The header image for today’s offering of an end run is courtesy of Grammarist and Google Images.
Health Friday is a series devoted to information regarding Big Pharma, vaccines, general health, and associated topics.
There are Important Notifications from our host, Wolf Moon; the Rules of our late, good Wheatie; and, certain caveats from Yours Truly, of which readers should be aware. They are linked here. Note: Yours Truly has checked today’s offering for AI-generated items; to the best of her knowledge and belief, there are none. If readers wish to post AI-generated items in today’s discussion thread, they must cite their source. Thank you.
Today’s offering is Part One regarding the current situation with Moderna’s latest entry into the mRNA-based “vaccine” platform: the company’s influenza “vaccine”, mRNA-1010.
On 11 February 2026, Dr. Vinay Prasad, director the CBER division of the FDA (Center for Biologics Evaluation and Research) sent an RTF letter (Refusal to File letter) to Moderna in response to that company’s BLA application (BIologics License Application) review request for mRNA-1010, a modRNA-based “vaccine” against several strains of influenza. Dr. Prasad cited the lack of a true placebo control group in a study by Moderna using this “vaccine candidate.” Please see the screenshots, below, from https://pharmacally.com/fda-issues-refusal-to-file-letter-for-modernas-mrna-1010-flu-vaccine/, 11 February 2026:


However, by a few days later, the situation had changed completely: the FDA “reversed course”, agreeing to review Moderna’s BLA (amended) application for mRNA-1010.
By 18 February 2026, a “Type A” meeting had been arranged between the FDA and Moderna regarding the RTF letter. Moderna agreed to submit an “amended” BLA application to the FDA for mRNA-1010. The FDA accepted the amended BLA application. The FDA will make a final decision on approving mRNA-1010 by 5 August 2026. Please see the screenshot, below, from https://www.biospace.com/fda/fda-reverses-course-on-modernas-mrna-flu-shot-application-promising-august-decision, Heather McKenzie, 18 February 2026:

Note: A Type A meeting at the FDA is a “high-priority” meeting; sometimes, Type A meetings are called “milestone meetings.” Per https://facetlifesciences.com/ and https://seed.nih.gov/ searches.
Please see the screenshots, below, on this “volte-face” from https://www.thefocalpoints.com/p/fda-reverses-course-will-now-review, “FDA Reverses Course, Will Now Review Moderna’s Controversial mRNA Flu Vaccine”, Peter A. McCullough, MD, MPH, 20 February 2026:


Then, from https://www.biopharmadive.com/news/fda-reverses-course-review-moderna-approval-application-influenza-812432/, “FDA reverses course on Moderna’s flu vaccine”, Delilah Alvardo, 19 February 2026 (the quotation in the screenshot is from Mani Foroohar):


Note that last sentence, by Mr. Faroohar. It clarifies three “behind-the-scenes” aspects of mRNA-1010:
**** One: That Moderna will do whatever it takes to get mRNA-1010 approved — including being “more assertive” with the FDA, and arranging it so that the FDA will “work with” Moderna.
**** Two: That Moderna will do a “post-marketing study” on mRNA-1010 AFTER it is FDA-approved — meaning that ANYONE who takes mRNA-1010 AFTER the FDA approves it is being treated as a “human lab rat” by Moderna. This is the same type of situation that occurred when the FDA approved Moderna’s modRNA COVID-19 bioweapon “vaccine”, mRNA-1273.
**** Three: That Moderna considers the FDA approval of mRNA-1010 as the “stepping-stone” to what the company appears to believe is the “Holy Grail” of modRNA-based “vaccines” — the company’s “combo” modRNA-based influenza + modRNA-based COVID-19 “vaccine”, mRNA-1083 (the clinical trials for which have been completed)
There are, in addition, two other aspects to the situation: What appears to be pressure on Dr. Martin Makary, MD (FDA Commissioner) to “knuckle under” to what Moderna wants; and, what appears to be a coordinated campaign to have Dr. Vinay Prasad removed from his position as CBER division director at the FDA. In Yours Truly’s opinion, these relate to the “sudden arrangement” of the FDA “Type A” meeting between Moderna representatives and the FDA almost immediately after the Refusal to File was issued by Dr. Prasad. Please see the screenshots, below, from https://www.biospace.com/fda/makary-prasad-under-fire-as-fda-turmoil-reaches-president-trump, Heather McKenzie, 20 February 2026:



Peter Pitts, by the way, was an FDA employee whose position was that of a “senior communications and policy adviser” of the agency (https://www.centerforbiosimilars.com/authos/peter-pitts.) It appears that Mr. Pitts and CMPI are involved in a campaign to have Dr. Prasad removed from the FDA.
In Yours Truly’s opinion: Dr. Martin Makary is compromised, due to his involvement with BIO.org/, the group that is implementing a campaign to have HHS Sec. Robert F. Kennedy, Jr., removed; and, Dr. Prasad is compromised, due to what appears to be lack of support within FDA, combined with innuendoes regarding his professional behavior at the agency.
In Yours Truly’s opinion, It appears that there is a combination of chaos, mistrust, internecine feuding, and active resistance going on within the FDA; plus, exterior pressure on the agency from companies and other entities to restore the FDA back to the “good old days”, when drugs and other biologics were authorized and approved in what may be called a kind of “rubber-stamp” process. It also appears that HHS Sec. Kennedy, Jr., is either being “kept out of the loop” regarding what is going on with the FDA; or, cannot, for whatever reason, root out personnel within the FDA who are fomenting trouble.
Yours Truly now turns to the Moderna-funded published paper on mRNA-1010, which was cited by Dr. Prasad as the reason for his issuing the Refusal to File letter to the company: https://doi.org/10.1038/s41541-025-01340-5. “mRNA-1010 influenza vaccine elicits distinct and enhanced humoral immunity compared to adjuvanted inactivated vaccines.” Paulina Kaplonek, et al. 15 December 2025. Moderna completely funded this study; all of the paper’s co-authors are either current or former Moderna employees; and, the current Moderna employees who are co-authors of the paper are also stockholders in the company. All of these in and of themselves, in Yours Truly’s opinion, represent massive conflicts of interest that should, under normal circumstances, disqualify the paper from any serious consideration by the FDA for a BLA application review. This is aside from the flaws in the clinical trial NCT05397223, on which the paper was based (more on this below in today’s offering.) Please see the screenshots from this paper, below:

Note the use of the word, “may.” This word is used in several areas of the paper, as in, “may elicit”; “may reflect”; and, “may induce.” In other words, Moderna does not KNOW if mRNA-1010 can actually be helpful against influenza. The company is guessing that it “may.” However, the company is still pursuing the BLA application with the FDA to get the “vaccine” approved — without having provided ANY proof that the “vaccine” actually does what it is “supposed” to do — which is, to prevent influenza infection better than the licensed influenza “vaccines” already on the market.
The following screenshots from the paper relate to how mRNA-1010 works, including: Figure 4B and Figure 4D, which show that the “vaccine” minimizes the crucial natural body’s activity of IgM cells (the “recognize an enemy and signal the other cells” immune cells); which show that there is an apparent increase of IgG4 cells (the “tolerate but never clear” cells); and, which appear to hint at mRNA-1010 being used as a kind of “universal influenza vaccine” candidate:
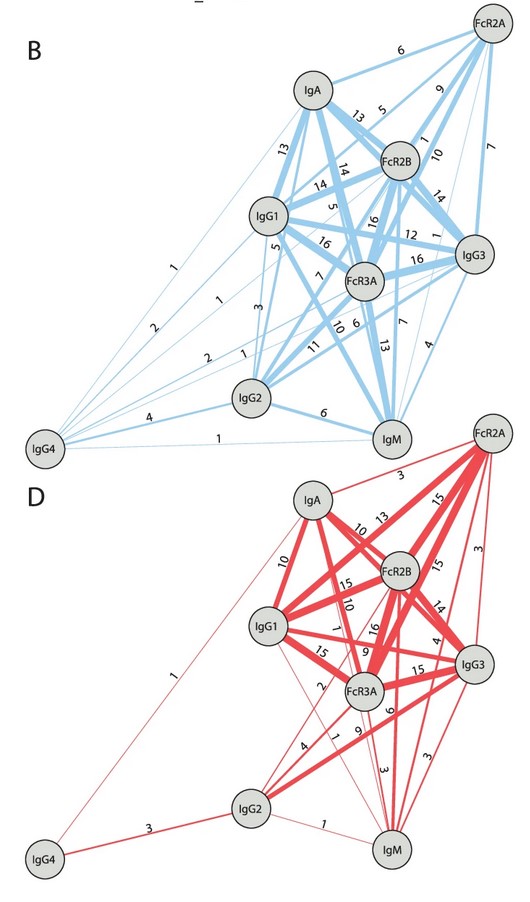
The blue image is the results of the “comparator influenza vaccine”, FLUAD; the red image is the results of mRNA-1010.
Then, from the section that discusses the results of Figure 4B and Figure 4D:

The Moderna paper co-authors did not prove that mRNA-1010 provides mucosal protection from influenza.
Following is a screenshot from the paper regarding the non-involvement of IgM cells induced by mRNA-1010:

Followed by the “hint” that mRNA-1010 may be used as a “universal influenza vaccine” candidate:

Finally, the Acknowledgements section, and the Ethics Declarations section, of the paper:


As shareholders in Moderna, the above employees (and co-authors of the paper on mRNA-1010) stand to make money off the sale and use of this “vaccine.”
**** Regarding the clinical trial which was the foundation for the Moderna paper that was published on 15 December 2025, NCT05397223, details of which are found here: https://clinicaltrials.org/study/NCT05397223. This clinical trial did NOT have a true saline placebo Control Group. Per the Clinical Trials website, there are TWO separate parts to the study. In Part One, study subjects received injections of: of mRNA-1345 (a modRNA-based “vaccine” against RSV); or, of mRNA-1647 (a modRNA-based “vaccine” against Cytomegalovirus);, or, of mRNA-1273 (the modRNA COVID-19 “vaccine”), all by Moderna. In Part Two, study subjects received injections of either: FLUAD (the “comparator” licensed inactivated influenza vaccine by Seqirus); or, of mRNA-1010. Please see the screenshot, below, from the Clinical Trials website for NCT05397223, the Secondary Outcomes Measures section:
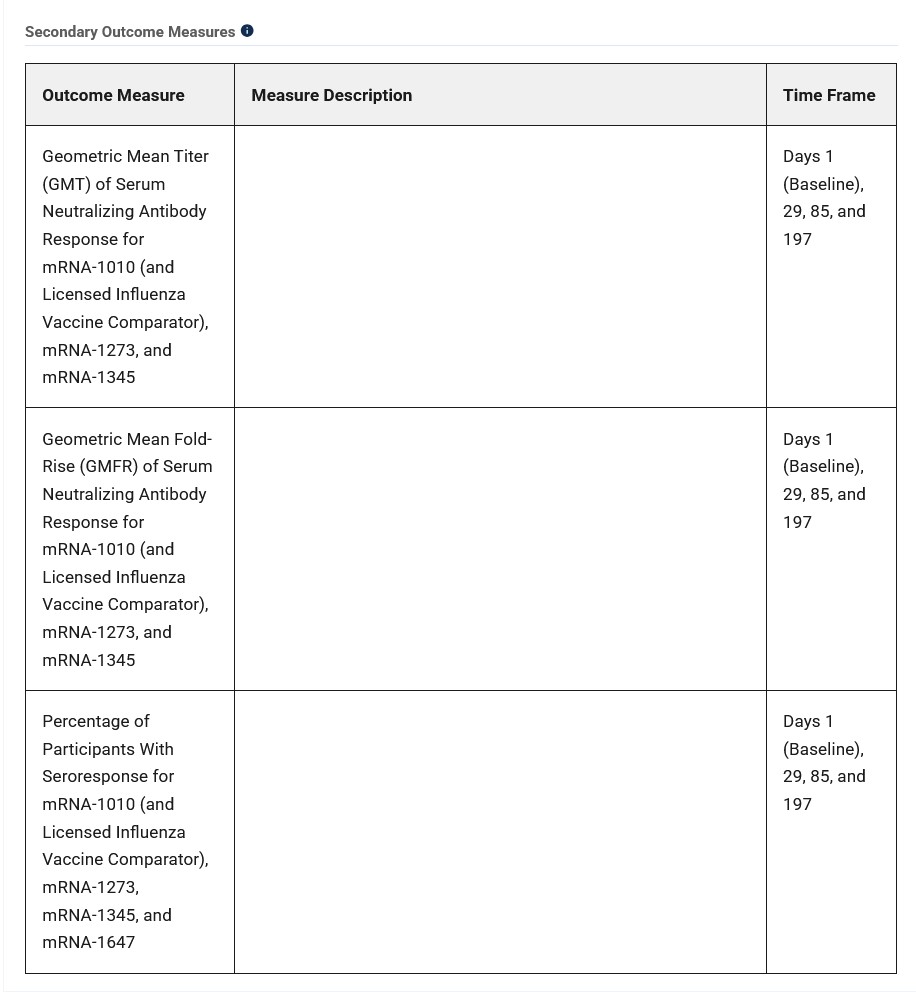
However, there is not a single word in the Moderna-funded paper cited above in which the outcomes for ANY of the “vaccines” used on the study subjects other than FLUAD or mRNA-1010, are found. Nothing for mRNA-1345, for mRNA-1647, or for mRNA-1273. It is unknown if any of these three “vaccines” induced any interactions with either FLUAD or with mRNA-1010. There are “No Results Posted” on the Clinical Trials website for NCT05397223.
Moderna has been in the process of developing and testing mRNA-1010 for the past several years. The company applied pressure, which apparently went all the way up to the Oval Office, in order to force the FDA to reverse course and agree to review the (amended) BLA application for mRNA-1010, despite the flaws of the clinical trial NCT053972723, and despite the Refusal to File letter sent by Dr. Vinay Prasad.
To be continued in Part Two.
THERE IS NO PLACE IN THE HUMAN BODY FOR AN mRNA-BASED, modRNA-BASED, saRNA-BASED, OR taRNA-BASED PRODUCT IN ANY FORM.
Peace, Good Energy, Respect: PAVACA
(intellectual Property Disclaimer and Notice: With the exception of linked items that are found on the internet, the ideas and opinions of today’s offering are by PAVACA. Credit must be given to PAVACA if ideas or opinions in today’s offering are used by other blog writers; by podcasters; or in social or print media.)
