The colorized vintage image of vaccination for today’s header is courtesy of MedPage Today and Google Images.
Health Friday is a series devoted to Big Pharma, vaccines, general health, and associated topics. There are Important Notifications from our host, Wolf Moon; the Rules of our late, good Wheatie; and, certaincaveats from Yours Truly, of which readers should be aware. They are linked here. Note: AI-generated items in today’s offering will be cited as such. If readers wish to post AI-generated items in today’s discussion thread, they must cite their source. Thank you.
The Moderna “combination modRNA influenza + COVID-19 vaccine”, mRNA-1083, also called mCOMBRIAX, has just been approved for use in Europe. mRNA (mCOMBRIAX) is a combination of the Moderna modRNA multi-strain influenza “vaccine”, mRNA-1010, plus the company’s “other” modRNA COVID-19 bioweapon “vaccine”, mRNA-1283 (mNEXSPIKE.) Yours Truly has written about the mRNA-1010 component of this “combo vaccine” here: https://www.theqtree.com/2026/02/27/health-friday-2-27-2026-open-thread-modernas-mrna-1010-and-the-end-run-around-hhs-sec-kennedy-jr-part-one/; and, https://www.theqtree.com/2026/03/06/health-friday-3-6-2026-open-thread-modernas-mrna-1010-and-the-end-run-around-hhs-sec-kennedy-jr-part-two/. Part One of the discussion of the mRNA-1283 (mNEXSPIKE) component is here: https://www.theqtree.com/2026/03/13/health-friday-3-13-2026-open-thread-meet-modernas-mrna-1283-mnexspike-the-other-component-of-mrna-1083-mcombriax-part-one/.
Today’s offering is Part Two of two regarding mRNA-1283 (mNEXSPIKE.)
The Package Insert for healthcare professionals for mNEXSPIKE is here: https://www.fda.gov/media/186738/download. Below is a screenshot of section 11 Description of this injectable:

Note that the Description does NOT mention that the fact that N1-Methylpseudouridine (either by that name, or by its IUPAC name [its spelled-out chemical components name]) is used in mNEXSPIKE. Nor does it mention that the N1-Methylpseudouridine in mNEXSPIKE serves as the “scaffold” for the mechanism of the “vaccine” — a lab-created element that destroys the RNA of the natural Uridine in the body, replacing it with a compound that does nothing beneficial: in fact, which serves as a “facilitator” in speeding the ingredients of the “vaccine” throughout the entire body; while, at the same time, severely damaging the body’s natural ability (via natural Uridine and its RNA) to regulate mood, help with learning and memory, and assist in normal “gut-brain axis” interactions.
Which leads into the Patent for mNEXSPIKE. Yours Truly will not present the entire Patent. Nor is Yours Truly advising people to read through the Patent document for every drug or “vaccine” they put into their body. However, there are multiple aspects of “the Devil is in the details” in the Patent for mNEXSPIKE regarding the ingredients that “may” be used in formulating this “vaccine” — details that appear to be lacking in the Package Insert. Keep this in mind when reading the following.
There are multiple Moderna-held Patents for mNEXSPIKE (https://www.modernatx.com/en-US/patents.) The most recent Patent is here: https://patents.google.com/patent/US12508278B2/en. The Title of the Patent is: “Lipid nanoparticle compositions and methods of formulating the same.” This Patent was published on 30 December 2025. An image of the Inventors and Dates section is below:
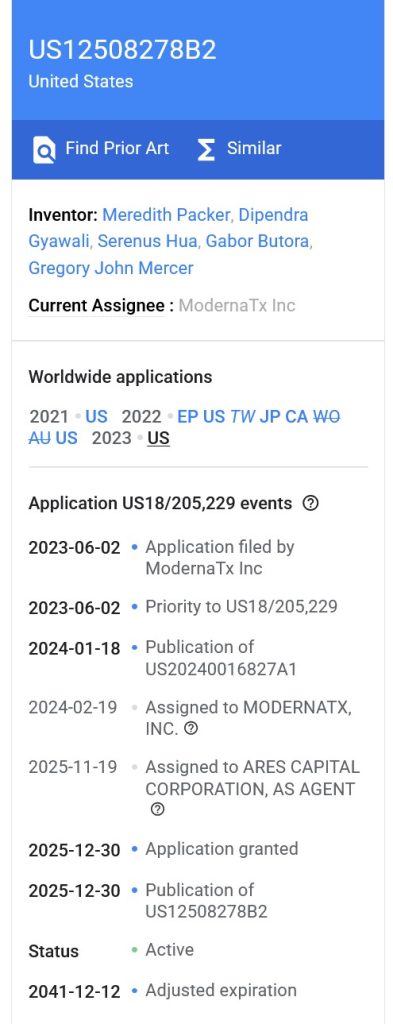
Note that the assigned Agent for this Patent is ARES CAPITAL CORPORATION (https://www.arescapitalcorp.com/), which is private-equity, real estate, and credit asset manager in “global alternative investment” (per Wikipedia.)
The mention of N1-Methylpseudouridine as a component of mNEXSPIKE is here in the Patent:

Which is followed by a list, in Claim 1, of twenty-two different “phospholipids” that Moderna “may” choose from in formulating the “vaccine.” Which list is then followed by a list, also in Claim 1, of nine different PEG-lipids (polyethylene glycol lipids) that Moderna “may” choose from in formulating the “vaccine.” The question here is: Are there different “phospholipids” and/or different “PEG-lipids” being used in different batches of mNEXSPIKE other than the ones listed in the section 11 Description of the Package Insert for the “vaccine”? Recall, from Part One regarding mNEXSPIKE, that the FDA Approval Letter that was sent to Moderna for this injectable specifically stated that the agency relied ONLY on the “data” and other information that Moderna gave to the FDA — there was no independent testing or analysis performed.
Then, there is the lipid nanoparticle (LNP) in mNEXSPIKE, which in the section 11 Description is SM-102. However, in the Patent document, there appear to be multiple types of LNPs from which Moderna “may” choose in formulating the “vaccine.” In addition, there are listed multiple percentage ratio ranges of these LNPs in multiple types of combinations with the PEG-lipids and/or with “non-cationic lipids.” These are in the Patent document under 1. Lipid Nanoparticle Compositions. The question here is: Are there then multiple “variations” of these formulations that would appear in different batches of mNEXSPIKE?
Then, there are the “Adduct Impurities” that are permitted in the formulation of mNEXSPIKE. Please see the screenshot, below, from the Patent, the section 1. Lipid Nanoparticles:

“Adduct impurities” — think the “loose DNA” in the Pfizer-BioNTech and in the Moderna modRNA COVID-19 bioweapon “vaccines.” Think the “Process 2” manufacturing method — and that “culturing bath” of lab-created E. coli derivative that is part of this process — that these companies use as part of modRNA COVID-19 bioweapon “vaccines” production. It appears that Moderna is fine with allowing impurities in mNEXSPIKE. One would think that, after more than five years of manufacturing modRNA COVID-19 bioweapon “vaccines”, the company would have come up with a way to remove impurities; or, at least, to reduce the percentage “allowed” in the “vaccine” to as nearly zero as possible, from the product before putting it on the market — and not allowing a range of percentage amounts of impurities to remain in the product.
Following are screenshots from the Patent US12508278B2 for mNEXSPIKE. Yours Truly believes it appears that Moderna may have submitted “data” to the FDA for approval of this “vaccine” based on only the limited set of ingredients that the company sent “data” about — while, at the same time, the Patent for the “vaccine” contains multiple lists of multiple variations of these ingredients from which the company “may” choose to formulate batches of the “vaccine.” In other words — is a “How Bad is My Batch?”-type of scenario being set up?
The ratio of lipid nanoparticles (LNPs) to the mRNA in mNEXSPIKE is allowed to vary:

The type of pseudouridine used in mNEXSPIKE is allowed to vary:

Which indicates that another pseudouridine OTHER than N1-Methylpseudouridine “may” be used in mNEXSPIKE.
The company reserves the right to substitute codons in the manufacture of mNEXSPIKE. Codons are three-nucleotide sequences of a strand of either DNA or of RNA:

The “rationale” for codon replacements:

Forms of administration for mNEXSPIKE:

Note that the Patent states that the “vaccine” can cross the Blood-Brain Barrier.
In summary: It appears that Moderna, in giving the FDA “data” involving the ingredients listed on the Package Insert for mNEXSPIKE, for which “data” the FDA granted “full approval” last year, did not inform the FDA that the Patent for the “vaccine” contains multiple variations for the ingredients used in the product; multiple variations of formulations for the product; and multiple forms of administration of the product. It also appears that Dr. David Kaslow, who signed the FDA Approval Letter to Moderna for mNEXSPIKE, either did not read the Patent document for this product; or, did not have it summarized for him to read; or, read either or both of the latter and was fine with it.
**** AND HERE’S THE REASON BEHIND THE DISCUSSION OF THE PATENT FOR mRNA-1283 (mNEXSPIKE) AND THE SCREENSHOTS FROM THE PATENT: It appears that Moderna is using clinical trials study subjects as HUMAN LAB RATS in their two latest Clinical Trials of “variant formulations” of mRNA-1283 (mNEXSPIKE.)
Clinical Trial NCT07266558 (https://clinicaltrials.gov/study/NCT07266558.) Please see below: screenshots of the study Overview, and of the Trial Description – Intervention and Study Arms section from the Researcher View:


Note the language, “Variant Formulation.” Note ALSO that this Clinical Trial is for HEALTHY persons age 50 – 64 years with NO underlying conditions — this appears to be the “setup” to getting mNEXSPIKE approved by the FDA for HEALTHY persons under age 65. Note also that mRNA-1273 (SPIKEVAX) may be given to a study subject.
Clinical Trial NCT07089706 (https://clinicaltrials.gov/study/NCT07089706.) Please see below: screenshots of the study Overview, and of the Trial Description – Intervention and Study Arms from the Researcher View:


Note that there is NO saline Placebo group in the clinical trial above. Note also the very small study subject pool (832 persons.)
What are in these “variant formulations” of mRNA-1283 (mNEXSPIKE) that the study subjects will receive in the clinical trials listed above? Refer back to the screenshots from the Patent for mRNA-1283 in today’s offering. Look at the multiple combinations of lipid nanoparticles to modRNA, the multiple types of pseudouridines, the multiple percentage ranges of allowed “adduct impurities” that can be used in formulations of this injectable. Also — Why is there NO saline Placebo group in NCT07089706?
Does anyone at the FDA understand what’s going on here?
THERE IS NO PLACE FOR AN mRNA, A modRNA, AN saRNA, OR A taRNA PRODUCT IN THE HUMAN BODY, IN ANY FORM. THERE MUST, FIRST, BE MUCH MORE RESEARCH PERFORMED ON THESE GENE-ALTERING THERAPY PLATFORMS AND PRODUCTS. THERE MUST, SECOND, BE IRREFUTABLE PROOF THAT PRODUCTS MADE USING THESE THERAPY PLATFORMS AND PRODUCTS ARE TRULY “SAFE AND EFFECTIVE.”
Peace, Good Energy, Respect: PAVACA
(Intellectual Disclaimer and Notice: Other than URLs and related items available on the Internet, the ideas and opinions of today’s offering are by PAVACA. Credit must be given to PAVACA if the ideas and opinions of today’s offering are used by other blog writers, by podcasters, or in social or print media.)
