The colorized vintage image of vaccination for today’s header is courtesy of MedPage Today and Google Images.
Health Friday is a series devoted to Big Pharma, vaccines, general health, and associated topics. There are Important Notifications from our host, Wolf Moon; the Rules of our late, good Wheatie; and, certain caveats from Yours Truly, of which readers should be aware. They are linked here. Note: AI-generated items in today’s offering will be cited as such. If readers wish to post AI-generated items in today’s discussion thread, they must cite their source. Thank you.
The Moderna “combination modRNA influenza + modRNA COVID-19 vaccine”, mRNA-1083, also called mCOMBRIAX, has just been approved for use in Europe. mRNA-1083 (mCOMBRIAX) is a combination of the Moderna modRNA multi-strain influenza “vaccine”, mRNA-1010, plus the company’s “other” modRNA COVID-19 bioweapon “vaccine”, mRNA-1283 (mNEXSPIKE.) Yours Truly has written about the mRNA-1010 component of this “combo vaccine” here: https://www.theqtree.com/2026/02/27/health-friday-2-27-2026-open-thread-modernas-mrna-1010-and-the-end-run-around-hhs-sec-kennedy-jr-part-one/; and, here: https://www.theqtree.com/2026/03/06/health-friday-3-6-2026-open-thread-modernas-mrna-1010-and-the-end-run-around-hhs-sec-kennedy-jr-part-two/.
And now, on to the mRNA-1283 (mNEXSPIKE) component of mRNA-1083. Today’s offering is Part One of two:
Moderna’s “other” modRNA COVID-19 bioweapon “vaccine”, mRNA-1283 (mNEXSPIKE), was approved for use in the United States on 30 May 2025. The FDA Approval Letter to Moderna is here: https://www.fda.gov/media/186740/download. The Approval Letter for mNEXSPIKE was signed by David C. Kaslow, MD, Director of the Office of Vaccines Research and Review (a subdivision of FDA’s CBER department [Center for Biologics Evaluation and Research]). Dr. Kaslow is a Biden-era “holdover” at the FDA; he has been with CBER since 11 October 2022.
It appears that Dr. Kaslow relied solely on the “data” that was given to the FDA by Moderna regarding the “safety and efficacy” of mRNA-1283, prior to that agency’s granting approval of the BLA (Biologics License Application) that Moderna submitted for the product. In addition, it appears that Dr. Kaslow believed that the “data” that Moderna supplied to the FDA was sufficient enough to warrant a bypass of the review process by the VRBPAC group of the FDA (Vaccines and Related Biological Products Committee.) Please see the screenshot from Dr. Kaslow’s Approval Letter to Moderna for mRNA-1283 (mNEXSPIKE), below:

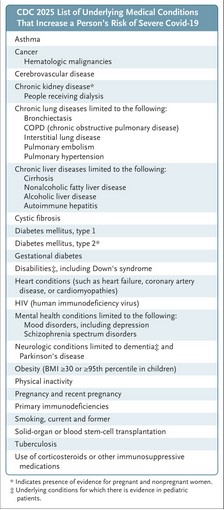
By the way, on 20 May 2025, Dr. Martin Makary (FDA Commissioner) and Dr. Vinay Prasad (Director of the FDA’s CBER division) published an “opinion piece” in the New England Journal of Medicine that purported to be their “opinions” regarding a “new approach” to COVID-19 “vaccination” in the United States. However, this was not really an “opinion piece” — it was implemented as FDA policy. This article by Dr. Prasad and Dr. Makary was published just 10 days before Dr. Kaslow sent the Approval Letter to Moderna for mRNA-1283 (mNEXSPIKE.) Did Dr. Prasad and Dr. Makary know in advance that Dr. Kaslow was going to issue this Approval Letter? Did HHS Secretary Kennedy, Jr., know? The “opinion piece” is here: https://doi.org/10.1056/NEJMsb2506929. “An Evidence-Based Approach to COVID-19 Vaccination.” Vinay Prasad, MD, MPH, and Martin Makary, MD, MPH. 20 May 2025. Below are two screenshots from this article: from the text; and, the Figure 2, which lists the “groups at severe risk from COVID-19.” The persons who would fall under one or more of the categories listed in Figure 2, even if these persons are under age 65, would “qualify” for “vaccination” with mNEXSPIKE:

The Package Insert for healthcare professionals for mRNA-1283 (mNEXSPIKE) is here: https://www.fda.gov/media/186738/download. Please see the screenshots of the following sections of this Package Insert, below: first, section 11 Description; then, section 12 Clinical Pharmacology, and section 13 Nonclinical Toxicology:


It would appear that Dr. Kaslow was comfortable in approving an injectable by Moderna (mRNA-1283, also called mNEXSPIKE) that contains the dangerous lipid nanoparticle SM-102; that contains at least PEG-related element (polyethylene glycol); and, that was not tested for carcinogenicity (the potential to cause cancer), for genotoxicity (the potential to damage genes, or to cause changes to genes, either of which can also cause cancer), or for impairment of male fertility.
It would also appear that Dr. Kaslow was comfortable with approving this modRNA COVID-19 bioweapon “vaccine” (mRNA-1283, also called mNEXSPIKE), which is purported to “prevent infection of COVID-19” — but, which claim has been proven to be wrong. From the FDA Package Insert for mNEXSPIKE:

The “prevent coronavirus disease 2019 (COVID-19)” statement is still being used by the FDA on COVID-19 bioweapon “vaccines” Package Inserts, despite the fact that Shrestha, et al., demonstrated in 2023 that “fully up-to-date vaccinated” Cleveland Clinic employees were more likely to become COVID-19 infected than those who were not “fully up-to-date” with COVID-19 “vaccination” (https://doi.org/10.1101/2023.06.09.23290893. “Risk of Coronavirus Disease (COVID-19) Among Those Up-to-Date and Not Up-to-Date on COVID-19 Vaccination.” Nabin K. Shrestha, et al. 13 June 2023.) Please the screenshot from this paper, below:

Why aren’t the Moderna “original” modRNA COVID-19 bioweapon “vaccine”, mRNA-1273 (SPIKEVAX), and the Moderna modRNA COVID-19 bioweapon “vaccine”, mRNA-1283 (mNEXSPIKE), the same? Please see the screenshot, below, from this article (https://www.drugs.com/medical-answers/what-difference-between-spikevax-mnexspike-3580787/, “What is the difference between SPIKEVAX and mNEXSPIKE?”, 2 September 2025:

However, regarding mNEXSPIKE, there apparently is a “blurred line” as to the use of it in persons under age 65 who do not have an “underlying medical condition.” Please see the screenshot below, from this article: https://health.mountsinai.org/blog/heres-what-you-need-to-know-about-the-new-covid-19-vaccines/, 23 September 2025; the advice is from Dr. Bernard Camins, MD, MSc, of Mount Sinai Hospital, in the “FAQ” section:

Note the language above, from Dr. Camins, regarding the ability of pharmacists to “prescribe vaccines off-label.”
Why is the N-terminal domain of the SARS-CoV-2 virus so important to Moderna that the company crafted a modRNA COVID-19 bioweapon “vaccine”, mNEXSPIKE, to utilize it? The N-terminal domain area is the “closest” (in terms of placement) to the receptor-binding domain (RCB) on the SARS-CoV-2 virus code. The N-terminal domain is also close to the S1/S2 cleavage site on the virus code. The receptor-binding domain is the part of the virus code that “makes sure” that the “payload” of the “vaccine” binds itself (enters into) the cells of the “vaccinated” person. These, in this writer’s opinion, make the N-terminal domain a “prime target” for the development of modRNA COVID-19 bioweapon “vaccines” that do not need to include the entire virus code, but still can “deliver the payload” through the receptor-binding domain mechanism. Please see: https://doi.org/10.3389/fcimb.2020.587269. “SARS-CoV-2: Structure, Biology, and Structure-Based Therapeutics Development.” Mei-Yue Wang, et al. 24 November 2020. Figure 1.) This is in contrast to the other modRNA COVID-19 bioweapon “vaccines” (by Moderna and by Pfizer-BioNTech) that contain the entire spike protein sequence (S1 and S2 areas.)
The issue regarding developing a modRNA COVID-19 bioweapon “vaccine” that only utilizes the N-terminal domain and the receptor-binding domain appears to have been determining exactly what “binding pocket” of the N-terminal domain would work. Scientists had been experimenting with the N-terminal domain to find such a “binding pocket”: for example, as described in this paper, from 2022: https://doi.org/10.1016/j.csbj.2022.11.004. “Structural and energetic analyses of SARS-CoV-2 N-terminal domain characterise sugar binding pockets and suggest putative impacts of variants on COVID-19 transmission.” Jonathan Lees, et al. 17 November 2022. Please see the screenshots from this paper, below; first, the Abstract; followed by the Graphical Abstract:

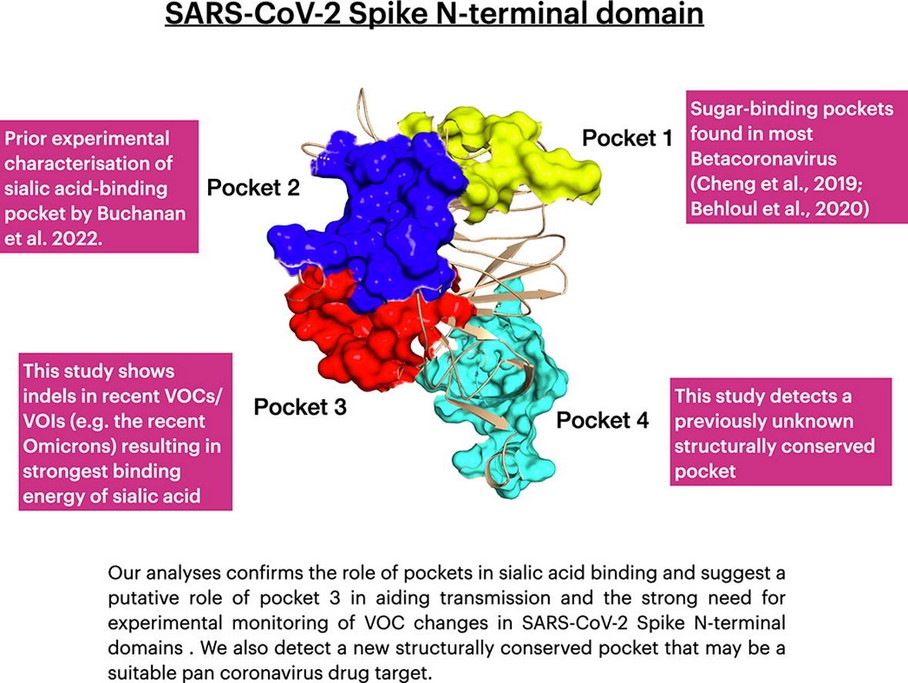
It appears that Moderna did find such an N-terminal “binding pocket” to use for the development of mNEXSPIKE. And, it appears that the company’s decision to “harness” the mechanism to the S1 area of the SARS-CoV-2 spike protein, as opposed to using the entire S1/S2 sequence, is due to the fact that the S1 area is that which appears to be the most “effective” in regards to the Delta and Omicron variants of the virus. Please refer to: https://pubmed.ncbi.nlm.nih.gov/38034565. “SARS-CoV-2 spike protein S1 subunit induces potent neutralizing responses in mice and is effective against Delta and Omicron variants.” Tarlan Mamedov, et al. 14 November 2023. Please see the screenshot, below, of the Conclusion from this paper:

Note that Mamedov, et al., used a tobacco plant to produce the S1 protein from SARS-CoV-2, which they then tested against Delta and Omicron virus variants.
In this writer’s opinion, mRNA-1283 (mNEXSPIKE) cannot be thought of as “mRNA-1273 (SPIKEVAX) Lite.” mNEXSPIKE contains the S1 spike protein of an Omicron variant of SARS-CoV-2. mNEXSPIKE contains the dangerous lipid nanoparticle, SM-102, that mRNA-1273 (SPIKEVAX) also contains. mNEXSPIKE contains N1-Methylpseudouridine (which will destroy the RNA of the “vaccinated” body’s natural Uridine, replacing it with a lab-created chemical compound that has no natural, beneficial mechanism for the body), which mRNA-1273 (SPIKEVAX) also contains. In this writer’s opinion, the only difference between mRNA-1273 (SPIKEVAX) and mNEXSPIKE is that the latter “vaccine” does not contain the S2 area of the spike protein.
To be continued in Part Two.
THERE IS NO PLACE IN THE HUMAN BODY FOR AN mRNA, A modRNA, AN saRNA, OR A taRNA PRODUCT OF ANY KIND, IN ANY FORM. THERE MUST, FIRST, BE MUCH MORE RESEARCH INTO THESE GENE-ALTERING THERAPY PLATFORMS AND PRODUCTS. THERE MUST, SECOND, BE IRREFUTABLE PROOF THAT THESE GENE-ALTERING THERAPY PLATFORMS AND PRODUCTS ARE TRULY “SAFE AND EFFECTIVE.”
Peace, Good Energy, Respect: PAVACA
(Intellectual Disclaimer and Notice: Other than URLs and related items available on the Internet, the ideas and opinions of today’s offering are by PAVACA. Credit must be given to PAVACA if the ideas and opinions of today’s offering are used by other blog writers, by podcasters, or in social or print media.)
