The header image for today’s offering of an end run is courtesy of Grammarist and Google Images.
Health Friday is a series devoted to information regarding Big Pharma, vaccines, general health, and other topics.
There are Important Notifications from our host, Wolf Moon; the Rules of our late, good Wheatie;, and certain caveats from Yours Truly, of which readers should be aware. They are linked here. Note: Yours Truly has checked today’s offering for AI-generated content; to the best of her knowledge and belief, there is none. If readers wish to post AI-generated content in today’s discussion thread, they must cite their source. Thank you.
Special Note regarding today’s offering: In Yours Truly’s opinion, it is very important that HHS Sec. Robert F. Kennedy, Jr., gets the URL link to today’s post. Please help in this regard if possible. Thank you. https://x.com/SecKennedy; https://truthsocial.com/@seckennedy; https://www.instagram.com/seckennedy/.
Part One of the series is here: https://www.theqtree.com/2026/02/27/health-friday-2-27-2026-open-thread-modernas-mrna-1010-and-the-end-run-around-hhs-sec-kennedy-jr-part-one/.
Part Two of the series, today’s offering, discusses two very important areas related to mRNA-1010, Moderna’s modRNA-based influenza “vaccine”, the (amended) BLA (Biologics License Application) for which the FDA has agreed to review, after the agency first refused to file the original BLA. The first area is the Patent document for mRNA-1010. The second area is the involvement of Blackstone Life Sciences in funding mRNA-1010.
**** Preliminary item before the first discussion area; keep this in mind regarding anything related to mRNA-1010: The EMA (European Medicines Agency) has just approved the use of Moderna’s COMBO modRNA “vaccine”, mRNA-1083, for use in the European Union. mRNA-1083 is combination of Moderna’s modRNA “vaccine” of multiple influenza strains, mRNA-1010 — PLUS, Moderna’s modRNA COVID-19 “vaccine”, mRNA-1283 (mNEXSPIKE.) Please see: https://www.stocktitan.net/news/MRNA/european-medicines-agency-s-committee-for-medicinal-products-for-rod4htf7c1gd.html, “European Medicine Agency’s Committee for Medicinal Products for Human Use Adopts Positive Opinion Recommending Marketing Authorization of mCOMBRIAX, Moderna’s mRNA Combination Vaccine Against Influenza and COVID-19”, 26 February 2026. mRNA-1083, also called mCOMBRIAX, is the “end-product” that Moderna is pushing to have the FDA approve for use in the United States. The first step in this process was to get FDA approval for the mRNA-1283 component, mNEXSPIKE. which the agency did on 30 May 2025. The next step is for Moderna to get FDA approval for the mRNA-1010 component.
>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>
First discussion area of today’s offering: THE PATENT DOCUMENT FOR mRNA-1010:
The Patent document for mRNA-1010 is found here: https://patents.google.com/patent/EP4274607A1/en. Note that this Patent was filed under a European Patent listing. The Patent listing screenshot is below, followed by a screenshot of the Title and the Abstract:
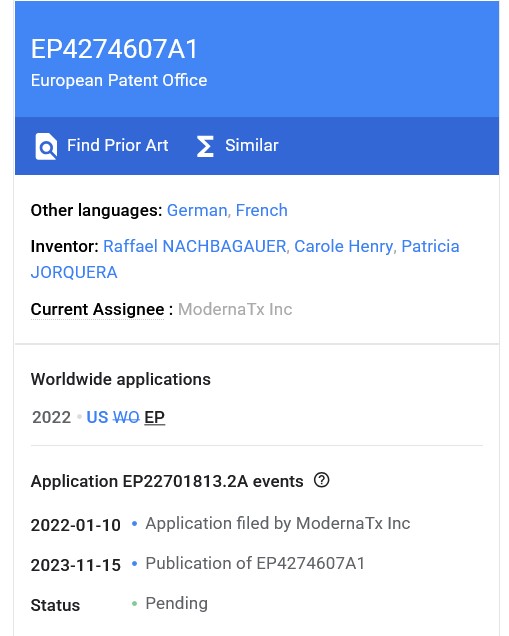

Yours Truly will not go through the entire Patent document for mRNA-1010. However, the screenshots of sections of the Patent document, below, are important, as they show what, in this writer’s opinion, are decisions by Moderna to lab-create an mRNA-based influenza virus “vaccine” that has multiple strains of virus and lipid nanoparticles variations — only a few of which “vaccine” formula variations could have been fully tested and the test results analyzed, either in lab mice experiments or in human clinical trials, before March 2026.
The Patent document for mRNA-1010 is what Yours Truly calls a “kitchen-sink” document. It lists and describes multiple modRNA-based influenza strains that “may” be used in the formulations of the “vaccine.” It lists multiple possible formulations for the lipid nanoparticles that “may” be used the formulations for the “vaccine.” It lists the multiple combinations of amounts of the modRNA-based influenza strains antigens that “may” be used in the formulations of the “vaccine.” These multiple variations and formulations are called embodiments in the Patent document.
Example: It appears that up to EIGHT (or more) different modRNA-based strains of influenza viruses “may” be used:


Example: It appears that multiple variations of modRNA strains ratios (from HA antigens or from NA antigens) “may” be used in the EIGHT strain “version”, such as 2:2:2:2:1:1:1:1;, or, as 3:3:3:3:1:1:1:1. Among others:

Which is also described in the Claims section of the Patent document; see below:

Note: “HA” refers to hemagglutinin, an influenza virus surface protein, assists virus particles to “attach” to red blood cells; “NA” refers to neuraminidase, an influenza surface protein that assists in virus replication.
Example: It appears that multiple types of lipid nanoparticles “may” be used in the multiple types of “vaccine” formulations; such, SM-102 (an “ionizable lipid”, already used in SPIKEVAX and in mNEXSPIKE); DOPE; SDPC; PDG-DMG; plus, OTHER types of lipid nanoparticles that Moderna will create for use:


Note that various types of pseudouridines, including N1-Methylpseudouridine (also used in SPIKEVAX and in mNEXSPIKE) “may” be used in the “vaccines” formulated.
Example: It appears that Moderna will determine how many modRNA-based influenza strains will be used in the “vaccine.” Moderna will also determine how many types of lipid nanoparticles (LNPs) will be used in the various “vaccine” formulations:

Note the language that TWELVE OR MORE influenza strains “may” be used in the “vaccine.”
**** Yours Truly will again stress that the European Medicines Agency (EMA) has just approved the Moderna “combo-vaccine”, mRNA-1083 (mCOMBRIAX), for use in Europe. mRNA-1083 contains mRNA-1010, plus mRNA-1283 (mNEXSPIKE, Moderna’s other modRNA COVID-19 bioweapon “vaccine” [the other being its “SPIKEVAX” product].) Look again at the details from the Patent document for mRNA-1010, above. How can a “vaccine” like mRNA-1010 be approved for use anywhere, given that Moderna can choose anywhere from ONE to TWELVE different modRNA-based influenza strains, plus ONE or MORE types of lipid nanoparticles, along with combining these in MULTIPLE FORMULA VARIATIONS, to produce this injectable?
Further down in the Patent document, there is the section Table 5. Experimental Groups. It is in this section that there is detailed discussion of the lab mice experiments with all three of the Moderna “mRNA-10….-vaccines” group, which comprises mRNA-1010, mRNA-1020, and mRNA-1030. This section describes the various influenza strains combinations used in these experiments:

From the section Pharmaceutical Formulations, it appears that Moderna will be using what Yours Truly will call the “crapshoot method” for choosing the amounts of ingredients to be combined into mRNA-1010:

And, from the Titer subsection of the above, it appears that Moderna will also use the “crapshoot method” to estimate (guesstimate) the amount of titer in the blood of a person “vaccinated” with mRNA-1010:

>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>
What is a possible “underpinning” to the “multiple options” for Moderna’s mRNA-1010 formulation? Perhaps it is this 2023 article: “Rethinking next-generation vaccines for coronaviruses, influenzaviruses, and other respiratory viruses.” Jefferey K. Taubenberger, David M. Morens, Anthony S. Fauci. https://pmc.ncbi.nlm.nih.gov/articles/PMC9832587/. 11 January 2023. Please see the screenshots from this article, below. First, the Abstract:

Next, from the Introduction:

Finally, from the section Mucosal responses:

If Yours Truly read this paper correctly, the main idea appears to be the development and use of modRNA-based “vaccines” to be administered intranasally. Note also that Dr. Jeffery K. Taubenberger, one of the paper’s co-authors, is now the Acting Director of the NIAID; and, who holds the Patent for the “universal influenza vaccine” that he invented in 2020 while working at NIH.
>>>>>>>>>>>>>>>>>>>>>>>>>>>>>>
Second discussion area of today’s offering: The involvement of Blackstone Life Sciences in the financial backing for mRNA-1010. Yours Truly begins here, with screenshots from the Moderna Earnings Call Transcript published by The Motley Fool on 13 February 2026 (https://www.fool.com/earnings/call-transcripts/2026/02/13/moderna-mrna-q4-2025-earnings-call-transcript/); first, the Date and Participants section; then, a portion of the Takeaways section; and, the Risks section:



Thanks to The Motley Fool for the above information.
Moderna has realized that their United States COVID-19 bioweapon “vaccine” market, and their modRNA-based influenza “vaccine” market, are shrinking. There were income losses for the company. Meanwhile, Moderna has multiple other injectable products “in the pipeline” for development and testing prior to submitting applications to the FDA for BLAs for these products. This, then, presents an issue regarding the funding of the company’s endeavors.
It appears that Moderna has found a solution to two of these issues — the situations with funding; and, with developing mRNA-1010 and with mRNA-1083 to get these products approved for use: the company obtained an “infusion” of up to $750 Million dollars from Blackstone Life Sciences in 2024: https://pharmaphorum.com/news/blackstone-bankrolls-modernas-flu-shot-750m, “Blackstone bankrolls Moderna’s flu shot for $750m”, Phil Taylor, 28 March 2024. Please see the screenshot from this article, below:

Note closely the language in the article screenshot above related to Blackstone Life Sciences receiving future “rights and royalties payments” from Moderna for the sales and use of mRNA-1010 (the multi-strain and multi-LPN modRNA influenza “vaccine” being considered by the FDA for approval), and of mRNA-1083 (the combination modRNA “vaccine” of mRNA-1010 plus mRNA-1283 [mNEXSPIKE, modRNA COVID-19 bioweapon “vaccine”], which was just approved in Europe this month.)
What is Blackstone Life Sciences? It is a subsidiary of Blackstone, an international asset-management and acquisition company that currently has over $1Trillion dollars in managed assets: https://www.blackstone.com/the-firm/. Please see the screenshot from this website, below; “AUM” = Assets Under Management:

The Blackstone Life Sciences subsidiary was founded in 2005. The headquarters location is: 314 Main St., 15th Floor, Cambridge, MA, 02142. Please see the screenshot showing the Blackstone Life Sciences investment in Moderna on 15 March, 2024, below, per https://pitchbook.com/profiles/investor/11151-91#investments:
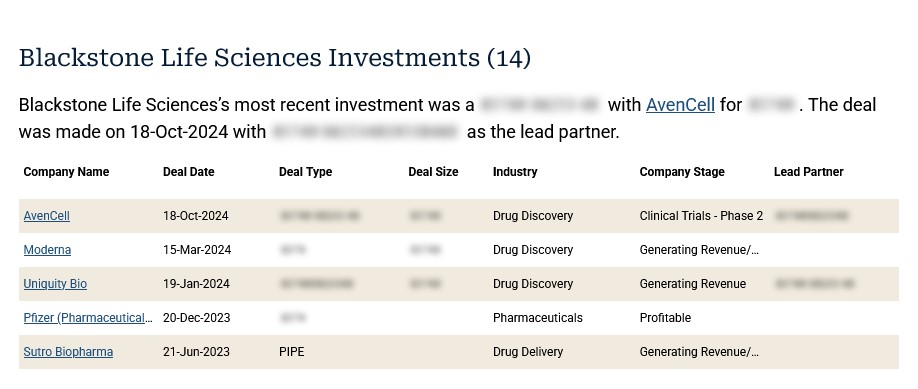
The above information, with thanks, is from the non-subscriber website page on Blackstone at Pitchbook.
Is it remotely possible that Moderna is under pressure to “make good” regarding the potential for rights and royalty payments to Blackstone Life Sciences for the sales and use of mRNA-1010 and of mRNA-1083? Is it remotely possible that this is what is behind the pushing by Moderna (which was ultimately successful) in the FDA’s sudden reversal of the Refusal to File letter for the BLA for mRNA-1010, and that agency’s subsequently agreeing to consider an “amended” BLA for this “vaccine”? Is it remotely possible that Moderna (and/or Blackstone Life Sciences) regard the huge potential of the United States market to be important in terms of “Return on Investment” — regardless of the potential for “vaccine”-induced damages from mRNA-1010 and/or mRNA-1083?
Is it remotely possible that the FDA — and Dr. Martin Makary, FDA Commissioner — do not realize that they are being used as pawns in a kind of “vaccine chess game” being played by Big Pharma and Big Investment entities? Has Dr. Makary read the Patent for mRNA-1010, or had it explained to him?
THERE IS NO PLACE IN THE HUMAN BODY FOR AN mRNA-BASED, modRNA-BASED, saRNA-BASED, OR taRNA-BASED PRODUCT IN ANY FORM.
Peace, Good Energy, Respect: PAVACA
(Intellectual Property and Disclaimer: Except for items cited above that are available on the internet, the ideas and opinions of today’s offering are by PAVACA. Credit must be given to PAVACA if the ideas and opinions of today’s offering are used by other blog writers, by podcasters, in social or print media, or in any other form.)
