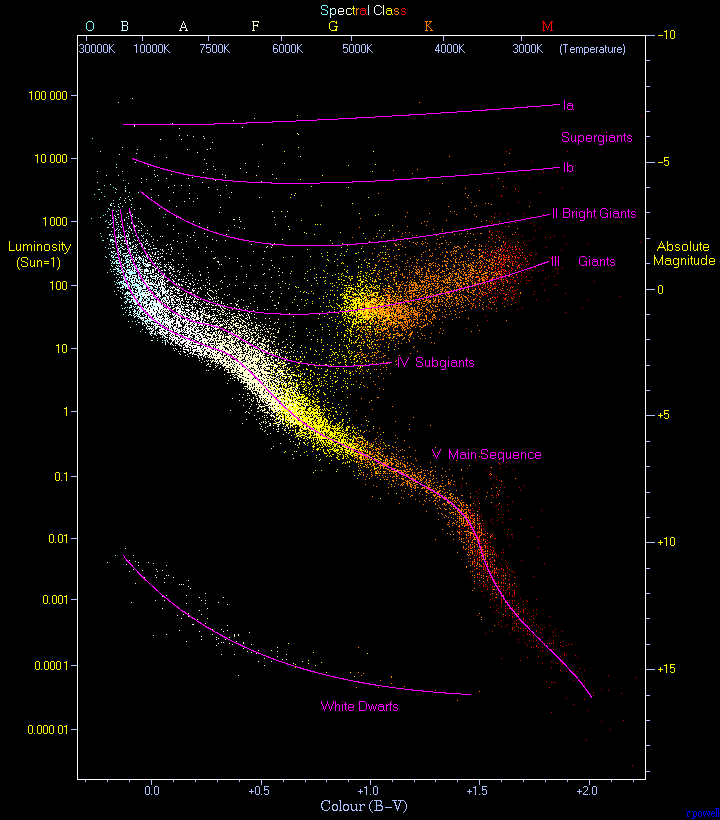
Once we had absolute luminosities in hand, something became apparent. You would expect a hotter star to be brighter, just as white hot coals in your fireplace are brighter than redder coals. And we could indeed calculate how bright they should be compared to cooler stars from the Stefan-Boltzmann equation. (An object twice as hot as another object emits sixteen times the energy as that other one does.)
When we looked at the luminosity of hotter stars, though, they were even brighter than they should have been. But there was a very simple answer to that. They were brighter than one would expect, because they were physically larger than the dimmer stars, just as a coal twice the size as another coal will emit twice as much light as the other, even at the same temperature.
So combine the two: Imagine a white hot coal twice the size of a red hot coal, and the white hot coal is now 32 times brighter than the red coal; more than can be accounted for just by its temperature or by its size.
Eventually we were even able to figure out the mass of these stars (especially when they were parts of binary star systems–we could determine the mass by watching how fast the stars orbited each other), and all of this was confirmed.
And all this largely from the data that Annie Jump Cannon meticulously collected, analyzed and cataloged.
Cecilia Payne Fills the Gas Tank
OK, now we are ready for Cecilia Payne-Gaposhkin.
She was at Harvard (yes, HAHvuhd again) in 1924, working on her doctoral thesis–she would go on to become the first female given a doctorate in astronomy by HAHvuhd…though it was actually Radcliffe, the associated womens’ college.
She took up an issue, that being what stars are made of.
That should have been pretty easy, right? We had their spectra with all of those wonderful absorption lines, after all. O stars had lots of helium in then. A stars had lots of iron and magnesium and silicon in them. And so on, down to M stars that had spectra of molecules in them like TiO2. That was how we divided them into their classes, after all!
But it turns out that many of these absorption lines weren’t from (say) ordinary iron or ordinary helium. They were from ionized iron, iron that had lost a couple of electrons. What difference does that make? The absorption lines (or emission lines under other circumstances) are caused by electrons absorbing (emitting) that precise wavelength of light in order to jump to a higher (lower) orbit.
When an atom is ionized, it has lost some electrons, and it hangs on to the remaining electrons more strongly, so it takes more energy for them to jump to higher orbits. This changes the absorption spectrum of that atom.
One way to knock those electrons off in the first place is to heat the atoms; that makes them move faster and when they slam into each other it could be hard enough to knock some electrons away. Thus the amount of ionized substances depends on their temperature.
This had first been realized by the Indian physicist Megnad Saha, but Cecilia Payne (she married Gaposhkin in 1934, so she was still Cecilia Payne in 1924) was the first to try to apply it to stars.
The prevailing theory at the time was that our Sun was made up of pretty much the same things as the Earth. All that calcium in the spectrum seemed to fit (there is a lot of calcium in the Earth’s rocks), as do other spectral lines from unionized (i.e., not ionized, rather than not a member of the UAW or Teamsters) elements. Meanwhile the hydrogen lines are very weak, especially compared to bigger stars.
Payne corrected for all those temperature effects, and came to the realization that the Sun…and other stars as well…were mostly hydrogen and helium. In fact the Sun is 74.9 percent hydrogen, 23.8 percent helium, and only 1.8 percent everything else.
This is so striking that astrophysicists today call everything that isn’t hydrogen and helium “metals” as a short hand. Since most of the elements in the periodic table are metals, that’s not a bad bit of scientific slang.
When Payne submitted her dissertation for review, it was criticized severely. She (unfortunately) backed down and wrote a paragraph into it dismissing her own data as spurious.
By 1929 her main critic, Henry Norris Russel, came to the same conclusion by a different method. He had the integrity to mention in his paper that Payne had got there first, but he still often gets the credit for discovering the stars are mostly hydrogen.
Mostly hydrogen.
So maybe (getting back, at last, to where we started) stars really did get their energy from fusing hydrogen. They certainly had the raw ingredient for it. The sun has the mass of 333,000 Earths, and three quarters of that is hydrogen. That is an absolute shitload of the stuff.
We knew from the binding energy curve how much energy is released (how much mass is converted to energy) per hydrogen atom, when four of them are brought together to form helium. We know the power output of the sun. Given those numbers it’s simple arithmetic to figure out how much the sun would have to “burn” and that amount is 620 million metric tons per second (a metric ton is a thousand kilograms, which on earth weighs roughly 2200 lbs).
4.26 million metric tons of this mass is converted to energy. That is a LOT of energy. And this happens every second. When you plug that into E=mc2, you get that number I quoted above, 3.838×1026 joules, and since that’s every second, that’s the number of Watts as well.
Divide that 620 million metric tons into the mass of the sun, and it’s clear that there’s enough fuel in the Sun to last billions of years–and indeed it has; we are about midway through that phase of the Sun’s life.
Tunneling Through Hurdles
But I am getting ahead of myself.
There was an additional hurdle the hydrogen fusion suggestion (not even really a hypothesis even now) had to clear before it could be taken seriously. And it was a difficulty Arthur Eddington had recognized clear back in 1920.
In order to fuse hydrogen into helium, you have to bring two protons together close enough that the strong nuclear force (which is so short range the protons have to be almost touching each other for it to take effect) overwhelms the electrical repulsion of the protons…which, if you’ll remember is a strong enough that we people could feel it (even out of those dinky little protons).
This can be done by making the protons move fast enough right toward each other. The repulsion causes them to slow down, stop, and reverse course…but if they’re moving so fast that they don’t stop until they get close enough, then they’ve climbed over the so-called “Coulomb barrier” (named after Coulomb, who first discovered the law of electrostatic forces) and can stick to each other.
How to make protons move fast? Heat them up. Temperature, after all, is simply a measure of the average kinetic energy of the atoms in a substance. Hotter temperatures mean higher speed of the atoms, particularly in a gas or superheated plasma.
At the kinds of temperatures we’re talking about, the electrons are stripped off the atoms, completely. You have bare protons zipping around in a swarm of loose electrons. (This is called a plasma, and it’s a fourth state of matter: solid, liquid, gas, plasma.)
The problem was, the interior of the Sun was believed to be at 17 million K, and even that temperature simply isn’t high enough.
But there actually is a way, and it’s supplied by quantum mechanics. Because of the Heisenberg uncertainty principle, the speed and position of particles isn’t precisely set at any given time, and if the speed isn’t set, the kinetic energy isn’t either. A particle with not enough energy (one would think) to jump over a potential “barrier” therefore gets to do so sometimes anyway. It’s much more likely if the particle is close to having enough, than if it is not.
This bit of quantum strangeness is called “quantum tunneling” and allows a particle which has no business jumping over a barrier to do so anyway, and physicists likened it to “tunneling” through the wall.
At the temperatures inside the sun, the probability of this happening is small, but not so small it never happens (as you see in the more familiar world where you fail to tunnel through blank walls unless you’re in a Road Runner cartoon).
If it were hotter inside the sun, the energy levels would be higher and the probability of tunneling through the barrier would be higher. But even as it is, it’s high enough that a tiny fraction of the protons do manage to “tunnel” through the barrier, and fusion can then happen.
But there is yet another hurdle, if you will pardon the expression.
When those two protons do glom onto each other, the resulting “diproton” is so unstable it simply falls apart right away.
But every once in a while, at the exact moment the diproton forms, one of the protons undergoes positive beta decay and becomes a neutron. In the process it releases a positron (anti-electron) and a neutrino. The positron finds an electron (they’re everywhere and literally anywhere in a plasma), they mutually annihilate and release a gamma ray–pure energy.
(The neutrino is a matter neutrino, not an antimatter antineutrino, because it counterbalances the antimatter positron, unlike in nuclear reactors here on earth where an antineutrino is created to counterbalance the electron produced by “regular” beta decay.)
A proton and a neutron will stick together. In fact this is hydrogen-2 or deuterium. Or rather, it’s a deuterium nucleus, known as a deuteron. (And yes, the joke is that the study of deuterons is known as deuteronomy.)
This beta decay at exactly the right time is a very rare event. And this is a good thing! Consider all those protons slam-dancing at 17 million degrees K for billions of years. If this event wasn’t rare, they’d be used up quickly rather than the supply lasting for billions of years. It’s not as if hydrogen is in a fuel tank until the sun is ready to burn it. No, it’s sitting on the fire, and has been sitting on the fire all along. It’s just that it’s burning very, very slowly.
The average survival time of a lone proton in the center of the sun is nine billion years. Yet it collides with a lot of protons at the temperature and pressure at the core of the Sun.
This was all outlined by Hans Bethe in 1939, at a Nobel lecture he gave.
The next step is for the deuteron to glom onto another proton. This takes, on average, about a second. The result is a helium-3 nucleus, two protons and one neutron.
After an average time of 400 years, two helium-3 nuclei will collide, and the result will be one helium-4 nucleus, and two freed-up protons, ready for another nine billion years on average of bachelorhood.
Six protons in, two protons out, plus one helium nucleus, plus gamma rays, plus two neutrinos. And a lot of energy. This is called the proton-proton chain.
Bethe also outlined another process, which involves four protons being added to carbon nuclei successively, with a couple of beta decays along the way, until an oxygen nucleus is created, which then spits out an alpha particle and reverts back to being the original carbon nucleus. This method is called the carbon-nitrogen-oxygen chain, or CNO chain, and it nets a helium-4 nucleus after consuming four protons.
It turns out that in stars more massive than the sun, this is the dominant mode. The temperatures are high enough to support it more readily than the proton-proton chain.
I’m now going to jump ahead to the modern understanding rather than going through the detailed history of how it was hashed out.
We know, now, that intergalactic gas consists of about three quarters hydrogen and one quarter helium. This gas is hot enough to radiate in X rays, but we can analyze the spectra.
There is only a trace of lithium in this gas, maybe a tiny bit of beryllium, and absolutely nothing else.
This is gas that was never part of a star. This is the original composition of the universe. [At least, as far as ordinary matter goes…but THAT is a future story.]
All of the “metals” we see today have to have come from somewhere. And indeed stars made them.
Because fusing hydrogen to helium isn’t the only way stars can make energy.
The Life of A Star
So let’s walk through this.
A cloud of (mostly) hydrogen gas…a very big cloud, trillions of miles across…contracts under its own gravity. As it contracts, it heats up (just like any other gas). But that’s no problem, gravity continues to crunch the cloud down.
The only thing that will stop the contraction is an equal but opposite pressure coming from the inside of the cloud. The pressure from the cloud depends on its size, a smaller cloud has less mass, less gravitation, and less pressure, so it will take less of this hypothetical internal pressure to get it to stop contracting.
I called it a hypothetical internal pressure, but it’s actually real. As the pressure and temperature at the center of the cloud go up, the hydrogen gas loses its electrons, the protons start slamming into each other, and at a temperature somewhat lower than at the center of the sun, some nuclear fusion begins to occur. If it’s a small cloud, that releases enough energy to heat the core up enough to stop the contraction. A bigger cloud will continue to contract, raising the temperature higher, to the point where more fusion happens, and then finally a balance is struck.
This balance is when the star becomes a well behaved, ordinary star, and it is now a “main sequence” star.
The main sequence is where all the hydrogen-burning stars go.
When I say “more fusion happens” I mean that more fusion happens for each ton of the star’s mass. In other words, it burns its fuel faster.
The bigger the star, the faster it burns its fuel, not just in absolute terms but in proportion to its mass. Bigger stars thus live much shorter lives than smaller ones.
It happens they are also a lot rarer than small stars.
One star in ten million is an O type star. These are 15 – 90 times as massive as the sun, but they are anywhere from 30,000 to a million times as luminous. If a star 90 times the size of the sun burns its fuel a million times as fast…well, you can see that it’s going to run out about 10000 times faster. Indeed they live only a few million years. Almost every O type star that has ever existed is long gone.
On the other end of the scale are the M type stars. About 75 percent of all stars are M type main sequence stars (at least, judging from the stars near the Sun). They are anywhere from 8% to 57% the mass of the sun, but even the biggest ones emit 7% of the light of the Sun. (The smallest emits 0.03% the light of the sun.) They’re cool and consequently reddish; they’re called “red dwarfs”.
(Red dwarfs may be 75 percent of all stars, but if you step outside at night and look up, you won’t see any red dwarfs. They’re simply too faint to be seen by the naked eye. The nearest star to us (other than the Sun, of course) is a red dwarf and cannot be seen without a telescope. This is not to say that you won’t see red stars…but those will be red giants. Which I’ll get to below.)
Red dwarf stars are long lived. It is estimated that one 16% the mass of the sun will last 2.5 trillion years. That’s an estimate, of course, because no one has seen one die. The universe isn’t even 1/100 th that age yet. Every red dwarf that has ever formed is still with us. (Even a “big” red dwarf 57% the mass of the sun should last at least 30 billion years, also older than the universe.)
OK, this is well and good. We have a pretty thorough description here of how hydrogen is made into helium. But not only is it still bottled up in a star…it’s also still not metals.
Remember that the material of the universe originally contained no metals, except maybe a smidge of lithium and beryllium. Yet we have these elements today…if not, you wouldn’t be reading this and I’d never have written it, because we would not exist.
Where did the metals come from? If they come from stars, how do they get out of the stars?
Well we need to follow this story further. (Kids, stop asking “are we there yet?” after every paragraph.)
What happens when a star runs out of hydrogen fuel?
It depends on the star. Those tiny red dwarfs, less than 25% the mass of the sun, are simply done. They shrink until the only thing holding them up is the mutual repulsion of the electrons. At this point they weigh maybe a million tons per cubic meter. They’re very hot, but that’s residual heat that slowly radiates away–no new energy is being created. Because they’re hot–hotter than they were as living stars, they are now known as white dwarfs, and are approximately the size of the Earth. Sirius B is a notable example of a white dwarf (I talked about it in one of my “stars” articles).
But wait.
Didn’t I say that no M stars had died yet? If so how do we have white dwarfs?
Because bigger stars also become white dwarfs. They take a more indirect route, but get there faster.
Stars bigger than 25 percent the mass of the sun follow a different path when they run out of hydrogen. They also begin to contract once again, but the temperatures in their interiors climb a lot higher.
They climb high enough, to 100 million K, that helium begins to fuse, three nuclei at a time, into carbon. (This is called the triple alpha process, because the three helium nuclei are three alpha particles.)
This happens at much higher temperatures. Under all of this heat the star expands. It gets downright bloated.
When the sun hits this phase it will probably bloat enough to swallow the earth.
That huge surface is actually rather cool for a star, it’s a hundred million miles (or more, for bigger stars) away from the raging inferno where carbon is being made.
The star is a giant, but it is red, hence the name “red giant.” It puts out a LOT more light than a red dwarf, in fact it puts out much more light than it did before. That pushes it up out of the “main sequence” and into the territory of the “giant” stars, to the top and right of that Hertzsprung-Russel diagram. These are giants in size, not mass…they’re no more massive than main sequence stars.
Helium converting to carbon produces less energy, kilogram for kilogram, than does hydrogen fusing to helium. Yet the giant star doing so must produce more energy to produce all that heat that makes it bloat.
In other words, all that helium “ash” from the hydrogen fusion, is going to itself be burned much faster than the hydrogen was. The star will get hot enough to do so, because it seeks balance.
Red giant phases don’t last very long compared to the time the star spent on the main sequence, happily burning their birthright of hydrogen.
If the star is the size of the sun, this is the end of the line. During this phase the star is a bit unstable, and may blow off some of its outer layers, producing a “planetary nebula” (called that because they used to be mistaken for planets in telescopes), and so a star like this might return some of the carbon it produced to space. But then the star dies, and it shrinks into a white dwarf. This white dwarf will contain carbon in it–a lot of carbon, but it does no good; it’s stuck in the white dwarf.
Of course, now when stars are formed from gas that already has metal in it, they return some of that too, but that’s not where that stuff came from. So where did it come from?
Big Stars are Metal Factories–complete with a shipping department.
Kids, we’re not there yet.
Stars considerably larger than the Sun, when they run out of helium in their cores, start to fuse the carbon. Again, this is at even higher temperatures. And again, this is a diminishing return. Less energy from the fusion, with a higher temperature having to be maintained, means this phase is short.
Also, around the core there is still some helium, and even the layers immediately outside the core are hot enough to fuse helium to helium, making more carbon, or helium to carbon making oxygen.
The star turns into a giant onion, each layer going inwards making bigger and bigger nuclei, and this (at last) is where all the good stuff forms, all the elements up to iron, in point of fact the elements that make up us.
Cooked in the centers of massive stars.
The only thing we need to close the loop, now, is to explain how all that stuff gets out of the stars. That would explain where all the metals that already exist in the Sun came from. Somehow, those metals were made in long-dead massive stars, then ended up in the cloud that contracted to form the Sun.
So here it is. The massive star eventually has a core of silicon, and there’s not enough other stuff in the core (though there is in layers surrounding the core) to keep going. The star heats up again, and commences to fuse the silicon into iron.
There are vast amounts of silicon in there, many times the weight of the earth.
The star rips through it in a day. Yes, a day.
It now has a core made of iron.
And now it cannot make energy any more. Because fusing iron consumes energy.
So the core collapses.
There’s a bit of a rebound effect. I say a bit. That rebound is actually one of the biggest explosions there is, a “core collapse supernova.”
The explosion is so bright, it outshines the other 100 billion (or so) stars in that galaxy, for a few weeks.
The last time one of these happened in our galaxy where we could see it was in the 1600s, just before the invention of the telescope, and the supernova was visible during the daytime.
Supermassive stars live fast and die hard. Bruce Willis has nothing on them.
That big explosion flings vast quantities of all the stuff the star has been brewing out into space, later to coalesce into new stars…and planets. And in the case of some star that blew itself to bits over four and a half billion years ago, the stuff eventually made us.
In the process, a lot of neutrons are created, and glom onto existing atoms, making heavier atoms, and until recently, it was believed that even gold, lead, and uranium were primarily produced this way. What an image: all the gold in your jewelry was once hurtling through space at a tenth the speed of light, blasted out of the guts of a star bigger than the sun.
What a pretty story. So pretty a lot of people like to say “we are made of star stuff.”
It is a fact that we owe our very existence to the death of big stars. Our bodies are made of atoms flung from their funeral pyres.
The Neutrinos Prove It
What’s the evidence?
There’s a lot of evidence, in fact, including the composition of nebulae (gas clouds), and especially the nebulae that have been blown out of supernovae. Stellar compositions are the evidence that started the whole thing, but shouldn’t be forgotten. A lot of “little things” all consistent with this framework.
But I want to focus on neutrinos.
In fact, this is why I undertook this whole damn series.
I wanted to talk about neutrinos. And connect them to stars…remember I talked about stars in the two science posts before this series. I was going to tie the smallest known particle of matter to the biggest discrete objects out there: stars (galaxies and galaxy clusters are bigger, but they don’t strike us as being objects but rather groups of objects).
But they are so ghostly, so non-reactive, that I would need to really justify their existence and tell the story of how they were discovered. And that entailed yet more background. I was going to just explain how they solved some problems with conservation laws…but then that meant I needed to explain those.
I thought maybe I’d write four parts. Then the doggone thing took on a life of its own. It ended up being twenty parts before I got to neutrinos. And another two before I connected them to stars.
So here I finally am.
One of the most important pieces of evidence that stars are, indeed, fusing hydrogen into helium, and so on as appropriate, is the neutrinos.
Those two protons coming together to make a deuteron, release a positron and a neutrino in the accompanying beta decay. This means that IF nuclear fusion powers stars, then ordinary stars are sources of neutrinos, and that most definitely includes our sun.
But also, a supernova, a dying star, gigantic numbers of neutrinos all at once in the fury of nuclear reactions going on all at once in the explosion–the reactions that give us all those heavy elements, elements heavier than iron.
There was a supernova of this kind 168,000 years ago in the Large Magellanic Cloud. The light reached us in 1987. A star known as Sanduleak -69 202 had just died.
It is estimated that this supernova released 1058 neutrinos. All at once.
I’ve thrown some big numbers at you over the course of this series, but that number is staggering. I’m not going to pretend to imagine how much that is.
Divide it by a trillion…it’s still 1046. Still a staggeringly huge number beyond our experience. And a hundred billion billion times as much as that ridiculously huge number I used for the power output of the sun.
If you had been a billion miles from that star when it blew up, here’s what you would have seen. The neutrinos hitting almost instantly–they were made in the core at the moment of the explosion but just zipped right on through everything. Then the light, actually delayed by all that matter being blasted out. Then the matter would have reached you as a blast wave to end all blast waves.
Except that you wouldn’t have seen the light or the matter, because you’d have been killed instantly by the neutrinos. There were so many of them that even at their ridiculously low likelihood of interacting with you rather than passing right on through, enough of them would have interacted with you to kill you instantly from the radiation.
It’s estimated that the light from the explosion–which, remember, outshines billions of stars–is one percent of the energy contained from the actual material blast. And that is one percent of the energy carried off by the neutrinos.
The light, bright enough to be seen from earth that far away (one of the first people to see the supernova was an astronomer at a major observatory in the Andes, outside taking a smoke break; he noticed that the Large Magellanic Cloud had a “new” star in it), was a sideshow.
So why are the neutrinos from the sun and exploding blue supergiants such a big deal?
Because we can detect neutrinos. And therefore, if we don’t see these neutrinos, something is wrong with our theory.
Large tanks of water, deep underground in mines so that nothing can get to them other than neutrinos, can be surrounded by flash detectors, which will register a hit every now and again. We can even tell, from the direction of motion of the products of the reaction, what direction the neutrinos came from. (And it’s a neutrino detector–we don’t have to wait for daytime or nighttime, it runs 24/7, and it doesn’t matter whether the sun or the supernova is “up” or not.
Twenty five neutrinos (a big signal for neutrinos) were detected from the supernova.
More importantly, these tanks have been detecting neutrinos from the Sun for years. That is a sure sign that nuclear fusion is happening there. And, they are of precisely the energy one would expect from the creation of deuterium from regular hydrogen.
There was just one hitch, with regard to the Sun’s neutrinos. We can calculate, from the power output of the sun, how many fusion reactions must be happening each second inside the sun, because we know how much energy each individual reaction releases. (It’s a geek’s story problem.) That gives us the number of neutrinos. We can figure out how many of them must be going through the detectors. And we know how likely it is that any given neutrino will be stopped inside the detector, letting us detect it. In other words, we know how many neutrinos should be detected coming from the sun, on average, during a given time period.
The number we detected was 1/3rd as much as it should be.
Ah, well, you solve one mystery (what makes the sun and other stars shine?) and you get presented with another mystery (where are the neutrinos?).
This is science moving forward.
And now, I think, I’m going to continue this series, even though it has reached the original planned conclusion.
I’m going to step from the neutrino, to something very, very big….much bigger than stars.
And then I’ll tell the story of that missing third…but that’s going to take a few installments.
Bonus Stuff
You put out more energy than the sun…sort of
The core of the Sun is at 17 million K, but what is its energy density?
How much energy is being generated in each cubic meter? The very high temperature has no bearing on this; some particular cubic meter of the sun stays at 17 million K because its surroundings are at that temperature. Heat leaks out of the core only where it meets the higher layers of the Sun. In fact it takes tens of thousands of years for a photon in the sun’s core to make it to the surface.
Energy density is how much energy is generated per…kilogram or cubic meter depending. Gasoline has a higher energy density than car batteries (even the ones for electric cars), for instance.
The energy density of the core of the sun turns out to be…wait for it…about 276.5 watts per cubic meter.
That is not a typo. Yes, we think of the core of the sun as a raging inferno, because there’s a lot of energy trapped in there. But as to how much new energy it generates every second, it’s actually quite sparse.
YOU produce 100-150 Watts just sitting on your butt reading this (more if you’re scratching your head really hard), because you have to keep your body temperature above room temperature. And your volume is a LOT less than a cubic meter. In other words, you generate more energy than a same-sized piece of the Sun’s core.
In fact a cubic meter of compost generates about the same amount of power as a cubic meter of sun’s core. (It just can’t do it for billions of years, so no, the sun isn’t a big compost heap.)
The reason the sun puts out so much power is that the core of the Sun is huge, roughly 200,000 miles across. That is a lot of cubic meters!!
So where did the gold come from?
I alluded to the belief that gold primarily came out of supernovas being an “until recently” sort of thing. So what’s the current theory? Core collapse supernovas leave behind either (for stars a couple of times more massive than the sun) a neutron star) or (very massive stars) a black hole.
What is a neutron star? It’s almost the ultimate collapse. It is what happens when even electron-to-electron repulsion can’t stop a star from collapsing, and the star doesn’t stop collapsing at white dwarf levels. Much of the star in a supernova gets blown away, but the remainder is usually much more massive than the sun. That remnant collapses. The electrons are forced into the nuclei, and combine with the protons to make neutrons. The entire remnant becomes one big ball of neutrons, with maybe a surface layer of white dwarf-style matter. The entire mass of the thing ends up in a ball perhaps ten miles across, weighing billions of tons per teaspoon.
When two of these neutron stars happen to collide–perhaps because two massive stars both went supernova and the neutron stars eventually lost all of their orbital energy to gravitational waves and then collided with each other–a lot of neutron debris splashes out there, decays and becomes heavy atoms, like gold. Entire earth-masses of gold are produced in this way and scattered across the cosmos. Now that we have observed neutron star collisions, we realize that most of the really heavy elements out there came from neutron star collisions, not from supernovae.
Obligatory PSAs and Reminders
China is Lower than Whale Shit
Remember Hong Kong!!!
中国是个混蛋 !!!
Zhōngguò shì gè hùndàn !!!
China is asshoe !!!
China is in the White House
Since Wednesday, January 20 at Noon EST, the bought-and-paid for His Fraudulency Joseph Biden has been in the White House. It’s as good as having China in the Oval Office.
Joe Biden is Asshoe
China is in the White House, because Joe Biden is in the White House, and Joe Biden is identically equal to China. China is Asshoe. Therefore, Joe Biden is Asshoe.
But of course the much more important thing to realize:
Joe Biden Didn’t Win
乔*拜登没赢 !!!
Qiáo Bài dēng méi yíng !!!
Joe Biden didn’t win !!!









Welcome to Steve’s Weekly Avalanche of Words! Bring your own coffee.
[just kidding]
ok

It’s too early in the morning for all of that.
It’s here all day.
Congrats Astros.
3rd WS in 5. Not too shabby.
And proving ya never did need to listen to Beltran and cheat, you dumb jackasses.
From last night’s game. Astros were up 3-2 in the series and 2-0 in game 6, but Boston was rallying in the 7th and had men on 3rd and 1st with only one out. The Astros pitcher had just thrown a 3-1 count with all three of those balls hitting dirt. He settled down, threw a strike for a 3-2 count, then this happened….
….sooooo clutch….this stolen base throwout is absolute perfection….just unreal accuracy and placement…
.
Honestly, Boston was desperate to have tried that against Maldonado…
.
https://www.youtube.com/watch?v=X-MyjsfyzWg
I’ve heard of a lot of “pitcher’s games” in baseball, but very few “catcher’s games”. This guy is impressive. Hitting the second base glove six inches off the bag and four inches off the ground is amazing.
In Little League, it was helpful that from home plate to Second Base was only about as far as we could throw in a semi-straight line.
So you just threw the ball as hard as you possibly could, and it would land right about perfect at the bag 👍😂
From my little league days, you threw it roughly at second base, and the second baseman was supposed to deal with it from there. This Maldonaldo guy fires it to where the second baseman’s glove needs to be when it gets there.
The coaching and drills involved have to be hilarious. “No, really, start going like you’re going to tag the guy out and watch out for the ball arriving.”
Two words for you:
Yadier Molina.
Yadi’s closing in on retirement age.
Hmm…..
This guy re-defines the word “fast.”
Just like we used to do it in Little League! 👍😁
LOL
🤸♀️ 🤸♀️ 🤸♀️ 🤸♀️ 🤸♀️
smiley2:
Another Wyeth painting, this time featuring both Rattler and Betsy taking a break from blueberry picking. I think the hat on the bed in the photo you shared belonged to Betsy.
“Distant Thunder” (1961) ~ Andrew Wyeth
Egg Tempera
Unless she’s just borrowing it to shade her eyes while he paints.
Nope. Betsy is wearing the same hat in the painting “Outpost” (1968). Also, I remember seeing a photograph of her wearing that hat.
thanks itswoot !
Wyeth was a master at egg tempera…working with very thin brushes…almost quills…he painted using a technique called cross-hatching and dry-brush.
meticulously detailed…look at those fir trees ! that grass !
layer by painstaking layer of color and texture..
keeping things mostly monochromatic, you will also notice, in almost all of his work, an area or two of color that sets is apart from the rest of the painting..
in this case, Besty’s blue blouse.
💝
I ran across a description by Wyeth of his watercolor dry brush technique that you might find of interest.
Scroll down:
https://arthur.io/art/andrew-wyeth/garret-room
What really makes the blue blouse “pop” is the yellow dog. The “triangle” of the very dark base of the trees, the blouse, and the dog is the visual “glue” of the painting. It moves your eyes around the painting, never leading you off of it. Masterful.
This is what you get from a genius, the very pinnacle of artistic talent. I paint, and I am envious.
Thin Ice
1969
tempera
Andrew Wyeth, Realism
can you see the suggestion of a face in those leaves ?
I see a lion.
love kitty 💟 🦁
Barely! Lower right.
I’m seeing the left side of a lion’s face in the entire right side of the painting.
Huh?!?
lion’s left eye
lion’s nose
lion’s chin
left eye very clearly visible
nose (nostril) almost looks human
white chin at bottom
Now I’m seeing a proboscis monkey at center right. I give up!
(I never was much interested in looking at ink blots)
Yes! I saw it before reading your description. It’s a bit Rorschach, but still…..
That came out very nicely. Kudos!
This is, BTW, for Steve. There has been so much going all which way that it’s hard to tell which comments go with what.
The Political Science Summary is perfection:
“What is government itself but the greatest of all reflections on human nature? If men were angels, no government would be necessary. If angels were to govern men, neither external nor internal controls on government would be necessary.”
James Madison, The Federalist Papers, No. 51, February 6, 1788
Steve’s summary is the best I have ever seen. It could easily fit in with Carlin’s quotes.
Unfortunately, I forgot about the other species of maggot that is drawn to government: The sort who doesn’t want power but rather loot.
While looking at some founding fathers’ quotes I came across this one, which is appropriate for the election crisis of 2020. We cannot “lose our country by a scrupulous adherence to written law” by not righting the wrongs of Nov. 3.
“A strict observance of the written laws is doubtless one of the high duties of a good citizen, but it is not the highest. The laws of necessity, of self preservation, of saving our country when in danger, are of a higher obligation…To lose our country by a scrupulous adherence to written law would be to lose the law itself, with life, liberty, property and all those who are enjoying them with us; thus absurdly sacrificing the ends to the means.”
Thomas Jefferson, letter to J.B. Colvin, September 20, 1780
“To lose our country by a scrupulous adherence to written law would be to lose the law itself, with life, liberty, property and all those who are enjoying them with us; thus absurdly sacrificing the ends to the means.”
_____________
Translation: The Constitution is NOT a suicide pact.
Not from the beginning, and not to this very day.
A country can exist without a Constitution.
A Constitution cannot exist without a country.
Lose your country, and the Constitution — whatever value it had — goes with it.
Jefferson is a man for our time.
What happens if you run two red dwarfs (dwarves?) together?
“What happens if you run two red dwarfs (dwarves?) together?”
___________
You get Siamese Faucis, of course. 👍
I’m expecting some bizarre responses to this….
I’m a waffle man, too!
Snow White gets to clean up a mess?
If the Seven Dwarfs were all red, she’d have been subject to the diktat of a politburo.
Lol.
Thank you, Steve, for another Joe-Biden-Didn’t-Win Saturday thread.
😊 👍
This one is epic!
Yes — 𝕁𝕠𝕖 𝔹𝕚𝕕𝕖𝕟 𝕕𝕚𝕕𝕟’𝕥 𝕨𝕚𝕟.
And F*ck Joe Biden and the donkey he rode in on!
amazing astrophysics post, Steve…can’t follow it all but I get the gist…

This is where I am. Look forward to these articles weekly. Can’t keep up with the incredible detail and thoughts. BUT, I get a bunch of amazing nuggets. Some laughs with the humor.
Bonus is the opening weekly verbal ass wuippn on the Chinee asshoes.
LOL … know what you mean. I let them sort of wash over me. I may not be able to repeat them, but they definitely open the mind in unexpected ways.
So, a little light discussion about firearms….
My dad grew up around firearms. His dad was part of a sheepherding group in western Colorado — he was the camp cook, and there was expected to be protein in the pot. There were some anecdotes regarding firearms from my dad’s childhood, including the time he shot a hole through the bedroom door while cleaning a 30-06.
Because my grandfather was frequently away, and went through five wives during his lifetime, my dad partially raised my uncle, the Marine. After they both went to California, my dad settled into a life with very little exposure to firearms, while my uncle built a family with a gun rack on the wall. My initial exposure to the discipline required to responsibly own guns was when my cousin and I were bouncing on the couch at his house, and he went right over the edge onto his face rather than touch the gun rack for support.
Over the years, the repeated ridiculous forays into gun-control crises annoyed me, and I finally selected a time when the Fiancee was out of town to go to a range and get NRA training in gun safety. I got the four laws, their practical application, courtesy at gun ranges, single-action/double action in wheelguns and semiautomatics, proper grips and sighting. It was a good class. I buddied-up with another person in the class (a guy from Sri Lanka who was doing contract work at Apple while his wife and family was in NYC….) and did weekly range time for several months. The range offered a plethora of rental guns….my (younger) buddy was pretty good with a bunch of ’em; the only way I could keep up was with hand-cannons like the Kimber Stainless II or CZ-75 ‘Tactical Sport’ — long sights and serious precision.
Unfortunately, if the fake news gun crises annoyed me then, it’s even worse now.
Everyone should take the course I did so that they can pick up a gun from a desk or the ground without endangering themselves and others, and so that they can competently carry it from place-to-place likewise. They should know the basics about how guns work and the four laws. And this should be drilled into their minds strongly enough that they won’t grab a gun rack when falling off a couch.
This whole “I won’t learn gun basics because I’ll be tainted” combined with the oogiedeboogie about guns causing crimes and spontaneously generating criminals to do them just makes me think these people are stupid.
Oh, and “if you find a loaded gun just laying there, how do you render it safe?”
Wheelgun — rotate the cylinder out, eject the cartridges.
Semi — drop the mag. Either cycle the one in the barrel out, or lock back the upper and shake it free. VISUALLY VERIFY THERE IS NOTHING IN THE BARREL.
Rifles — varies.
Oh, and just for fun — the Fiancee informed me that she just found out that her sister now has a gun. !!!!! Her friend was upgrading, and passed her previous gun on to her. !!!!!
Reminds me of when my aunt in TN had a home invasion and subsequently armed-up. Funny, the way that works.
I’d like it better with the “Died” date filled-in.
IKYM.
I’ll bet he hasn’t gotten any of the poison death shots.
I would bet my house he’s been on HCQ then Ivermectin from October, 2019.
From Paul B McCray, the inventor of the SARS Corona virus infected humanized transgenic mouse, who concluded in 2007 that human monoclonal antibodies prevented clinical disease in the mice:
https://pubmed.ncbi.nlm.nih.gov/34340553/
Nafamostat
build back better = first, destroy America…then… let the globalists build something else back…”better”.
👉 The Great Reset = build back better
“build back better” how about they build it first and if we like it we will come.
Thus far it seems to resemble a cage with lots of controls.
Bye,Done… Build Back Butthead…
This is so accurate it is scary.
got that right.
blech
barf
The last of the Mo-Vegans…
Recently read to reduce costs, some grocers are pulling “slow movers”. Looking forward to this vegan crap disappearing from stores.
No, that will never, ever move until it rots in situ.
Because, of course, the desire to cut costs must be tempered by social awareness (or something like that).
Oh that is hilarious!
from Judgment At Nuremberg
…it was FEAR…it swept over the land…
Burt Lancaster as Dr Ernst Janning, a Nazi judge during the Third Reich, testifying…
The Dems have been using that one too.
yes…I caught that, too.
All forms of government that skip over direct and continuous assent by the people become corrupt, and justify their errors with things like “progress” and “forward” and all that garbage.
BACKWARDS TO SANITY! BACK TO REALITY! BACK TO HUMANITY!
It WORKS under the BIDENAZIS.
“Progressive” and “forward” have been buzzwords for decades. I never did get why certain people used them. But that would make sense.
And I want to live in 1790. Sigh….
BUILD “BACK” BETTER!!!
🙂
Excellent find.
Maybe one day many of the current crop of Nazis will also come to realize that they are excrement.
Another great article Steve. Finding out why mankind doe’s not live very long in the scheme of things, we put out too much energy. Something I wish I had moar of….
Yeah, gotta hit the road again. Maybe I’ll explain later. Got to go 😬
Every step forward for life involves controlling more and more energy.
Which is why the environmentalists, who want us to use less, are to be fought tooth and nail.
They don’t just want us to be more efficient (which would be laudible), they want us to stop.
buttgig needs ETERNITY LEAVE
He will not LIKE his eternity … guaranteed … 💥☄️⚡️🔥💥☄️⚡️🔥 .. 🤨 ..
All I can think of when I see this is, those poor children.
The children will have all their creature comforts met. It appears that they will be loved as well. But they are being raised without a mother, on purpose. They have been placed in an abnormal environment whose ideology is destructive. I am so sorry for them. It would be better to have fewer creature comforts in a loving home with a mom and a dad, where foundational principles are taught.
100% agree.
EXCLUSIVE: International Journalist Shares on Italian Government’s Actions Setting Up Rome Protesters Using Same Tactics as US Capitol Police on Jan 6
.
Italy and the US are close. Unfortunately, they are so close (since the Obama Administration) that they are using the same tactics on their people standing up against their dictatorial dissent.
.
https://www.thegatewaypundit.com/2021/10/exclusive-international-journalist-shares-italian-governments-actions-setting-rome-protesters-using-tactics-us-capitol-police-jan-6/
FTA: Communists everywhere are making their moves now. We must stand together and stop them.
Some evil stuff here.
===
.
.Authors of ‘The transHuman Code’ and ‘Artificial Humanity‘ to host the introduction of
‘The Code to the Metaverse – Programming Our Future For Good’ at The Vatican, Rome
.
https://www.globenewswire.com/news-release/2021/10/20/2317655/0/en/WISeKey-to-introduce-The-Code-to-the-Metaverse-Programming-Our-Future-For-Good-at-the-forthcoming-Vatican-Collegio-Teutonico-meeting.html
TRANSUMANISM
the fusion of technology + the human body & mind & DNA.
think, for a moment, about the impact all of this is having and will have on the generation of children being born (if they make it) and coming of age during these times.
adopted & raised by trannies, homosexuals, lesbians, other perverts, other Lefties… along with the vast array of other psychos prowling the planet looking for whomever they can devour, twist, torment, abuse, experiment on, control…and ruin…and corrupt.
sorry
should be TRANSHUMANISM.
… 🥺🤚 .. 💔😢 .. 😔 ..
It’s like they want people to become & reproduce The Borg. Demonic!
Contrast w/ Tank in the Matrix “100% pure homegrown human, a genuine child of Zion!”
Many otherwise smart people have been lured into the snare of transhumanism by the doctrine of perfectability.
IOW, attempting that of which only God is capable.
For me it is comforting to know that the one one who is perfect is God.
Accepting once imperfections is freedom.
I feel sorry for the people who are willing to give up their freedom hand it over to a computer that is programmed by other imperfect human in the false assumption that it makes them as perfect as God. The sad think is with all the money they have they seem unhappy people and their souls are filled with fear of dying.
Isn’t Facebook RENAMING itself to something new? I heard that it’s tied to this “metaverse” stuff.
I would suggest a renaming to “Lugubrious”. Catchy and relevant to their true objectives.
Maybe they should rename it AssPortrait.
Seems more fitting.
LOL!!!
Yes it is alarming what they are tying to do get one spiritually hooked on their program. Believe Bannon spoke about that.
I am so glad I never allowed myself to be entrapped by Facebook. All sounded so nice since many family and friends are scattered around the country to stay in touch.
The whole Facebook was a scam for entrapment and hold people there.
I mean their is something like email where people can share pictures and stay in touch. Right now snale mail sounds really good 🙂
Catholic twitter has been on this like white on rice. Heresy doesn’t begin to say it.
They are making themselves gods. They want to create transhumans and robots in their own self-created world where humans and the normal way of life are not needed. They are perverting the created order of the universe and must be opposed. The pope is working with forces of evil.
It’s creepy as f***!
What is the metaverse? Meaning explained (nypost.com)
What Is the Metaverse? | NVIDIA Blog
Metaverse – Wikipedia
For some reason I can’t seem to save stuff to Pinterest for the last couple days. So I am shamelessly borrowing images & posting them on Gab for fun. So many of the memes shared here are worth more than 1000 words. Thanks for all the great finds & shares Q-Treepers. You guys Continuously Rock!!!
God Bless each & every one. Have a wonderful weekend!
Here’s something to consider
https://mynews.one/bulletin-evidence-that-lapalma-eruption-earthquakes-are-artificial-attack/


From a “really close” zoom in, per the article

“Whatever is going on on La Palma, may have begun naturally, but as of 11:45 PM eastern US time on Friday, October 22, 2021, it now clearly looks as though some type of man-made attack is taking place.
It LOOKS like somebody wants a lot of Americans dead.”
“Back in the 1990’s several scientists closely studied and even went inside the then-dormant volcano and found that the entire southwest flank had become unstable and could slide into the Atlantic Ocean. That unstable land mass is the size of the island of Manhattan, NYC, and would generate a tsunami wave.
Computer models showed the tsunami would begin hitting the US east coast within 7 to 8 hours, and would smash almost all the major cities, with a wall of water 30 meters to fifty meters high. That would mean a wave 100-164 FEET tall, lasting 5 to 45 minutes as it flooded 12-15 miles inland.”
That’s weird. Maybe it’s some sort of algorithm that causes the quakes to show as a grid?
No clue! Hope someone w/ a solid science background weighs in (my degree doesn’t count here)
If positions are reported to the nearest (say) 100 m, plotting a bunch of data will cause it to land on a grid.
Yeah, seems right.
So if you go to the website and zoom in on clusters of quakes in other places, although they are not as dense as the La Palma ones, they do appear to be shown in a sort of grid pattern. I think this is an algorithm issue.
Here’s the site:
https://www.emsc-csem.org/Earthquake/Map/gmap.php
Interesting. Good catch. Maybe it is a function of the sensing equipment? Maybe it’s like a GPS grid overlaying the info? No clue here, but definitely curious…
It’s a function of roundoff.
If positions are reported to the nearest…oh, I dunno, maybe 100 meters, all your numbers are going to end in 00. That will look like clumping at round numbers.
That works in 2D as well.
Gotta be something like that GPS thing, I’d think.
See Steve’s common sense takes nearby 🙂
“…lasting 5 to 45 minutes…”
no worries ’til they say “… 40 days and 40 nights…” !
😉
Nice!
Should raise tons of questions if true.
If true would explain much also. Example why Biden is so hell bent on getting everyone vaccinated. Cause Mr Global says so or else?
Meanwhile the only thing that could create a grid like that would be satellites.
If China was doing such, you’d think we would know.
However if this were true there are enough people across the world that would have access to what is being reported and I’d suspect there’d be many multiple reports of this happening long ago.
That said though, using their map, zoom does provide for what is said (grid pattern). Also other clusters heavily in the red do not produce a grid pattern. So what’s up? Elaborate hoax if it is one.
Wonders if there is another mapping program we can look at?
Great points. Another Just the facts Ma’am situation me thinks!
Let’s first be sure that what we are seeing isn’t just an artifact of lack of precision in measurement.
Let me illustrate by analogy…your bathroom scale might read off to the fifth of a pound. That doesn’t mean you lose or gain weight only in 1/5th pound increments, though it will look like it if you keep a daily log of your weight.
If positions are being reported to the nearest 100 meters (or some other number) when plotted they will form a grid regardless of the truth of the matter.
Thanks for this common-sense insight. Steve 🙂
As mentioned, other clusters on the world map that are of the same size, for which there are two others, do not demonstrate the grid pattern. Only the one at the Canary Islands does it. Maybe later I’ll go look for another map. Maybe go back and look at past data for volcanos too as what you say may be normal for laying out detection devices for volcanic areas.
Real Men lay down their lives for others in so many ways revealing God’s image!
The Lover of Our Souls cries out to Each of Us with Eternal Love!
To be the Beloved Bride & (part of the) Body of Christ, Eternal Mystery–Dive In!!!
More details and mystification on how a live round was on a movie set. It sounds like safety went out the window. Who made THAT call?
In reading about “live rounds” on a movie set, I am getting some contradictory info.
Some articles say that “live” on a set means “loaded with blanks.”
Some say that it means an actual round.
Something about this is SO fishy. I wonder if we will ever really know the truth. It is Hollyweird, where they spin tales for a living.
I agree. There is something VERY fishy about it. Very. Another Baldwin brother was questioning the account. For the safety shield to not be in front of the directors is a BIG red flag.
Agreed.
This is legit. It’s him. First photo from NY post:
From LinkedIn:
Formerly Kirkland & Ellis, and Skaden Arps…
MeThinks the entire narrative is FUBAR.
Movies ARE FANTASY. GUNS ARE ALWAYS REAL.
Until we have the written procedures for weapons AND ammunition on “Rust” movie set, we ARE simply tuning into other folks guesstimate AND we pontificate.
IMO.
“Live” should include EVERY round with any amount of explosive.(Primers are also explosive)
“Cold” gun is, IMO, STOOPPID. MEANINGLESS in the safety realm.
“Unloaded”, NO round, including blanks, dummy rounds in the weapon. Applies to a magazine attached to the weapon.. THIS makes sense.
Asshoe actors / actresses MUST THEMSELVES VERIFY Blank rounds OR dummy rounds loaded. AND verify weapon is UNLOADED – SAFE. WHOEVER handles a weapon capable of firing a round IS LIABLE for their actions, including weapon discharge.
Was taught early on NEVER, NEVER, NEVER be at the business end of a weapon. NEVER, NEVER, NEVER point a weapon at someone unless it is self defense and situation requires engaging the threat – fire a round.
Now factor in movie making FANTASY LAND. Weapons ADD DANGER TO LIFE, property…
^^^ THIS ALONE DICTATES the most stringent safety processes are VIGILANTLY applied with NO DEVIATION in movie fantasy land.
—-
Absolutely tragic this gal was murdered AND some dude was wounded.
But, I am happy for Baldwin’s misery. Baldwin chose to point the weapon, point the weapon in an unsafe direction AND pull the trigger. STOOPID on STEROIDS. In a “Just” world, Baldwin would be held accountable. Along WITH whoever loaded that round. AND, everyone that contributed to unsafe weapon handling.
—-
Sadly the “coincidences” with the attorney widower as suspicious.
AND, SADLY, TWO TIER Justice will be on full display soon enough.
Yeah, I hope he pays the price, but I doubt he will.
These are the two take-aways I have been hearing from people: justice will not be done, Baldwin will be protected; and another body to add to the Clinton count.
I think it’s just interchangeable terminology. It’s like people referring to revolvers as pistols. “Pistol” can mean any handgun, but it also specifically refers to semi-automatic handguns vs. revolvers.
A “cold” gun has nothing in it, not live rounds or blanks. The assistant director said the gun handed to Baldwin was cold.
I don’t know what a gun with blanks in it is called, but I suspect “hot,” because there should be no scenario on a movie set where live rounds have to be dealt with. So I’m speculating that a gun is either cold, with nothing in it, or hot, with blanks in it. But I don’t know for sure.
Good thoughts.
“The shot heard round the world.” ???
Maybe, but this would be really out there for that.
seen on Gab, & claimed to be possibly only 10% of the actual number (per that study couldn’t it even be as little as One Percent?)…

This being reported by a DS wire service is interesting.
https://www.reuters.com/article/us-health-china-bgi-dna-idUSKCN2ED1A6
Special Report: China’s gene giant harvests data from millions of womenBy Kirsty Needham, Clare Baldwin
Don’t trust China for anything. Period.
And here’s another wrinkle —
https://vgacquisition.com
http://www.weforum.org/agenda/authors/richard-branson
The business acquisition / shell company of Richard Branson, VG Acquisition Corp., completed its purchase of 23 And Me, the personal DNA testing firm, in July, 2021.
Richard Branson is an “Agenda Contributor” to the WEF.
Boy, that is a trap if there ever was one. Media blitz, daily TV ads, people enamored with finding out some iota of relation to any so called minority or other culture.
Underhanded Down Under…
@BeachMilk
1h
·
TRANSLATION: “We acknowledge that the COVID vaccine is killing and maiming thousands of people, but we won’t enter this information in our database until we investigate and find a good reason not to”.
https://gab.com/BeachMilk/posts/107150896161422678
Another Oz outrage

@BeachMilk
3h
·
·
Edited
Tee-shirt manufacturers in Australia are ‘testing the waters’ to see if a child-sacrifice theme will be a Christmas seller.
https://gab.com/BeachMilk/posts/107150842958985878
Trump takedown effect?
@Artraven
1h
·
JURNO BEFORE TRUMP AND AFTER


THIS IS TOP TIER OVER AT WAPO
MICHAEL SOMETHING OR OTHER…
https://gab.com/Artraven/posts/107150881608372404
LOL!
The opposite of a before & after coming to Jesus countenance (not saying Trump is Jesus, but Believers likely know what I mean) 🙂
THIS explains a LOT. I never really liked the supernova explanation – it seemed like hand-waving to “a big messy explosion had this fine result.
But the idea of what can be regarded as “pure neutronium” being belted out, losing gravitational compression, and the decomposing chunks then undergoing massive decay toward iron – oh, that is highly explanatory – including a lot of “highly theoretical” stuff.
In another matter, only tangentially related, what does the “goldilocks zone” look like for a red dwarf? If there is one, then that has implications, and also explains a few things.
There is something called an R process, where nuclei are force fed a bunch of neutrons all at once during a supernova. I’m sure that does amount for a lot of heavier-than-iron nuclei, but you’d expect that number to trend downward the higher up you go.
Which it largely does. It wasn’t that ridiculous a thought, and though I haven’t read the literature by any means I am sure it is still believed that SOME heavies do get made in supernovas.
Apparently they’re still considered the major source of elements up through rubidium (Z=37), and about half of all isotopes heavier than iron can be accounted primarily this way.
The R process results in a lot of nuclei with way too many neutrons in them and ordinary beta decay will push them up the periodic table.
As for the goldilocks zone, I couldn’t say other than the obvious: that zone will be much, much closer to the star. I’ve seen claims that the “year” of such a planet would be a hundred days or less, much less the smaller the star is.
I know decades ago the conventional wisdom was that such a planet would get blasted by solar flares a LOT, and they’d be proportionately larger than ours are. Imagine the tops of the oceans being raised tens of degrees, perhaps even to boiling by a flare every few years and you can see that’d not be a friendly place for life to form. (Burn the land and boil the sea…) A lot of conventional wisdom about planetary systems [formed back when we only knew of one, this one], however, has been blasted into oblivion now that we have observed thousands of them. I doubt, however, that would affect this particular conclusion about the flares.
Dr. Zandre Botha, PhD on the Stew Peters show w/ revelations of post “vax” blood issues. Sorry if this has been posted before…
blob:https://www.brighteon.com/90244ef6-6615-48ef-b539-0384305bf357
@Minataur
1h
·
NEVER BEFORE SEEN: BLOOD DOCTOR REVEALS HORRIFIC FINDINGS AFTER EXAMINING VIALS – HEADS UP! https://www.brighteon.com/0fb5d26d-304b-4aae-9f22-cd2c41f39a7d
NEVER BEFORE SEEN: BLOOD DOCTOR REVEALS HORRIFIC FINDINGS AFTER EXAMINING VIALS – HEADS UP! (MIRRORED)NEVER BEFORE SEEN: BLOOD DOCTOR REVEALS HORRIFIC FINDINGS AFTER EXAMINING VIALS – HEADS UP! (MIRRORED)
Mirrored from Bitchute channel Jim_Crenshaw at:-
EXCLUSIVE!…www.brighteon.com
View Link Feed
https://gab.com/Minataur/posts/107150877954639773
Here’s her website & it lists Covid & post-injection protocols
https://www.drzandrebotha.com/
Some structures she observed in post-shot blood she described as “self-assembling” & Not biological. She found some similar structures in a shot vial (J & J?) that has chain of custody docs & was provided by a lawyer, I believe…She provides her observations but not what she’s seeing might be, because she doesn’t know & hopes other experts will weigh in…
I Really hope Wolf sees this video & weighs in. His insight will be invaluable here, imo…
At 16 minutes she starts describing symptoms & she & a surgeon similarly had after being exposed to “vax” material or “vax’d” patients…
Apparently this show was from Oct. 4
This info is from his site:
https://www.redvoicemedia.com/2021/10/never-before-seen-blood-doctor-reveals-horrific-findings-after-examining-vials/
Dr. Botha came forward, today, on “The Stew Peters Show”
*Note: Dr. Botha mentioned treatment for vaxxed while speaking to Stew, here are her recommendations:
Treatment for vaccine injury
NAC
Ivermectin
Food grade hydrogen peroxide
Molecular hydrogen therapy
Glutathione
Ozone therapy
Zeolite
Important to support the heart with potassium
Selenium is also good at detoxification of cells
Chelation therapy
It was a J&J vial that Dr. Botha analyzed. Her findings confirm the “lighting up” of the “dot-like” structures that Dr. Madej found in the J&J vial that she analyzed. Also the black “shaped” structures.
Her video of the black circles & the silver lines connecting on 6 equidistant points around the circles are very intriguing too.
TWC “we’re all gonna die!!!”, as usual.
Anthony Watts at WUWT shreds them all the time, as do Joe Bastardi and others. Atmospheric rivers are a normal part of the hydrologic cycle(s). How else would all that water get from the ocean/ground into the atmosphere… And at the same time, they don’t see that the biggest “greenhouse gas” IS water vapor… Yet another scam, indeed one they used to practice and “learn” for the COVID scam….
TWC is worse than the Enquirer, CNN, MSDNC, ABC… Unwatchable. TWC weather SKEER reporting methods have been adopted by “local” weather news, everywhere. Also, unwatchable.
Weather for this fool is online forecasts. NO video. NONE. Scan the daily forecast. At times drill down to hourly AND look into live weather radar.
ALL of the video weather is simply forecasting DOOM AND RECORD BREAKING LIES.
^^^ Can be likened to Faux Weather mimics Faux News horse shit. ^^^
According to Babylon Bee, you might be looking at Oceanfront property if you live, erm, east of Kalifornistan 🙂
https://babylonbee.com/news/new-computer-models-showing-california-under-water-causes-huge-spike-in-support-for-climate-change

^^^ Truly great news! Truly miss living near the ocean. 🙂
TOC Ready Room 23 Oct 2021: A ‘domestic terrorism’ retraction, and an emerging plan in Burma – The Optimistic Conservative (wordpress.com)
Let’s Go Brandon!
Oh, I’ll do it, too.
!!!
No, don’t do that.
If the IRS sees a bunch of $601 transactions, they’ll learn to simply ignore them.
Pick some random number between $600 and $1000. Change that number every time you do this. (Obviously if your bank balance isn’t that high, you’ll need to adjust the range.) But if we really want to jam the system up we should each use a different number every single time.
Right! Just not $666!
Done properly that could be an accusation leveled at them…
True…
Oh! That’s rich….LOL!
For the Satanist’s Witchmas tree

UK info found on Gab

Greenwald!

Hopium on Gab?
@ToniBDufour
2h
·
·
Edited
Prepare for a week that will change history.
-DJT
https://gab.com/ToniBDufour/posts/107150795198597433
Source of this claim?
Someone shares Scott’s sense of humor 😉
@Artraven
2h
·
OK TRUMP,
JUMP OUT OF THE BUSHES
TELL US ALL WAS A SILLY JOKE AND YOU JUST
WANTED THE SNOWFLAKES TO SEE HOW BAD THE DEMS AND RINOS
ARE.
ANY MINUTE NOW…..
https://gab.com/Artraven/posts/107150859222523830
It isn’t funny when people actually think that’s what’s going to happen.
They’re going to be thinking that all the way to the gas chambers.
I really hope & pray it doesn’t come to something like that…but it’s not looking too good.
But keep in mind DP’s find of the Live Feed in Italy showing an empty public square, while the person showing that propaganda on their phone was standing in the midst of a massive crowd of protestors in that same square.
What we see &/or hear isn’t necessarily what is Actually going on. The FJB chants seem to indicate things are Not at all as the media mouthpieces & usurpers of our Republic would have us believe…
Remember that D-Day diversion w/ inflatables etc…history repeats!
Lock Him Up in [Market-Ticker-Nad]
“vax” horrors unabated
@TerryBucci
2h
·
VAERS data released Friday by the CDC included a total of 818,044 reports of adverse events from all age groups following COVID vaccines, including 17,128 deaths and 122,833 serious injuries between Dec. 14, 2020, and Oct. 15, 2021.
https://childrenshealthdefense.org/defender/vaers-cdc-deaths-injuries-covid-vaccine/
 17,000+ Deaths Reported After COVID Vaccines, Including New Report of 12-Year-Old Who Died After Pfizer VaccineVAERS data released Friday by the CDC included a total of 818,044 reports of adverse events from all age groups following COVID vaccines, including 17,128 deaths…Children’s Health Defense
17,000+ Deaths Reported After COVID Vaccines, Including New Report of 12-Year-Old Who Died After Pfizer VaccineVAERS data released Friday by the CDC included a total of 818,044 reports of adverse events from all age groups following COVID vaccines, including 17,128 deaths…Children’s Health Defense
View Link Feed
https://gab.com/TerryBucci/posts/107150819875655441
https://childrenshealthdefense.org/defender/vaers-cdc-deaths-injuries-covid-vaccine/

PA power!
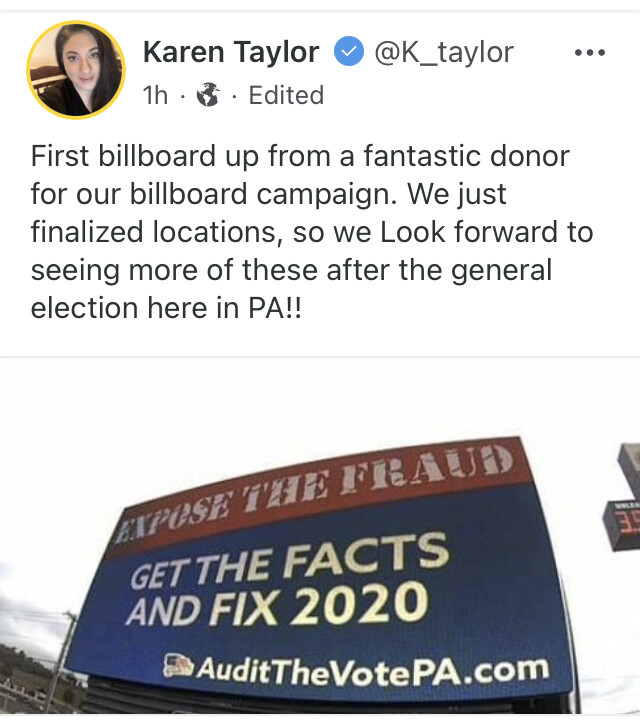
Is WI ramping up to an audit too?
Absolutely outrageous.
Yes!
Yep. That’s been working since we were kids.
Heart-breaking
Almost can’t believe CDC published this – unless the people down low in the organization are getting scared, and are trying to save themselves from the fall of Walensky, Fauci, Woodcock and Biden.
Truth-tellers Must Arise, Everywhere!!!
Even my fairly lib son-in-law is at least somewhat “vax” hesitant because he’s heard of the heart contraindications for young men…& he does love to work out!
If they are suddenly telling the truth, something is in the wind.
Not sure of the date when that was actually published. It could be like that “quiet” revision of “covid death” numbers down by like 95% Twice…
Dr. Simone Gold
@DrSimoneGold
1d
·
We are seeing the failures of government everywhere, from healthcare to bare shelves.
We cannot rely on bureaucrats to tell us what’s best for ourselves and our families.
Push back against the mandates.
Push back against blocking early treatment.
It’s OUR choice to make!
1,483 likes
84 comments
586 reposts
Flank steak this morning was $16.99 a pound. Roasts are OUTRAGEOUS prices. I had to go to two stores to find the cut I was looking for because at the first one, both the meat and deli counters were closed. I realize eastern mid-St. Louis County doesn’t really wake up before nine on weekend mornings, but I’ve NEVER seen that at that store.
Um?
@NeonRevolt
1h
·
They say the “Dutch Oven” is a key to a happy and lifelong marriage.
manoffiji
@manoffiji
1h
·
@NeonRevolt In college we were hanging out with a group of people watching a movie and my wife-to-be was in the room and I liked her a lot but I figured if she didn’t think farting was funny it wouldn’t be worth my time. So I ripped a huge fart and she thought it was funny. We’ve been happily married 18 years now.
https://gab.com/NeonRevolt/posts/107151709838557016
Makes me think of a classmate I had in elementary school. He could break wind like artillery fire, and could burp most of the way through the pledge of allegiance….
One time he did both at the same time…. We were expecting him to disappear 😆
superhuman powers, or perhaps subhuman!
My daughter said she once belched, farted, sneezed, & hiccupped all at the same time & she thought it was one of the best experiences of her life–yikes!
Throne of Blood (clots)

Banksters being questioned
[video src="https://media.gab.com/system/media_attachments/files/088/517/776/original/7f44c35b70feef8e.mp4" /]
McAfee (or McAtee)??? !!!

Is anyone shocked?
Bueller?
Bueller?
LOL
IVM crushing the curves!
@K2xxSteve
5h
·
This is a fascinating article. Despite similar overall vaccination rates between the U.S., the U.K., and Australia, look at the case numbers and profiles, and then look at what else is going on with Ivermectin.
https://www.thedesertreview.com/opinion/columnists/freedom-and-ivermectin-prevail/article_05d8061e-3035-11ec-bfe8-2b94d8993caf.html
The Desert Review is claiming that the exploding number of prescriptions for Ivermectin in the U.S. is responsible for slowly eradicating Delta, despite rampant censorship and threats on doctor’s medical licenses. Meanwhile Ivermectin is virtually banned and/or heavily restricted in the U.K. as their numbers of cases continues to linger, and then Ivermectin is banned completely in Australia as their number of cases has exploded despite vaccine mandates and tyrannical lockdowns.
It’s fascinating to see a sea change in Ivermectin use in the United States, and for it to now be considered in “widespread use”. Tons and tons of credit should go to Drs. Pierre Kory and Paul Merik at the FLCCC, and to the likes of Joe Rogan for really pushing this hard and skewering Sanjay Gupta recently as well. Pretty amazing to see. #Ivermectin #FLCCC #MedicalGab
https://gab.com/K2xxSteve/posts/107150837656055461
https://www.thedesertreview.com/opinion/columnists/freedom-and-ivermectin-prevail/article_05d8061e-3035-11ec-bfe8-2b94d8993caf.html
Add in users of horse paste, undocumented but not insubstantial I bet.
No doubt–Yum!
Constructing a narrative 😉

Kyle Rittenhouse day in court
@diamactive2001
2d
·
Andy Ngô :
A use of force expert gave testimony at Kyle Rittenhouse’s pretrial hearing outlining how the teen’s decision to shoot was reasonable given the circumstances. Two of the men tried to take his gun after pursuing him & the third rushed at him w/a pistol.
https://apnews.com/article/wisconsin-kenosha-homicide-shootings-police-brutality-8d68c528a53bddfa2ced544be1438e55
 Force expert: Rittenhouse decisions to shoot were reasonableKENOSHA, Wis. (AP) — An Illinois man who shot three people during a protest over police brutality in Wisconsin last year was justified because the men confronted…apnews.com
Force expert: Rittenhouse decisions to shoot were reasonableKENOSHA, Wis. (AP) — An Illinois man who shot three people during a protest over police brutality in Wisconsin last year was justified because the men confronted…apnews.com
View Link Feed
https://gab.com/diamactive2001/posts/107138815414267873
https://apnews.com/article/wisconsin-kenosha-homicide-shootings-police-brutality-8d68c528a53bddfa2ced544be1438e55
Show trial. Should never have been charged.
Absolutely. A True American Hero!!!
“Stop the world I wanna get off, it’s Too Weird for me!” Randy Stonehill, Great Big Stupid World

Seen on Gab…sauce?

Replying to, “…sauce?”
NONE that I see.
MeThinks Joe, Hoe, Nanzi and Chuckie are bounced OUT, it is all meaningless blather.
While hope remains, optimism is this regard is seriously lacking.
Hell, them dumb bastards in DC can’t even trash BiteMe armed with the 25th OR even push the impeachment of Joe AND Hoe.
There is no mechanism to reverse a certified election that does not include an approval by the Supreme Court. Let alone a Democrat run Congress.
Yup. Uncharted reality.
Courts AND Legislatures could get creative. But they WON’T. They are all corrupt.
^^^ MIRRORS WHY WE ARE IN THIS FU SITUATION.
No doubt!
I used to read WND, worldnetdaily, A Lot during the Obama usurpation & especially enjoyed much of their reporting on the Natural Born Citizenship clause. They cited some example of a Governor in Minnesota, iirc, who was thrown out after being elected, sworn in, & in office for some weeks or months. This was back in the 1800’s. Presumably his election was “certified” before being overturned…
Scofflaw SCOTUS! To the rescue! I mean recusal!
Wolfie,
My transhumanism post is on the Utree @
https://utree.home.blog/2021/10/22/klaus-schwab-in-all-his-glory/
In several different comments.
Thank you!
OK – thanks! Will pull it out later this evening.
Crap!
That worse than any photo or comment I’ve posted!
Not really sure that’s KlauSS, but it’s nasty anyway.
& that’s really saying something 😉 you have to try harder!!!
It’s up now!
https://www.theqtree.com/2021/10/23/transhumanism-the-great-new-reset-is-the-same-old-serfdom/
If they fail us, we might have to come up with an appropriate mechanism to save the republic.
“A strict observance of the written laws is doubtless one of the high duties of a good citizen, but it is not the highest. The laws of necessity, of self preservation, of saving our country when in danger, are of a higher obligation. … To lose our country by a scrupulous adherence to written law would be to lose the law itself, with life, liberty, property and all those who are enjoying them with us; thus absurdly sacrificing the ends to the means.”
Thomas Jefferson to John B. Colvin, September 20, 1810
Doubt there is enough of us who will step up. Plenty that talk the talk, but ultimately as a population we have gone soft.
Many have, that’s true.
LOL Sounds like you’re about to re-up for another tour of Whoop A–!!!
From the old days when Real Men, not girly men, wore the uniform. Bring back honor…AND testosterone!
Keshel has all the stats, and has for months. Everyone who’s even half-way tuned into this horror knows that Trump (and a lot of other people) won. What he doesn’t have is the mystery path to point B.
Yes! Congress is about as illegit as Joe & Hoe!
30% price change in a day???
@NaturalNews
13h
·
The #price of shipping LNG rose nearly 30% between Monday and Tuesday #shippingrates #supplychain
https://www.naturalnews.com/2021-10-21-shipping-liquefied-natural-gas-rate-doubled.html
 Cost of shipping liquefied natural gas has more than doubled in a monthThe natural gas and energy shortages sweeping across Europe, Asia and the United States have now affected the cost of shipping liquefied natural gas (LNG).
Cost of shipping liquefied natural gas has more than doubled in a monthThe natural gas and energy shortages sweeping across Europe, Asia and the United States have now affected the cost of shipping liquefied natural gas (LNG).
One…http://www.naturalnews.com
View Link Feed
https://gab.com/NaturalNews/posts/107148840928551603
https://www.naturalnews.com/2021-10-21-shipping-liquefied-natural-gas-rate-doubled.html
curiouser & curiouser
@threesevens
14h
·
The husband of Halyna Hutchins, the woman killed by Alec Baldwin, recently joined the law firm Latham & Watkins–the same firm retained by Michael Sussman.
https://www.reuters.com/legal/legalindustry/alec-baldwin-shooting-victim-was-wife-latham-watkins-lawyer-2021-10-22/
 Alec Baldwin shooting victim was wife of Latham & Watkins lawyerThe husband of Halyna Hutchins, the cinematographer who was fatally shot by actor Alec Baldwin with a prop gun on Thursday, is a corporate lawyer in Latham & Watkins’…http://www.reuters.com
Alec Baldwin shooting victim was wife of Latham & Watkins lawyerThe husband of Halyna Hutchins, the cinematographer who was fatally shot by actor Alec Baldwin with a prop gun on Thursday, is a corporate lawyer in Latham & Watkins’…http://www.reuters.com
View Link Feed
355 likes
77 comments
263 reposts
https://gab.com/threesevens/posts/107148664075031986
https://www.reuters.com/legal/legalindustry/alec-baldwin-shooting-victim-was-wife-latham-watkins-lawyer-2021-10-22/
It’s all very curious. It’s always hard for me to”think like a bad guy” and flesh out what the significance could be. Connections to Hillary and Sussman, okay. But what would be the motive for killing the wife of this man? To warn people not to talk?
Seems like nearly anything is on the table when dealing w/ demonic darkness!
Yes. I believe it’s a message sent to all the lawyers at Latham & Watkins:
‘We know where all your family members are at all times and we can take out your loved ones if you are subpoenaed and give Durham any incriminating information you may be aware of on the Clintons or Sussman. Keep your traps shut!’
I’m sure Baldwin has a long list of blackmailable offenses and can easily be controlled by the cabal.
Are you suggesting that Alec Baldwin was MK’d???
I’m offering my opinion that he may have done things or been involved in things that he would not want exposed, or perhaps owes someone favors. He may have been told to aim and fire and may not have been sure what would come of it. I don’t believe he was mind controlled into pulling the trigger. That was a choice he made.
Thanks for the clarification Ellie. Sad & seemingly evil all around 🙁
I’d say that I believe what Ellie does, but also that some nearly unbelievable mind control technology exists, and works far better than the public knows.
Most people can’t make such a claim credibly. I’m not saying I can, but I’m closer than most.
IMO this could easily have been MK, because Baldwin has anger issues, and can thus overcome the inhibitions needed to make an obsessive/compulsive implant that involves violence go forward.
Most people would not point a gun at another person because of training that would supersede any surreptitious implanted thought. They might successfully express an obsessive implanted thought like “Man, I’d like to shoot that bitch”, but this urge cannot overcome their religious or firearms training or strong bias (like, say, a commandment – THOU SHALT NOT….).
Baldwin would be perfect. The surreptitious MK would assure him that it’s a dummy gun, no bullets, and it’s OK to shoot her, because she won’t get hurt. It’s just training – but it’s BAD TRAINING.
And why is Reuters pointing out the relationship to Sussman?
Because somebody wants that message driven home – WIDELY.
People who DON’T have any iron in the fire, stoke the flames for the outcome. HILLARY will not be indicted. That is the message.
This is not just science. It’s MORE. This is real evil. MK is demonic. It’s ERROR – intelligent error – intentional error.
This is like that kid’s car blowing up in Georgia. We are up against a group that are FAR WORSE than the MAFIA.
Thanks for this very insightful perspective.
All of this is Way above my pay grade, so I’ll cling to Jesus & pray as He leads whenever I’m tuned in enough to recognize that “still small voice”…
Wonder if he was in treatment or on meds for “anger issues”… Ample opportunity for MK to be inserted there…
Yes!
Now – and this is very interesting – there are several drugs which are surprisingly effective for OCD. Those are clearly NOT desirable for facilitating MK, which depends of subconscious urges bubbling up into conscious action.
Yep. One might think that he had caught the MK “Delta Variant”….
Cast members are threatening to reveal Coke usage to keep up the rapid fliming schedule
Makes sense!!!
I believe fully that MK Ultra is used on many. I just don’t think it was needed for Alec Baldwin. He is completely onboard with these evil entities and, I think, he would willingly carry out one of their missions if they asked.
There are a lot of other explanations coming into view now, as well. Some reports that the guns were unattended on a cart. Two prior misfire events. Same union objecting to safety issues, who had the misfires, walking off the set right before the “number three”. And the guy who handed the gun to Alec and told him it was “OK” – on the set of another fatal accident.
______________________________________
Production crew walked off Alec Baldwin movie set hours before shooting in row over conditions and were replaced ‘on the fly’ by locals: Workers feared for gun safety after ‘TWO misfires’ days before Baldwin accidentally shot dead cinematographer. The lawsuit is going to be brutal. Some have noted, Q said many times, “The Hunt for Red October.”Now Colin Powell, who was like the James Earl Jones’ character just died, NASA just tweeted about the “Hunter Moon” in October, some rapper was just pushing his Red October sneakers, and the star of The Hunt for Red October, Alec Baldwin, just shot a girl who said on her web page she grew up on a Russian military base surrounded by nuclear submarines. Probably all coincidence, but curious none the less.
4chan Hollywood Insider Anon says daylight shoots never use blanks because you can’t see the muzzle flash, and insurance costs force a no-daylight-blank policy, actors never point the gun at a person for safety, it was live rounds which are not allowed on set, and the death fits with occult sacrifice practices, which he says are mind-bogglingly common in that culture. You can rule nothing out today. Interestingly about a third of the way down in the thread there is a picture of a goofy guy holding a picture of a small boy in his underwear, and Danny Devito making a guilty mouth expression next to him, with no comment. I advise you click on the guy’s ID in his first post and go down the thread reading his highlighted accounts of how Hollywood works. Very interesting, if true.
from the chans
100% what happened and by whom.
100% what happened and by whom.
Interesting software glitches going on here!
That would be me and my messy links
Could be – but there are many other messy things – trust me!!!
Also worked for Kirkland & Ellis and Skaden Arps.
IMHO could be the same type of scenario as the boyfriend of Kemp’s daughter having the inexplicable single car fatal crash right when the Ga election fraud happened…a warning but in this case..the husband is joining the evil empire so his loyalty isn’t in question.
Maybe someone doesn’t like Baldwin….a lot doesn’t like him
They killed that governor’s daughters boyfriend.
you don’t kill the one you want to coerce.
you kill or severely harm one of their “hostages to fortune”
Why do they call this a prop gun? It is obviously a fully functional fire arm. A prop would have a plugged barrel, no firing pin etc.
Good odds on at least a civil suit out of this.
Because it’s owned & managed by the property department? What are we missing because of this squirrel in drag???
I think that’s correct.
Just a guess here…
LOL!
Does anyone have an opinion of Jon Rappaport’s work? Specifically, the Matrix Revealed info he is selling?
https://blog.nomorefakenews.com/2021/10/23/cia-news-and-the-matrix-revealed/
This is the first I’ve seen of the Matrix Revealed. No opinion there.
Frankly, Rappaport is no different from the rest of the journalists out there who found out the truth and were rebuffed when it came to reporting it. They all have sources feeding them info. Who knows what is real and what isn’t. Well, other than western civlization thrived when the Church was strong.
Raven Constantine
@Artraven
1h
https://www.youtube.com/watch?v=zjbP6e20W2c
Ted Cruz TROLLS Rich Democrats With HILARIOUS New Bill! Sending Illegals To Their Country Clubs!
BEAGLES LIVES MATTER!
I’ve never had one, but have nothing against them.
We’ve lived next door to one or two for the past 2 decades. Their barks are Exceedingly Annoying. Our neighbor used to yell “Shut Up!!!” in frustration even louder than their barks. They do seem to have decent personalities though.
We (family of origin) had a beagle mix once & she was both cute & sweet but as an older dog her “death breath” was brutal. We (husband & I as young marrieds) had to watch her for a bit when I was pregnant (when all sorts of strange smells made me want to puke) & she’d come sit near me & pant & that cloud of funk made me green AND not with envy!
LOL I really know what you’re talking about. Being pregnant and smells…I couldn’t eat Mexican food for a long time after living near a restaurant..it was positively erpy to me !
I couldn’t stand the scent of turmeric, which we barely ever used but Wow that was So Bad 🙁
Crazy how the sensation of those things stay with us long long after
yes!
She needed her teeth worked on.
For sure!
Did you mean for this comment to be under the Girls’ Eyes meme?
No.
OK, my mistake 🙂
I thought yall were talking about the Beagles that were tortured and euthanized in Fauxi-Mengele sponsored labs
I thought that was just some outrageous catturd thing until seeing other reports 😡
My dad started to dislike them when my mother told him that when he died she was going to get a little beagle for company
In the arena of ideas with a possible clot shot bot…
Original Gabber
·
10m
@Rawed @Artraven Savage is people trying to help “free your mind” (think Morpheus in The Matrix) so that you might re-think your stated intention to see your children “vax’d”. Few, if any, of those you might perceive as attacking you are likely objecting to parental autonomy, I know I’m not.
I honestly grieve for the path you seem determined to trod. My family has been in the crucible of special needs for more than Two Decades. It’s incomprehensible to me that a loving, responsible parent would consider injecting their kid(s) with unknown experimental substances that have been repeatedly shown to have extremely dangerous consequences.
In Extremely Limited cases perhaps there is some justifiable reason to seek the “vax”. If you have Any Clues about what is Really Going On with this genocidal campaign you would flee far from it & defend your children with your life, Not put their lives on the line for a Lie.
The Covid clot shots (assuming the real substance & not saline is injected) appear to train one’s body to make the spike protein & then one’s own immune system goes into overdrive destroying itself. Some reports are that the “vax” recipient’s immune system deteriorates by about 5% on a weekly basis. This is setting many people up to have a very Dark Winter!
Please do some Real Research, including listening to “the other side” to see what dangers might be in store if subjecting someone to the shots.
Though I highly doubt you are this open-minded, here is a post that could start you on a journey of enlightenment…God Speed & may He give you True Wisdom to raise your children well…
https://www.theqtree.com/2021/10/20/the-clot-shot-the-explanation-thereof-and-the-faucist-lysenkoist-cdc-that-pretends-not-to-understand/
 The Clot Shot, The Explanation Thereof, And The Faucist-Lysenkoist CDC That Pretends Not To UnderstandI. The Clot Shot First things first. Nobody would be calling ALL of the various full-length stabilized SARS-CoV-2 S1 subunit spike protein vaccines “the clot shot”…The Q Tree
The Clot Shot, The Explanation Thereof, And The Faucist-Lysenkoist CDC That Pretends Not To UnderstandI. The Clot Shot First things first. Nobody would be calling ALL of the various full-length stabilized SARS-CoV-2 S1 subunit spike protein vaccines “the clot shot”…The Q Tree
View Link Feed
https://gab.com/ValerieCurren/posts/107152551690838110
2h
·
this “individual” is being pretty well savaged on @Artraven‘s timeline/post. I wonder if they are some type of China bot or Deep State plant. They follow no one & have no followers. Are they just sweeping up data on people passionate enough to try to talk sense into a seemingly clueless (or outright abusive) “parent”…
Rawed
@Rawed
4h
·
@Artraven Not up to you it is up to the parents of the children. Mine will get vaccinated soon
https://gab.com/ValerieCurren/posts/107151970974199133
The “bot” was among many comments to this meme, language warning, by ArtRaven

The problem is “woke,” scared, and brainwashed parents.
No doubt. Deliberately inflicting a horrible evil on your children while thinking you are protecting them is insane & apparently by design…
Over 100,000 more on the way plus the untold thousands from Afghanistan plus others that walked in and not counted. F J B
RutRoh
Interesting
https://twitter.com/TerriersVu/status/1452002853648670721
It’s more complicated than that. Dean Cain talked about it, and so did John Schneider. They both have been on set with props and real weapons.
Unless they’re frisking people at the gate, and the whole place is behind razor wire, anybody could sneak in 6 cowboy action bullets with powder and lead.
This was done to send a message to Sussman and everybody else in these law firms.
The Cabal is saying “you circle the wagons with us, or WE will shoot you.”
This is CANKLES-LEVEL protection.
If so…thank the Lord it was Baldwin that did the job for them instead of some MAGA leaning actor.
^^^ THIS I believe.
Reposting:
The tweeter says prop guns can’t fire live rounds. That’s not true. It depends on who is defining “prop.” Sometimes (probably many times) a prop gun is a real gun. The guns in the movies have to look authentic.
Yes – so much confusion here!
probably by design…fog of war & all…
There may be a leap or two in this.
So I went to twit world to see the above images & found this below, not in replies, & I’m not signed in but maybe some type of algorithm still recognizes me or my computer???
Feels like an alternate universe there right now…

Jacqueline
@Jacquel83423685
Looks to me like Buckingham Palace picked Tiffany Blue for their window coverings!!! LONG LIVE THE KING!
jedilizzy
@jedilizzy1
·
6h
Replying to @Jacquel83423685
Can someone please tell me what this means in potato language? https://abs-0.twimg.com/emoji/v2/svg/1f974.svg
1
7
Monica Shipley
@MonicaShipley9
·
5h
Jackie Kennedy wore a Tiffany blue outfit. When John John n Carolyn’s bodies “were found” they were cremated. Their ashes were “put” in Tiffany Blue Boxes. Trump’s Inauguration Day, Melania gave “big mike” Michelle Obama a gift in a Tiffany blue box. It’s all about John John etc
SteveMcAuliffe
@beholdcosmicwav
·
2h
Replying to @Jacquel83423685
Buckingham Palace deserted. All regalia stripped from the railings. Just 2 forlorn guards watching over a mausoleum
surely this is some type of troll with all the misspellings???
PATRICK E KENNEDY
@OfficialKing_Uk
·
7h
I Will Never Give Up You
My Patriots, My People Of This Great Nation.
I Will Never Give Up
I Will Never Fall
I Will Never Quit
I Will Never Bend
I Will Fight With Every Breath Of My Body
Why becouse i am a Kennedy your New king Of our Kingdom of Britannia.
https://twitter.com/OfficialKing_Uk/status/1451899847460720648
keem_113
@kr123_321
·
5h
Happy 123 day… 10.23 > 123
https://twitter.com/kr123_321/status/1451931988030599170
I’m guessing that this is Red Meat for The Wolf to tear apart!
Finally some truth coming out.
Autopsy images here too (from some time back)
blob:https://www.brighteon.com/24a295e3-915f-4a29-b908-16b858225f3f
@Minataur
48m
·
Dr. Cole: Covid-19 ‘Vaccine’ Is A Poisonous Attack On Populations – & “It Needs To Stop Now!” https://www.brighteon.com/8999e6d6-e8c8-4998-88bb-08cb9802094d
Dr. Cole: Covid-19 ‘Vaccine’ Is A Poisonous Attack On Populations – & “It Needs To Stop Now!” (MIRRORED)Dr. Cole: Covid-19 ‘Vaccine’ Is A Poisonous Attack On Populations – & “It Needs To Stop Now!” (MIRRORED)
Mirrored from Odysee channel @neverlosetruth at:-
https://odysee.com/@neverlosetruth:0/Dr.-Cole-on-COVID-Shots–quot-This-is-a-Poisonous-Attack-on-our-Population-and-it-Needs-to-Stop-Now!-quot-:a
Please…http://www.brighteon.com
View Link Feed
https://gab.com/Minataur/posts/107152618604025253
Oh, that’s beautiful!!!
This one is worse. She’s a KAPO. She’s a top Hitler lieutenant – eager to fulfill the plan. She has had opportunities to call off the “final solution” without penalty. That 16-2 vote was the perfect opportunity to put the brakes on this evil plan. She could even have hedged, and said “let’s wait 6 months for more science to come in”.
BUT NOOOOOOOOOO……..
This one is headed for Nuremberg II. No doubt about it.
Walenky is following FAUCI’s orders.
Something we can do. Something WE CAN DO.
https://nonvenipacem.com/2021/10/23/action-item-fda-accepting-public-comments-on-upcoming-decision-to-experiment-on-children/
I did it yesterday. People need to click on the link in the article, click on Comments, and choose a category from the drop-down list (someone suggested “Individual Commenter”). Write, and send.
Me too.
I doubt they’ll read mine past the first couple of sentences though.
I think that was Individual Consumer, but there would be either one or the other. Pick that one. 🙃
This afternoon, spoke on the phone with an old family friend. The last time I spoke with her was in May of this year. At that time, she was doing very well, sharp as a tack (she’s over 85), getting her vegetable garden ready, and so on.
Today — 6 months later — everything was different. She sounds like an elderly person in dementia. She says she’s living in the same retirement community where her sister and brother-in-law are (the truth is, she’s still in her own home). I had to explain who I am before she understood. She no longer leaves her house unless one of her daughters is taking her to an appointment. The vegetable garden is gone.
She told me that her daughters made sure she got the Pfizer “vaccine”, both shots, since I spoke with previously. And she just got a “booster shot.”
The mental changes in her based on our conversation of today indicate to me that this may be neurological damage caused by the “vaccine.”
That’s simultaneously heartbreaking and rage inducing.
The most vulnerable (children and seniors) being attacked by brainwashed loved ones.
If the truth ever permeated mainstream consciousness could they handle it or would they double down in denial ?
The latter, Molly. Democrats and RINOs have been doubling down since Barry was first elected in 2008. Oh wait, they have been doubling down since Bush’s election in 2000. Oh wait, I could continue…
I’d think mostly they would double down in denial, not wanting to believe what they’ve let themselves in for.
If some provable cure for the jab damage could be found, and made widely known, I feel sure there would be no shortage of takers.
I feel very sad for them and sincerely hope some such cure will be found.
Every single article that deals with combatting the shot I C & P and mail it to myself or else I’d space out where I read it.
I’m so sorry for your loss of a dear friend (at least as you knew her to be). Truly tragic 🙁
I am so sorry for your friend and you. It is hard to loose a longtime friend specially if she was still very active.
My kids wanted us to take the jab but I rejected it and my husband did also. They know they cannot change my mind 🙂
We lived a little longer have seen many things and sense when something is not right.
I realize that the changes in my old friend’s memory and cognition “post-vaccination” and “post-booster-shot” are strictly anecdotal, but perhaps there’s a bigger picture —
There are elements in the virus spike protein that appear to be from bat neurotoxin viruses, along with elements from other bat viruses, that were mixed into the Gain-of-Function experiments at the Wuhan lab in the creation of the chimeric virus called COVID-19. These spike proteins are also contained in the “vaccines” by Pfizer-BioNTech, Moderna, Johnson & Johnson, and other “vaccines” for the virus that have either mRNA or adenovirus DNA “delivery systems” for the spike protein into the recipient’s body.
One of these bat neurotoxic viruses, the Nipah virus, is STILL being experimented with at the Wuhan lab AND IN OTHER LABS, including in the U.S. — and that Peter Daszak’s EcoHealth Alliance, at the root of the Wuhan lab GoF work, is STILL GETTING NIH FUNDING for Nipah virus experiments.
It’s not without the realm of possibility to posit that the COVID-19 virus, AND/OR the “vaccines”, AND/OR the viral “shedding” that’s appearing to be a real phenomenon among the “vaccinated”, can ALL negatively impact the brain and neurological functions. Dr. Zandre Botha came on the Stew Peters show and, in reporting her findings on what she saw via 400x and 1000x microscopic examination of a J&J “vaccine” vial contents (horribly deformed blood cells, square-shaped black items, etc.), ALSO spoke about how SHE HERSELF, being in contact with “vaccinated” patients who come to her presenting with various brain function issues post-“shots”, feels as if her brain is “spinning inside her head”, among other things.
Which will be ignored and attributed to her age.
So so sad and unnecessary.
Sadly I have noticed mental changes in my siblings who’ve taken the jab. They don’t seem to notice BUT I see it.
related to that aka Obama era protest to protect Freedom & ranching rights?
@NotYourPolitics
14h
·
They call the Trump-loving, Christian conservatives, “Far-right”.
https://apnews.com/article/coronavirus-pandemic-canada-health-public-health-idaho-0d2015c826bb01a2cd04b31254c870c5
 Report: Far-right anti-government group grows significantlyBOISE, Idaho (AP) — A far-right group launched by anti-government activist Ammon Bundy is rapidly expanding nationwide and making inroads into Canada, according…apnews.com
Report: Far-right anti-government group grows significantlyBOISE, Idaho (AP) — A far-right group launched by anti-government activist Ammon Bundy is rapidly expanding nationwide and making inroads into Canada, according…apnews.com
View Link Feed
https://gab.com/NotYourPolitics/posts/107149580827761794
https://apnews.com/article/coronavirus-pandemic-canada-health-public-health-idaho-0d2015c826bb01a2cd04b31254c870c5
Ammon AND his family ARE Patriots.
Yes! Didn’t one pay with their life, executed by FIB or something? unarmed in a car?
Likely you are referring to LaVoy Finicum.
Video ~17 minutes. Start about 9 minute mark. Lavoy is gunned down shortly after.
https://www.washingtontimes.com/news/2016/feb/3/fbi-video-robert-lavoy-finicum-shooting-oregon-fai/?utm_source=GOOGLE&utm_medium=cpc&utm_id=chacka&utm_campaign=TWT+-+DSA&gclid=CjwKCAjw5c6LBhBdEiwAP9ejG7sgTfwY9qDdimidVWMvtgaspsH5C5oahwNppzrJI8Jg7YVk0zmgvBoCuPUQAvD_BwE
Thanks, KBK. We Have to Remember the ones that have been lost in this War for Freedom!
This is awesome! How did CBS show it???
[video src="https://media.gab.com/system/media_attachments/files/088/537/788/original/d7660bd915ffe81d.mp4" /]
@candijo
1h
·
Dads stepping up and getting involved at their kids school.
More of this please.
https://gab.com/candijo/posts/107152608898863536
That is fantastic!
Yes!!!
CBS aired it because it’s black men coming to help. That’s great but I guarantee you if it had been white fathers there would’ve been an outcry of fear and loathing
Good point, or just plain ignoring it…
If it had been white fathers, the school and the news would call them domestic terrorists.
But as it turns out, there was never any chance of that.
Practically none of the white kids have SIVS (Spastic Impulsive Violence Syndrome).
We need similar in Detroit schools (& homes & neighborhoods)…
“We need similar in Detroit schools (& homes & neighborhoods)…”
______________
Especially if they lower the retirement age to 35, so all these Dads have time to babysit their hellions at school. 👍😂
You’re hilarious. It’s more like responsible dads watching over the out-of-control fatherless in the neighborhoods…
Did they all hit the Lotto?
How are they able to wander around a school building from 8am to 3pm every day?
Why doesn’t the school system control the violence?
Why are the children so insanely violent in the first place?
If the parents won’t raise the kids to not be thugs, and the school system won’t provide a safe environment for education – or even provide an education in the first place – then what’s the point?
The whole thing just smacks of a ‘feel good’ story that studiously avoids asking ANY of the obvious questions about why such a ‘solution’ was necessary in the first place.
And why do all these middle aged fathers have no place else they have to be from 8am to 3pm on weekdays?
When I look at these kinds of things, I don’t see the ‘aw shucks’ and the ‘isn’t that sweet’ aspect.
I just see a temporary band-aid on a critical wound, and questions that I know will go unanswered, because that’s how everything is.
Because nothing makes any sense in the world.
All most excellent points, Scott. But that fluff piece still puts a smile on my face as it is a tiny drop in the solution ocean…
It’s nice that all the Dads are retired, and have all this time to babysit their little hoodlums, yakin’ it up, havin’ a good time 👍
LOL
Seen on Gab

Time to round up the CATL and take them down…..
Oh, and apparently IN-N-OUT Burger has a new menu item (H/T Babylon Bee 🙂 )…
https://babylonbee.com/news/in-n-out-unveils-exciting-new-burger-the-lets-go-brandon
(catch the price)
LOL
Emerald Robinson Rocks!
https://alexberenson.substack.com/p/urgent-covid-vaccines-will-keep-you/comments
GOOD! She got this. It’s HUGE.
Yes!
AND logic
TikTok?
ChiCom MK?
Please, all patriots – take TikTok and Zoom and all other ChiCom apps off your phones, unless you want CCP spies in America to know your whereabouts and everything else 24/7.
???
TikTok and Zoom are used by the CCP to watch you and study you.
NOT me! No cell & none of those things on the computer. Can’t vouch for family though 🙁
Interesting AND conveniently “coincidental” how many businesses & schools were conducted over Zoom during the fake-a-demic. Not planned at all/s
Good news for cancer patients…
https://www.wnd.com/2021/10/revolutionary-cancer-treatment-wakes-immune-system-destroy-tumors/?utm_source=Gab&utm_medium=PostTopSharingButtons&utm_campaign=websitesharingbuttons
@HerbstDW
19h
·
Revolutionary cancer treatment wakes up immune system to destroy tumors https://www.wnd.com/2021/10/revolutionary-cancer-treatment-wakes-immune-system-destroy-tumors/?utm_source=Gab&utm_medium=PostTopSharingButtons&utm_campaign=websitesharingbuttons
 Revolutionary cancer treatment wakes up immune system to destroy tumors(STUDY FINDS) – A revolutionary cancer treatment is combining several therapies to successfully destroy tumors, scientists from the Massachusetts Institute of Technology…WND
Revolutionary cancer treatment wakes up immune system to destroy tumors(STUDY FINDS) – A revolutionary cancer treatment is combining several therapies to successfully destroy tumors, scientists from the Massachusetts Institute of Technology…WND
View Link Feed
https://gab.com/HerbstDW/posts/107148455584930609
Big Pharma: “Quick, hide the formula!”
yep 🙁
GRRRR! They can’t convict and sentence this monster soon enough.
I want that insane egotistical pos on a pike, but dont let it kill him. Let him suffer as long as possible to make up for the humans and animals he has put in harms way for DECADES. And anyone who wants to help him can join him. Yeah he deserves a trial first but a fast one.
Beauty wants to take out The Beast

We’re gonna find out how CHINA moderates us.
AND – there is MUCH about this known publicly already, so if Facebook holds out on giving up the evidence, Texas can get the judge to penalize them GREATLY and DEMAND the known evidence.
Doesn’t Trump have a lawsuit against FB &/or Twitter? Didn’t FL put similar laws on the books? Turn it into a multi-state Truth Dig?
Canadian on Gab perspective:
@TankmanbradRequested
Sorry, I dont do follower accepts any more (traitors out there)
Here is Link to 200+ files exposing fraudulent COVID Globalist agenda, free to access, updated fairly often with more: https://www.dropbox.com/sh/y3udbd2de37u3kj/AADtWyJKKhULxpwMGYNY3gtza?dl=0
The Q code is the url as well.
Canada is defiled and lost. A degenerate narcassist asshole who stole an election is telling us all we have no identity, so he will eagerly build us a terrorist-loving, commie one. Canada = DEAD AND GONE
https://www.dropbox.com/sh/y3udbd2de37u3kj/AADtWyJKKhULxpwMGYNY3gtza?dl=0
Possible useful Covid resource above
Fake blue drift exposed, just like Russ Ramsland’s report on 2 counties in Texas during the 2018 election.
@GolddiggerPatriot
22m
·
Emery County, Utah (2020 Election)
City: Huntington
Big Lie
Trump 4207 Biden 572
Big Truth
Trump 4408 Biden 371
https://gab.com/GolddiggerPatriot/posts/107153013892362996
In Real Votes Bye-Done got less than 10% of Trump’s votes 🙂
Bonus “coin” image on a Steve post!!!
Nothing to see here
https://www.thegatewaypundit.com/2021/10/chinas-military-declares-biotechnology-warfare-fundamental-guiding-principle/?utm_source=Gab&utm_campaign=websitesharingbuttons
CCP is a pestilence.
Jesus & Freedom the cure!
LOL, this guy must be in trouble. The DemCom Messiah to the rescue. Zero shuckin and jivin while the moron candidate nods his head
“We don’t have time….”
They ALWAYS say this shit.
Whether it’s Barky Barky or Pigly Osi or Angry Greta, it’s always the same – we don’t have time to figure out if it’s the right thing – we just have to DO IT NOW!!!
Wow did I miss that mellifluous voice & serene visage/s What’s he so angry about? I thought their side was winning!
https://s.w.org/images/core/emoji/13.1.0/svg/1f1fa-1f1f8.svg #AZ https://s.w.org/images/core/emoji/13.1.0/svg/1f1fa-1f1f8.svg #PATRIOT https://s.w.org/images/core/emoji/13.1.0/svg/1f1fa-1f1f8.svg III% https://s.w.org/images/core/emoji/13.1.0/svg/1f1fa-1f1f8.svg
@SteveEmery03
·8m
It’s been proven that the dossier was a product of Hillary with help from George Soros and McCain, we know Fauci funded and then lied about the creation of the virus, that the FBI set up the January 6th protesters, we proved that Joe Biden stole the AZ election and likely all the other swing states, and still not a single arrest! Republicans absolutely suck #MAGA
Interesting, concerning Dominion.
The U.S. Government & The Third-Party Vendor Are OneA lawsuit in Colorado alleges that a private company currently trying to chill free speech in America is actually the government itself.
https://emeralddb3.substack.com/p/the-us-government-and-the-third-party
Jason Miller
@JasonMillerinDC
·5h
Changing the company name ain’t gonna help, Zuck…
“Facebook needs to change its CEO, not its name: The public has lost trust in the network and the only way to regain it is for Zuckerberg to stand down, professor of communications says”

https://twitter.com/FDSheckler/status/1451863147401338882
She comes from the same gene pool as all of the WH staffers. Harry Potter, TikTok, Selfie freaks
Replying to @FDSheckler
and @JUSTcatmeme2
OMG. I suppose that as producer Alec Baldwin is responsible for her employment.
This gets worse by the hour.
This is all very wrong, but makes loads of sense.
There will be several chumps in this operation – IMO we know two of them now – Biden and this chick. They’re not innocent – not at all – they are “disposable dark side” – and yes, this is all very satanic.
No WAY should this chick have ever been an “armorer” – that is a total LAUGH.
Supposedly her father was/is a long time set armorer…probably called him and he says his kid wants the job ?
Assistant director took the gun off the cart without checking and handed it to Baldwin.
Zero chain of custody control.
It’s all WRONG – it’s absolutely WRONG.
She should have demanded back the weapon from the assistant director and checked it in front of all of them, then handed it DIRECTLY to Baldwin. That’s the only way to be safe.
C’mon…this is the new and improved generation. Lax and rules are so constructing ya know. Policing other people is sooo authoritarian and Baldwin is superchill with the cool kids
I read that her father has been an armorer on a number of movies. She wasn’t sure she was ready for the one she did before this one, but it went okay. It’s hard to believe they put her in charge of something that important.
I’m going to be completely honest. I’ve never spent a day of my life as an “armorer”, but I am sure I could do her job more safely than she could – and probably almost as competently.
Absolutely! You know gun safety and have common sense! Experience helps, too.
“…but I am sure I could do her job more safely than she could —”
___________
No doubt.
But could you do this…?
😂
https://twitter.com/i/status/1451863147401338882
LOL.
Absolutely DEMONIC.
She is the daughter of a famous armorer, so really its nepotism. I dont remember his nane. Supposedly her father trained her and im sure Baldwin abd the other producers were so lazy they just expected her to be competent.
Stupid nfers should be bankrupted over this, when you consider the safety issues in totality.
Baldwin is probably already figuring out how to hide his money.
You know about his wacky wife ? She’s probably freaking out..she uses his fame to make her own weird self famous
With union troubles and crew upset that’s the time to be really diligent about things
Yeah, I have suspicions there for sure.
Know what else? This is covering up HUGE vaccine news – TWO giant items.
With the other guy , Laudrie kind of going off the radar a new sensation is needed ?
Yup. New distraction for the masses. Especially conservatives (Alec Baldwin misfortune).
Many of us can read all about Baldwin without taking our minds off any of the important issues, the Fauci news, etc. We can multi-task. (And the Baldwin issues could tie in with the others.) But maybe not so for people who only follow things peripherally and are not presented deeper perspectives on the news. This is a perfect distraction because everyone wants to solve a mystery.
Yup. Exactly.
Gotta follow it, to some extent, no matter what.
She barely seems trained. Something is not right with her.
“Something is not right with her.”
___________
What gave it away?
I suspect that she was technically proficient, but that 80% of the difficulty in being an on-set armorer is keeping the outsized egos in line.
Yeah, I’ll bet her old man was good at his job, whipping the big heads into obedience. Bet she was a FAIL, and part of the safety problems noted.
Though I tell you, if there was a bad union guy involved, it would be a sweet op.
Rile up his buddies against the production, make accusations of safety problems, PROMOTE some safety problems, get everybody to leave in a huff, and be connected to the non-union replacements – who are really professional trouble – who finish the job.
“I suspect that she was technically proficient,”
______________
I don’t, not even a little bit.
“She is the daughter of a famous armorer, so really its nepotism.”
______________
Everybody knows armorer skills are hereditary. 👍
.
.
.
.
.
.
.
.
.
🙄
.
.
.
.
.
.
.
.
. 🤣 😂🤣
Here’s the Daily Mail article – good investigative work.
https://www.dailymail.co.uk/news/article-10123003/I-wasnt-sure-ready-Doubts-head-armorer-24-charge-guns-Alec-Baldwin-film.html
[in my best Steve Buscemi voice]
Why hire a professional firearms instructor, when you could have dancin’ Bambi, the pierce-tat Boom-car music chick?
😁
Oi o th police etc are calling the shooting by Baldwin a tragic accident? I thought that was determined by an impartial investigation.
Looks like an accident, but I don’t buy it.
But why and why again would there ever be real live bullets on the set?
If she was competent and loaded the gun she should know what real ammo looks like and what the blanks look like
There shouldn’t be any live bullets on the set, thus somebody brought them in. A competent armorer would FORBID them on the entire site, and threaten to fire anybody caught with live ammo, report them to their union, etc.
I half suspect the assistant director of swapping in a live round, but my prime theory is that it was done by one of the non-union who were brought in. They would do it AFTER the armorer checked the guns.
The non-union team brought at the last minute strike me as the killers.
I have thoughts that the whole movie was a setup, based on knowing the camera chick was linked to Sussman.
Something is really wrong with it.
I’ve even had thoughts that the whole thing is a LIE to shut up Sussman, but I know better. It’s real.
This is a perfect example of where incompetence provides cover for evil.
Interesting and plausible. Makes me wonder if the walkout that morning was organic or if those people were paid or coerced to do it.
See my theory above, based on how real IC provocateurs operate. Sometimes just ONE of them can take down an entire project, division, or even a company. Just one really good provocateur in the crew – better TWO – could have wrought all of this, to create the “safety narrative”, etc., and then they bring in the real talent to slip in the bullets, etc.
Right!
I just posted this on UTree. Guns were left on a table outside the building on the set.
https://www.dailypress.com/nation-world/ct-aud-nw-alec-baldwin-movie-set-shooting-death-20211023-ffphv7jy5nawreth4xfmtjttse-story.html
So the armorer had set out some guns that I guess people assumed were safe to use, on a table. Could they have been tampered with? Halls grabbed a particular one and handed it to Baldwin. Did he know that one had live ammo? Or, if he had nothing to do with it, maybe all those guns had been seeded with live ammo. Investigators probably already know the answers to these questions.
Here was my answer on the U Tree…..
My top suspect is a dirty union. Yeah, one of theirs got killed. That’s COVER.
They seed the guns. They walk off.
“Boom.”
Check this line out in the DM article:
#################
“It is the same union that had been threatened to galvanize an industry-wide strike in protest over poor working conditions including low pay and laxed safety. IATSE Local 44 – whose members were involved in the Rust production – said in a statement to its members that no union members were on the set on Thursday. ”
“One text message that was circulating on social media, shared repeatedly by union members, refers to a ‘walk out’ by staff the day before the tragedy. ”
“The text message claims that Halyna was one of the few people who decided to stay. She belonged to IATSE Local 600 and had been campaigning for better conditions for her team when she was killed.“
Very plausible.
If you keep asking questions like that, how are they supposed to cover this up? 😂
Check this out. https://www.thegatewaypundit.com/2021/10/dave-halls-man-claimed-gun-alec-baldwin-set-safe-also-present-brandon-lee-died-prop-gun-mishap-1993/
Dave Halls, the Man Who Claimed Gun on Alec Baldwin Set was Safe, Was Also Present When Brandon Lee (Bruce Lee’s Son) Died of a Prop Gun Mishap in 1993
That’s just like Stefan Halper being right in the middle of Watergate.
“That’s just like Stefan Halper being right in the middle of Watergate.”
____________
It’s ironic, I tell ya.
LOL. Maybe even “iron-sight-on-ic”!!!
“It’s ironic that Halls was also there when Brandon Lee died.”
___________
Yeah, ‘ironic‘.
That’s just the word I was thinkin’ of…
From Gab:
Sounds good to me.
Check this out.
Like that’s a good thing…
Right?!
Basically saying the social credit system is slavery. A person isn’t free to do anything except what the govt allows
The government will never allow a movement it does not approve of to exist.
Or fake control & manipulate, like the Proud Boys!
He’s been raised under socialism – he’s a good commie!
Talks of taking away peoples homes makes him laugh like a donkey. 🙄
Weird, I cannot Like your comment but could like Wolf’s below just fine…
So systematically stomping freedom is a good thing? whatever
Another “coincidence”. NOT suggesting Balwin is innocent. Baldwin should have verified the weapon was unloaded, should have NEVER pointed the weapon at a person AND NEVER pulled the trigger.
FTA Emphasis added
In 2000, Halls was the second unit’s first assistant director on The Crow: Salvation, the sequel to the film in which Bruce Lee’s son Brandon Lee was killed in an on-set firearms mishap in 1993.
https://www.thegatewaypundit.com/2021/10/dave-halls-man-claimed-gun-alec-baldwin-set-safe-also-present-brandon-lee-died-prop-gun-mishap-1993/
That’s a pretty bizarre “coincidence” 🙁
Marine Exchange (@MXSOCAL) Tweeted:
RPT 10/22: 163 total ships inport LA/LB incl 105 at anchor or hold areas & 58 at berths. Of the 163, 106 are container ships incl 77 at anchor or in hold areas & 29 at berth. 51 vessels in hold areas; 42 container ships, 2 tankers, 4 bulk, 2 gen cargo, 1 combo gen cargo/container https://t.co/ALvpdTNvLK
Added to the sidebar!!!
I’m a little late to the party.
But I see it was a smashing success.
Thank you Steve, for another Science Saturday,
even if it’s Monday now.