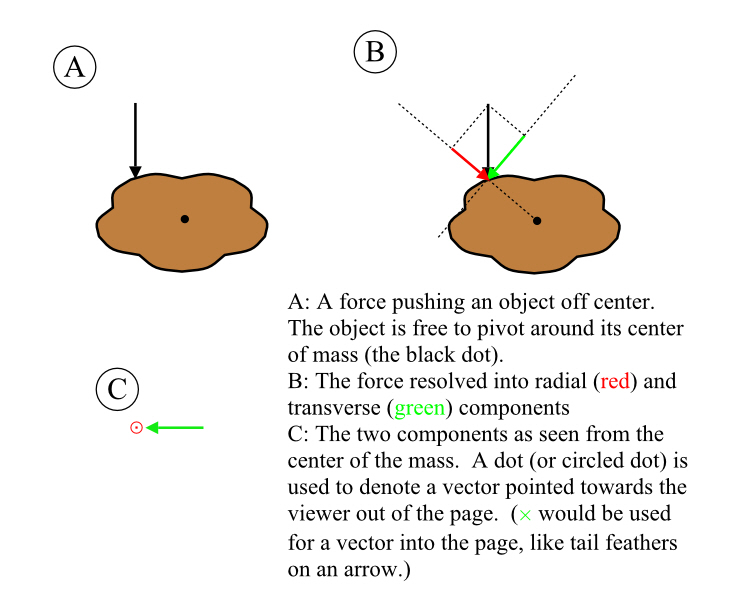
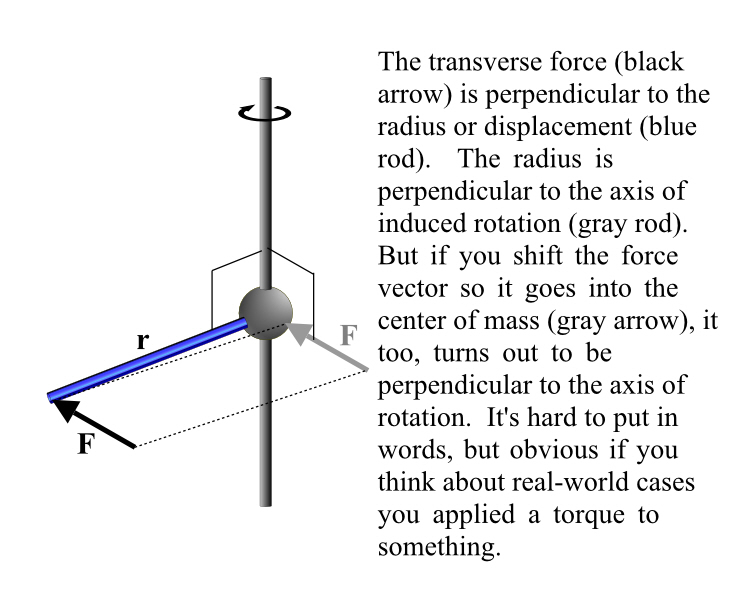
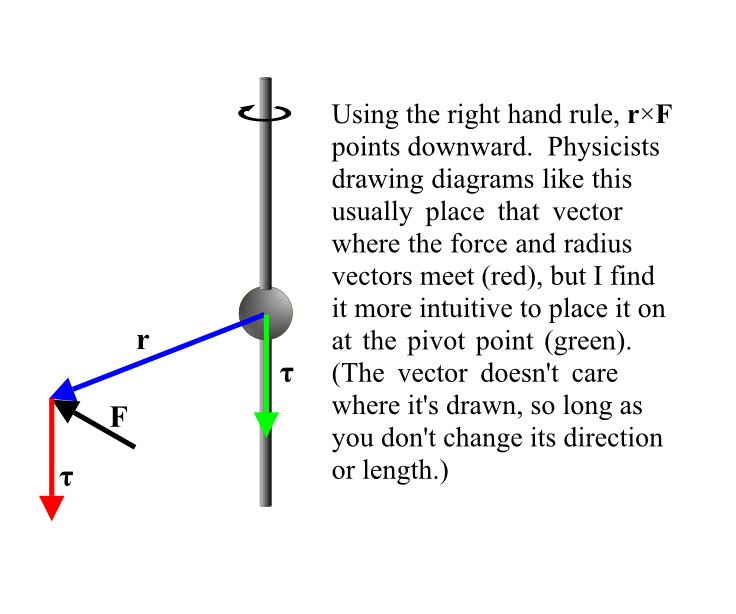
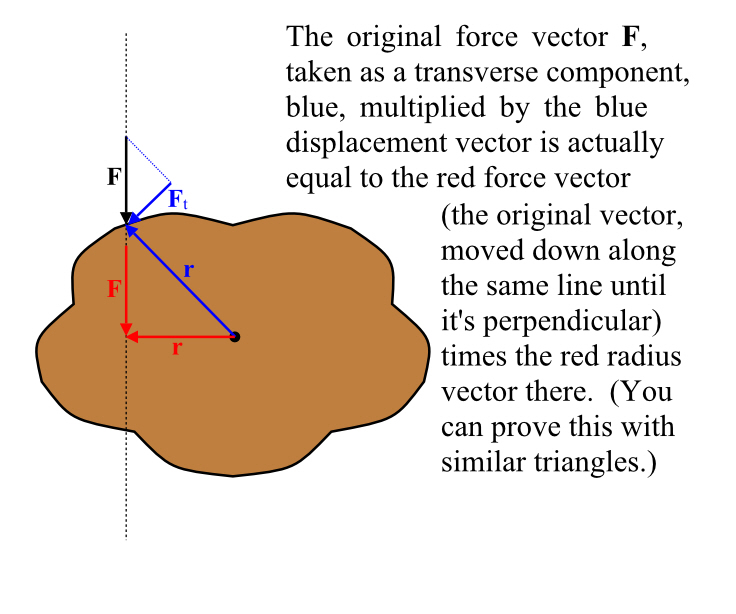
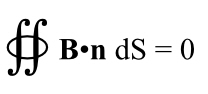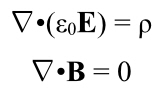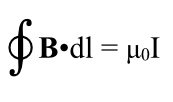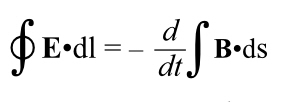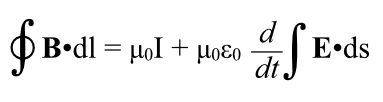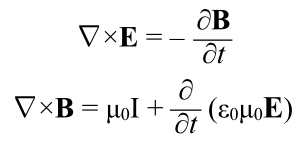Justice Must Be Done.
The prior election must be acknowledged as fraudulent, and steps must be taken to prosecute the fraudsters and restore integrity to the system.
Nothing else matters at this point. Talking about trying again in 2022 or 2024 is hopeless otherwise. Which is not to say one must never talk about this, but rather that one must account for this in ones planning; if fixing the fraud is not part of the plan, you have no plan.
Kamala Harris has a new nickname since she finally went west from DC to El Paso Texas: Westward Hoe.
Colorado Statehood Day

Once upon a time, this actually mattered. Colorado became a state on August 1, 1876. Because of the year, it is known as the “Centennial State” and I remember, buried in the Bicentennial hype, Centennial hype as well. We even managed to get Congress to order the mint to strike us a medal. (If you took a tour of the Denver mint and bought the souvenir set, you got a cent, nickel, dime, quarter, half dollar, and one of these, all of course struck at the Denver mint. [Yes, you did not get the bicentennial Ike dollar.])


Of course today this matters not one damn bit. I was once proud of this state, and am still proud of what it once was. But now August 1 is just a date when many absolutely stupid or outright tyrannical laws passed by our so-called “representatives” convened in the City and Cesspit of Denver, become effective.
Do I blame the Democrats? Yes. Do I blame the Republican RINOs? Yes. Do I blame the Libertarian Party for pulling enough votes from the Republicans that Democrats started getting elected? No. (Many republicans do blame the LP for that.) If the Republican party had done its f*cking job instead of continuing to fellate the Left even when it had veto-proof majorities in the state legislature in the mid 1980s, there’d have been no need in anyone’s mind for a Libertarian Party. [Which, by the way, was founded in Colorado Springs…]
If you think I am just a wee bit angry about this, well, it likely seems that way superficially, even though in reality, my attitude is completely different: I am a great deal angry about this. Just thought I’d clear up any possible confusion.
Lawyer Appeasement Section
OK now for the fine print.
This is the WQTH Daily Thread. You know the drill. There’s no Poltical correctness, but civility is a requirement. There are Important Guidelines, here, with an addendum on 20191110.
We have a new board – called The U Tree – where people can take each other to the woodshed without fear of censorship or moderation.
And remember Wheatie’s Rules:
1. No food fights
2. No running with scissors.
3. If you bring snacks, bring enough for everyone.
4. Zeroth rule of gun safety: Don’t let the government get your guns.
5. Rule one of gun safety: The gun is always loaded.
5a. If you actually want the gun to be loaded, like because you’re checking out a bump in the night, then it’s empty.
6. Rule two of gun safety: Never point the gun at anything you’re not willing to destroy.
7. Rule three: Keep your finger off the trigger until ready to fire.
8. Rule the fourth: Be sure of your target and what is behind it.
(Hmm a few extras seem to have crept in.)
Spot Prices
All prices are Kitco Ask, 3PM MT Friday (at that time the markets close for the weekend).
Last week:
Gold $1802.80
Silver $25.26
Platinum $1065.00
Palladium $2760.00
Rhodium $19,500.00
This week, markets closed for the weekend at 3:00 PM Mountain Time
Gold $1815.20
Silver $25.56
Platinum $1053.00
Palladium $2747.00
Rhodium $19,500.00
Gold broke out and up into the 1830s this week but much of that gain was lost by close on Friday. Silver is up a bit too, the PGMs however are down (or steady).
Part XII: The Rest of Special Relativity
Introduction
I had to cut Part XI, which introduced the four ground-breaking “boom” papers Albert Einstein published in 1905, short two weeks ago because I simply ran out of time. Our lupine host might be willing to tolerate a post as much as twelve hours late, but I don’t care to do that.
So I’m going to pick up where I left off.
But first I’m going to drag out a soapbox (Stop that twitching eye, it’s at rest in our reference frame!) and explain a couple of things.
I have seen people criticize Einstein for not being a real scientist, on the grounds that he didn’t do real experiments, but rather a lot of “though experiments.” I walked you through a few of them last time (all those examples with the moving trains).
You’re invited to imagine that Einstein did a bunch of thought experiments, and that other scientists accepted them as Holy Writ and that is how the theory of special relativity became accepted as being true.
But Einstein didn’t rely on his thought experiments. And neither did anyone else.
Science is like any other line of work. People specialize. Scientists can be divided into two broad groups, theoreticians and experimentalists. And of course there’s usually at least some of each in a scientist. But the archetypal theoretician is someone who shouldn’t even be allowed to touch a screwdriver lest he put out his own (or someone else’s) eye with it. Whereas many scientists are quite handy with tools and design and build very intricate equipment. And this distinction doesn’t just exist in pure science. I recall overhearing a fellow (engineering) student complaining to a prof about what a klutz he (the student) was in the lab courses and wondering if he were cut out for this line of work and the professor practically fell all over himself explaining that no, there was plenty of room in engineering for people who were good with the theory. (Those would be the sorts who design things and do not build the prototype!) It was pretty obvious to me from his talk that this particular professor was himself one of the more theoretical types. (He didn’t teach one of the labs!)
Einstein started by trying to explain things prior experiments had shown, did his “thought experiments” to come up with a theory, and put the theory out there…to stand or fall as people did more experiments. He was about as pure Theoretician as one can imagine, but he himself and everyone around him knew that even the most elegant theory was useless until validated by experience.
As I alluded to Einstein’s paper on the photoelectric effect had such sweeping implications about the very nature of reality that it took sixteen years to earn him his sole Nobel prize (he didn’t get his Nobel for relativity). Scientists certainly didn’t take that as Holy Writ, nor did they take anything else Einstein produced as Holy Writ.
Not until experiments upheld it, and it became plain that Einstein’s theories explained them better than anything else. If they hadn’t, he’d never have become known to absolutely everyone.
In 1905 Einstein was a 26 year old clerk in a patent office. He had the requisite credentials in science, but he still had to prove himself as a scientist. But even after he was considered absolutely solid as a theorist, that still didn’t mean that everything he put out there was considered Truth. Not until checked. And even then, there’s always the possibility someone will do some experiment somewhere that will put a gigantic crack into one of Einstein’s theories. And a good scientist knows this.
And on a related but different topic:
General relativity is often presented as though Einstein started with the Michelson-Morley experiment (which failed to detect any difference in light speed in a vacuum regardless of direction, even though Earth was presumed to be moving through an aether that serves as the medium light traveled through). This wasn’t actually the case; he was trying to reconcile a seeming inconsistency or two in electromagnetic theory (more about which, soon). But let’s set that aside. I’ve personally known people who can’t abide special (and especially general) relativity because, they claim, it “reifies space” (makes nothing into something). They don’t like quantum mechanics either, because (as we will eventually see) it’s non-deterministic. Of course some of these people are so confused they conflate relativity and quantum mechanics, accusing relativity of being non-deterministic (it’s quite deterministic–just not in the manner you expect), and so on.
But be that as it may, special relativity has its detractors, and they often start by suggesting a different explanation for Michelson-Morley’s “null” result, which seemed to show there is no aether, no medium for the propagation of light (just like sound requires air to propagate) that we (Earth) are plowing through One I read many years ago was that perhaps the aether is real, but is, locally, being “dragged along” with Earth, basically, “entrained.” So Earth can be moving through the aether, but because some of it is sticking to Earth, Earth is really dragging some bits of the aether through the rest of it. If so then here on the surface of the earth, the aether will seem stationary with respect to us, or us stationary with respect to the aether. So measuring the speed of light in different directions, in the expectation that we’ll find out how fast we’re moving through the medium it propagates through, will return a zero result. However, if we did the experiment far away from Earth, we might just discover that we are moving through an “aether.”
OK, that could indeed be an alternative explanation for the Michelson-Morley experiment.
But that’s not enough. A proposed alternate has to not just explain one thing, the one thing that got the ball rolling on a train of thought (um…pun left there even after I realized it) that became the theory being targeted as well as that theory does. [Note though that Michelson Morley isn’t where Einstein started from…but let’s pretend for the sake of argument that it is.] The alternate had better explain everything else that the target theory explains, as well as if not better than the target theory. And it would be nice if it also explains things the target theory does not, especially things that the target theory actually gets wrong.
If it can’t do this it’s worthless in our current context and can be shelved, perhaps to be brought back in a different context when we learn more, but more than likely, never to be brought back at all.
The entrainment suggestion, if true, would have certain other consequences which are very different from special relativity’s consequences. Those consequences simply aren’t true. It also doesn’t explain time dilation, which is absolutely real, measured in the laboratory, nor length contraction, nor mass deficits (another thing I haven’t got to yet), all of them measurable. So at that point, it’s not worth considering given what we know today.
If Einstein had decided to entertain entrainment as an explanation, and followed that through to its logical consequences, his work would have been worthless, because those consequences wouldn’t match reality.
I’ve beaten up on flat earthers before and I will do it again now. It is possible, in many cases, to come up with a flat earth theory that explains one phenomenon that suggests that the earth is instead almost perfectly spherecal. Differing sun angles at two different places on the earth? Well, that’s because the sun is close enough that parallax puts it in a different direction as seen from those two places. This is an alternative to the round earth theory that says the different sun angles are due to being on two differently oriented parts of the surface of a sphere, looking at a sun that’s far enough away you can approximate it as infinity. But that actually falls apart when you add a third point. And it doesn’t explain how nighttime can exist in some places at the same time as daytime in others. No doubt a sufficiently clever flat earther could conjure something up to explain that (I can’t). But that would be a different flat earth theory, because the particular one I alluded to earlier cannot explain how it can be dark in Tokyo and light in New York City, at the same time.
There is no one flat earth theory that can explain everything that the round earth theory does; and there’s nothing relevant that the round earth theory cannot explain. If one believes the earth is flat because there is a flat earth theory that can explain away everything, their logic is defective if those flat earth theories contradict each other. The mere fact that an alternate explanation can be made for every single thing a currently-accepted theory doesn’t throw that theory into doubt, and cannot unless all of the alternate explanations are the same explanation or at least not inconsistent with each other.
OK, hopefully after all that you have a sense of the rigor to which a proposed alternate theory will be subjected to. And hopefully you recognize that, at least back in the day science was science rather than SJW activism, the currently-accepted theory would not itself be the currently-accepted theory if it had not already run that gauntlet, going up against an older theory. And so on, back to Galileo, who founded the scientific method. (Before that, it was pure theory, pure thought experiment, rarely if ever checked against reality.)
OK, so on to more Special Relativity.
The Doppler Effect
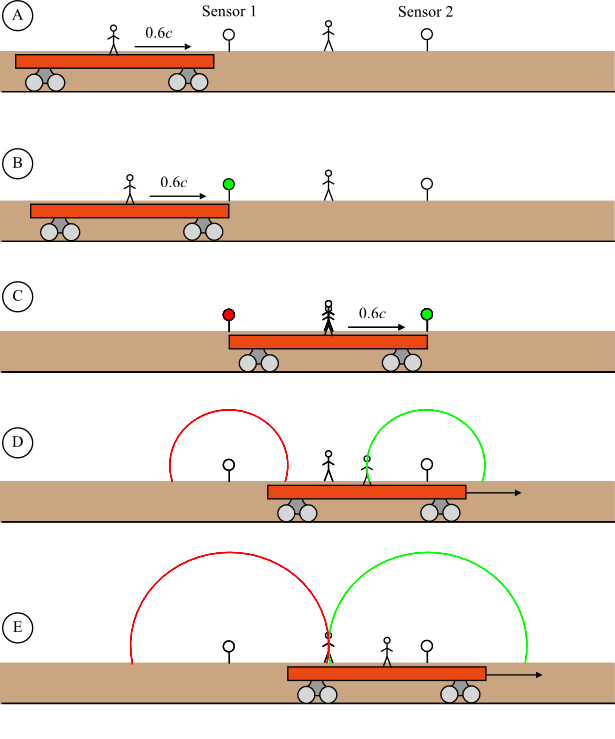
Imagine, if you will, that I am now stepping onto a moving soapbox (so your eyes can start twitching now if they want to).
But for now I’m going to move at a fairly sedate speed, about 76.7 miles per hour, one tenth the speed of sound (at sea level, on a “standard” day with standard temperature and air pressure). And let us assume the air is perfectly still with respect to the ground (which means this is not Wyoming or anywhere on the Great Plains). So I am moving at that speed through the air.
This speed is also 34.288 meters per second. And the speed of sound under these circumstances is 342.88 meters per second.
I strike something with a hammer. The sound from this radiates outward from where the hammer fall happened, at 342.88 m/s, in an ever expanding circle. But it does so through the air, not relative to me. After the first second, the sound has gone 342.88 meters but I have also moved 34.288 meters, so the sound wave in front of me is only 342.88 – 34.288 = 308.592 meters ahead of me. Similarly, the sound wave directly behind me is 342.88 + 34.288 = 376.168 meters away. If at that instant I strike with the hammer again, there will now be two sound waves, expanding outward. They won’t be concentric, the smaller, later wave’s center is 34.288 meters away from the center of the larger, earlier wave.
If you are standing directly in front of me, you will hear the first hammer blow at some time, then you will hear the second hammer blow. But you will not hear them a second apart. Remember that the forward edges of the waves are 308.592 meters apart, not 342.88 meters apart, and that corresponds to a difference of 0.9 seconds.
If I continue with the hammer blows, one second apart, you will hear hammer blows every 0.9 seconds. If you turn that into a frequency, it’s 1 / 0.9 = 1.11111111… hertz (Leftist lurkers: keep writing ones until I grow tired).
If someone else is standing behind me, they will hear hammer blows every 1.1 seconds, for a frequency of 0.90909090 hertz (and the somewhat more intelligent Leftist lurkers can take on the more intellectually challenging job of writing alternating zeros and nines until I grow tired).
A sustained tone is simply many, many pulses every second, and the same thing happens to them as to my hammer blows one second apart. Their spacing gets reduced by one tenth (for people in front of me) or increased by one tenth (for people behind me). That in turn increases/decreases the frequency by 1/9.

This affect was first noticed by lots of people when trains would pass through towns, and blow the train whistle as they went by. They’d hear a certain pitch as the train approached, then the pitch would drop as the train went past, and the train receding into the distance would be blowing a lower note on its whistle. Many thought the engineers were playing some trick with the whistle, but they weren’t (train engineers had better things to do than to make sure they trolled absolutely everyone they saw along the side of the track, with their whistle).
To put this more mathematically:
fheard = ( c / (c + vs) ) femitted
Vs is the velocity of the source through the medium, c is how fast the waves propagate through the medium. Vs should be treated as a positive number if the source is moving away from you, negative if it’s moving toward you. So in our example where vs is 1/10th the speed of sound, c /(c+vs) reduces to 1/1.1 for a source moving away from you, and whatever the frequency I blow, you’ll hear a frequency 0.90909090 times that.
A similar analysis gives a slightly different result if the source is stationary but you are moving towards or away from it:
fheard = ( (c + vr) / c ) femitted
You end up dividing by c/(c+vr) instead of multiplying by it, or alternatively, multiplying by (c+vr)/c, and vr is the receiver’s velocity through the medium, positive when you move toward the source. So if YOU are standing still and making the note and I am travelling towards you on my magic mach 0.1 soapbox, I will hear a frequency 1.1 times what you made, traveling away from you I’d hear a lower pitched frequency, 0.9 times as much.
There is a more general formula covering the case where both you and the source are moving through the medium, at different speeds, but it’s not important here. I’ll give it to you anyway.
fheard = ( (c + vr) / (c + vs) ) femitted
It sort of looks like a combination of the two others, doesn’t it? If you think about it, the two other formulas come from this one, if you set either the receiver’s velocity to zero, or the source’s.
Things get much more interesting if you move at the speed of sound, or faster than it, or if the source and the recipient are not moving directly towards or away from each other.
OK, now to look at special relativity.
Light has a frequency, that frequency, if it’s one our eyes can detect, is a color. Higher frequencies look blue or even purple, lower frequencies will look orange or red.
Wouldn’t it stand to reason that if a light source is moving towards you, it would look bluer, and if moving away, it would look redder?
Yes that makes sense. But wait a minute!
Light doesn’t propagate through a medium. It simply propagates. So all that stuff up above where I derived the Doppler formulae under the assumption that sound propagates through a medium and its your speed relative to the medium that affects what you hear…is crap when applied here.
But nevertheless, light does do Doppler shift. It just doesn’t do so quite the same way. The formula won’t involve your speed relative to the medium (which doesn’t exist), but rather go directly to your speed relative to the source, since that’s the only thing that could possibly matter. There won’t be two velocities built into this, but rather just the relative velocity between the two.
Now it’s:
fseen = [ sqrt( 1 – v2/c2 ) / 1 + v/c ] femitted
v is positive if the seer and emitter are moving away from each other, negative if they are moving toward each other.
Note that our old friend sqrt(1-v2/c2) shows up again, but this time it’s in the numerator, so this is 1/γ this time.
fseen = femitted / γ [ 1 + v/c ]
This formula does not have to be used on just the frequency of light waves. You can apply it to any occurrence that has a regular period. For example you could be travelling away from Earth at close to light speed, and use this to see how far apart it seems that the Earth is at the same spot in its orbit. Since that’s a yearly event (by definition!) you can therefore see how often an event that happens on a certain calendar date will appear to happen from your point of view.
In particular, you can see how often Billy receives Bob’s annual messages (and vice-versa), from our “Twins Paradox” example last time. When I discussed this example, Billy was on a spacecraft headed for Sirius at v/c = 0.8, outbound for the first leg, then stopping and returning. The twins had agreed to send each other messages once a year, and due to the press of time two weeks ago I simply asserted how often the other twin would receive a message. But now we have the mathematical tool to back up my assertion.
This made γ = 1/0.6 or 5/3. 1 +v/c = 1.8, so the denominator above is (5/3)(9/5) = 9/3 = 3, so you divide the emitted frequency by 3.
So as Billy travels away from Bob, any regular pulse (like an annual message from Bob announcing Bob just got a year older) will come in at 1/3 the rate it would arrive if they two weren’t moving with respect to each other. So Billy gets the message once every three years while outbound, as I noted.
On the return trip they approach each other so now you multiply γ (still 5/3) by 1 – 0.8 = 1/5 and get 1/3 which, remember is the denominator, so multiply the once per year frequency of Bob’s messages to Billy, and see that Billy gets three of them a year not one.
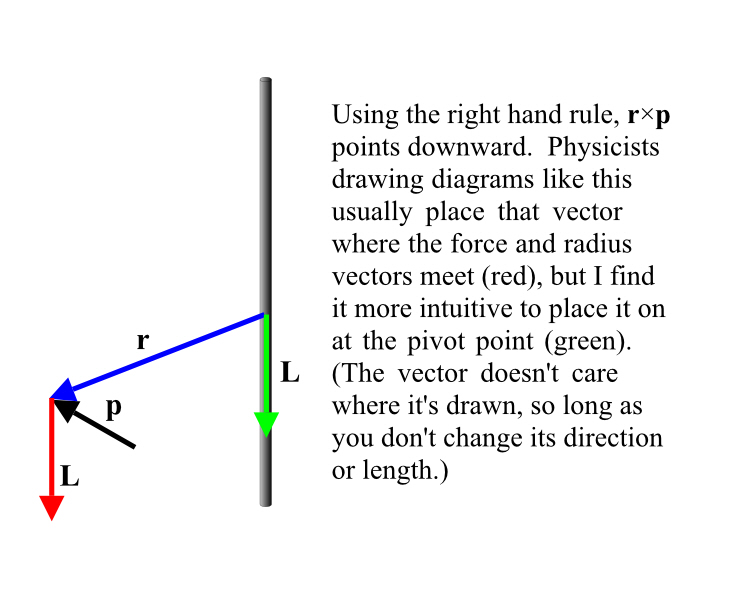
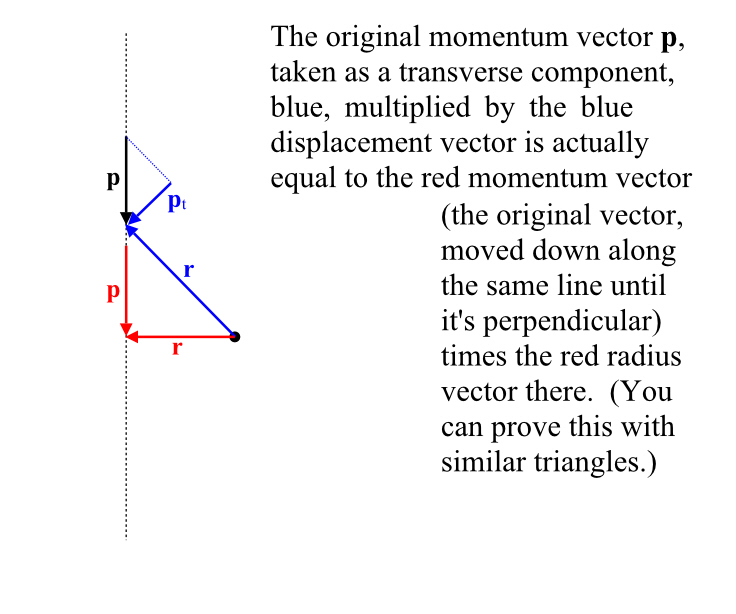
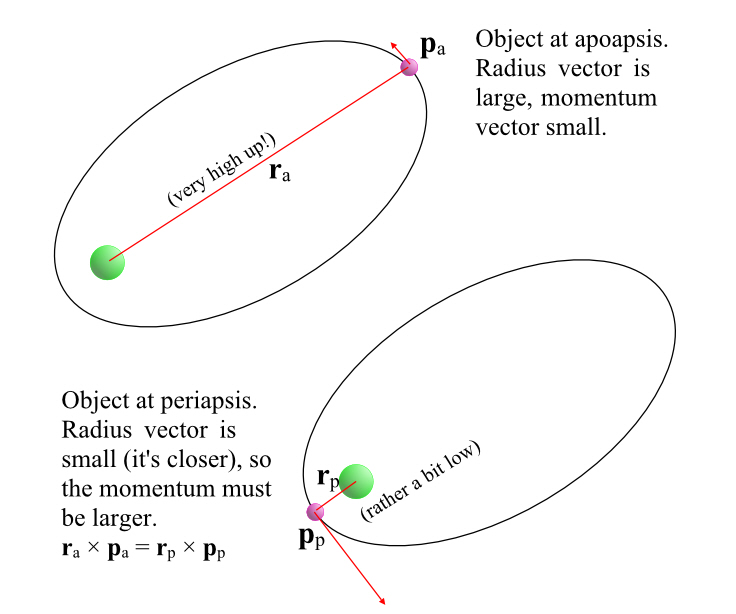
Relativistic Momentum
One consequence of all of this is that, if I am watching a moving person fire a weapon, the velocities do not add up. For example, if Bob were to see Billy fire a phased plasma rifle in the 40 watt range, straight ahead of him, and the beam from the phased plasma rifle travels at 0.5c (from Billy’s point of view), Bob will not see the beam of the phased plasma rifle moving away from him at 0.8 (Billy’s speed away from Bob) + 0.5 (velocity of the beam) = 1.3 c. Nope, no way, no how.
Velocity doesn’t add up like you’d think based on your much-slower-than-light experience.
Here’s the formula, on the left is the speed that you see as you watch someone, who is moving, fire his phased plasma rifle.
Vtotal = (vbolt + vperson) / ( 1 + vperson vbolt / c2 ).
Vtotal is the total velocity you see. vperson is the velocity the person firing the rifle is moving. vbolt is the muzzle velocity of the rifle. In other words vbolt and vperson are the two velocities you are trying to add, the velocity of the person in your reference frame, and the velocity of the rifle plasma bolt, in his reference frame.
So in our example, the top is 1.3c, and the denominator is ( 1 + .4c2/c2 ) so the total velocity is 1.3/1.4 times c. Which works out to .928c. That’s how fast you’ll see the phased plasma rifle’s bolt move from your reference frame.
The formula works in such a way that any two speeds slower than light will add to another speed slower than light.
If you are dealing with situations much, much lower than the speed of light, the bottom of the formula becomes 1 and you can just add velocities like you’re used to doing, a 60 mph pitch straight ahead on a train moving at 50 mph will look like 110 mph to someone watching the train go by. It will be very very very (immeasurably) slower than 110 mph in fact.
Momentum
But if you cannot add velocities, then you also cannot simply add momenta (momentums) because momentum is simply the mass of the object times its velocity. Indeed momentum itself doesn’t seem to be conserved in collisions!
However, there’s such a thing as relativistic momentum, which is conserved. It’s essentially our old friend γ times the classical momentum. Which means, of course, that at very low speeds, it looks just like the momentum we are used to and that momentum therefore looks to be conserved.
Even F = ma gets called out. Doing a unit analysis, force is mass times distance over time squared. But mass times distance over time (without squaring it) is momentum, so force can be thought of as momentum over time. We already have a relativistic momentum, so now just by dividing by time we have a relativistic force.
Force, of course, allowed to operate over a distance without being balanced out, is work. You can, through some rather messy algebra (which my college physics text…you guessed it…left as an exercise for the student), get from there to a formula for relativistic kinetic energy.
This is:
Ek = γmc2 – mc2
If an object is not moving, γ is one, and the kinetic energy is zero. We can sanity check this for very low speeds by using an approximation for γ which is that γ is approximately 1 + 1/2 v2/c2…. with the further terms all vanishingly small.
Plug that value of gamma into the equation above and you get:
Ek = (1 + 1/2 v2/c2) mc2 – mc2
Multiply out the first term:
Ek = mc2 + (1/2 v2/c2) mc2 – mc2
The first mc2 and the last one cancel each other out. The middle term’s two c2s cancel each other out as well which leaves you with the familiar:
Ek = 1/2 mv2
So again we see a case where a familiar classical formula is equal (to within an immeasurably small amount) to the relativistic formula for the same thing, at very low speed.
Our situation as people who move very slowly compared to light is just a special case, and classical mechanics only holds true in that special case. It’s close enough, in fact, that for daily life you can just ignore the special relativity aspect of things. Which Galileo, Newton, et. al. did do, out of not knowing it was there.
Returning to our formula for relativistic kinetic energy:
Ek = γmc2 – mc2
The first term has γ, which in turn has a dependency on velocity. The second term does not depend on velocity; it’s a sort of energy that just depends on the mass of the object.
In fact mc2 is now called the “rest energy”.
If you add the rest energy of some particle to the kinetic energy of that particle, the mc2s cancel out and the total energy is simply
E = Ek + Erest = γmc2
But of course, unless you just arrived here from the nineteenth century (or earlier) by time machine, you recognized the rest energy formula right off the bat:
Erest = mc2
Interestingly that famous formula is only half of the real formula for total energy.
But it does imply that even a totally stationary mass has energy locked up in it.
HOw much energy? 1kg times the speed of light, times the speed of light. Which is 1kg x 299,792,458m/s x 299,792,458m/s = 89,875,517,873,681,764 joules, 89 quadrillion joules, still slightly more than our national debt. Ten million 100 watt light bulbs could be run for 89,875,517 seconds with this energy; that’s almost exactly 2.85 years! A billion watts for three years!! Out of one lousy kilogram of mass.
Of course, we don’t know how to convert all of any mass into energy.
But pretty much any time we release chemical or nuclear energy, we convert some of that mass into energy. Chemical reactions release so little energy per kilogram (compared to this ridiculously huge number) that we can’t actually measure the mass change. But nuclear reactions do have a measurable effect on mass, as we shall begin to see when my narrative returns to the atom next time.
As I pointed out previously, this throws the conservation of mass into the toilet. Since mass can turn into energy (and vice versa), we now have a conservation of mass-energy. To be honest though, many physicists simply think of matter as just another form of energy, and talk about the conservation of energy without qualification since energy is seen to include matter now.
Now I’ll be honest with you that derivation seems to me like a lot of hand waving. At the end you just added the thing you were subtracting out back in and called it “rest mass.” But there is no real doubt any more, “rest energy” is real. We see it turn up every time we look inside the atom.
Revisiting Electromagnetism
Recall that Einstein’s original paper on relativity was titled “On the Electrodynamics of Moving Bodies.” We haven’t even mentioned electricity and how it behaves at these velocities, though.
Einstein got all of this stuff by looking at electromagnetism, not from trying to figure out why Michelson-Morley got a null result.
Remember from last time: “If you move a coil of wire through a stationary magnetic field, a current is induced in the wire. The problem is, if you looked at it from the point of view of the wire, the effect is due to an electrical force. But from the point of view of the magnet, the effect is due to a magnetic force.”
So, two different reference frames, each getting a difference in the mechanism for getting the current flowing in the wire. This looks like a contradiction, and it worried a lot of people at the end of the nineteenth century. But it turns out that if you bring relativity into play, it gets resolved.
The too long, didn’t read is that magnetism will turn out to be electricity–with relativistic effects.
I’ll illustrate that with an example; this is going to force you to remember a lot of electromagnetism.
Imagine a long, straight wire carrying a current. You’re sighting down that wire, looking in the same direction as the current. The wire is running past your eye and diving into your computer screen, so the current flows into the screen.
(And remember that current is treated as if it were a positive charge moving, not an negative charge, so in reality the electrons are coming toward you. [And gee, it’s nice not to have to talk about “electrical fluid” any more.])
Even though current is flowing, there is as much negative charge in any part of the wire as positive charge. There’s no net electric charge, and therefore there is no electric field.
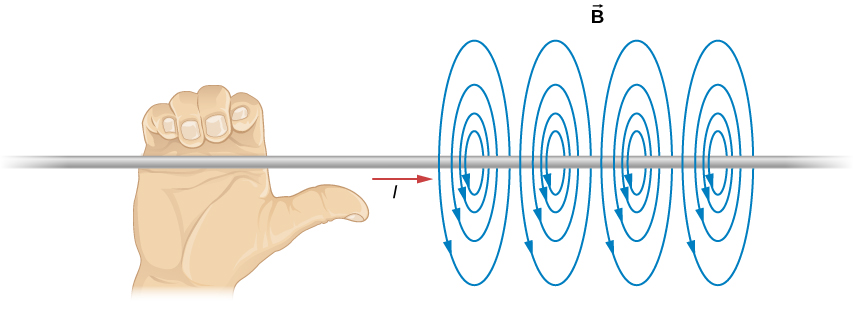
However, every current creates a magnetic field, In this case, it runs in rings around the wire according to the right hand rule. Orient your right thumb in the direction of the current (which, remember, is defined based on notional positive charges moving, so it’s in the opposite direction of the motion of the electrons). The fingers of your right hand point in the direction of the magnetic field. Thus from your vantage point the magnetic field lines run in clockwise circles around the wire. To the left of the wire they run upwards, to the right, downwards.
OK, so imagine a positive test charge sitting near the wire, to the right of it as seen by you. If it’s stationary with respect to the wire, it just sits there. There’s no electric field, so it’s not being pulled towards (or pushed away from) the wire. And it’s not moving through the magnetic field so no F = qv x B because v is zero.
OK, now imagine that test charge moving, away from you, into the computer screen. Now we have a velocity, and qv is a vector pointing into the screen. But, where the test charge is, the magnetic field points straight down. Use the right hand rule, and the test charge feels a force towards the wire thanks to its interaction with the magnetic field, created by the flowing current.
So: send the positive test charge alongside the current, it gets drawn toward the wire by the magnetic field induced by the current. Still with me?
OK, let’s back up. Let’s run this scenario again, but momentarily forget the magnetic field.
The notional particles carrying the current are positively charged, and they must have a certain spacing as they move along, if they are further apart than that spacing, then the wire would have more negative charge in a certain length than positive and the wire would have a net charge and there’d be an electric field.
Now let’s ride along with our test charge outside the wire. It is now moving closer to the speed of the current than it was when stationary. And it is now moving with respect to the negative stationary charges in the wire.
So it sees the negative charges get closer together, because of relativistic length contraction.
And it sees the positive charges get further apart, because it’s moving closer to their velocity so the length contraction that was always there, is now lessened. In fact, if the charged particle is moving at the same speed as the current, the partices making up the current are as far apart as they can be because they’re at rest in the postive charge’s frame of reference.
If you crowd the negative charges in the wire closer together and space the positive charges further apart, which is what our moving test charge sees, now the wire does have an electrical field, one due to a net negative charge in the wire. The positive test charge is now attracted to the negative wire by an electric field.
How much is it attracted to the wire? Exactly as much as the magnetic field did when we looked at our test charge as if it were moving through a magnetic field.
They are, in fact, the same effect! A magnetic field is just what someone sees due to relativity acting on distributions of electrical particles.
Note, we got this by applying length contraction to the charges in the wire, not through the laws describing the interplay between electricity and magnetism. Length contraction, etc., must be implicit in Maxwell’s equations, but Maxwell certainly never noticed!
And thus, another thing gets explained by relativity. In fact it was the first thing to be explained–this is what Einstein was trying to solve after all, but the point is all the pieces fit together, quite nicely.
And 116 years later they still do.
Conclusion
The most important of these pieces is that Joe Biden didn’t win.
Obligatory PSAs and Reminders
China is Lower than Whale Shit
Remember Hong Kong!!!
中国是个混蛋 !!!
Zhōngguò shì gè hùndàn !!!
China is asshoe !!!
China is in the White House
Since Wednesday, January 20 at Noon EST, the bought-and-paid for His Fraudulency Joseph Biden has been in the White House. It’s as good as having China in the Oval Office.
Joe Biden is Asshoe
China is in the White House, because Joe Biden is in the White House, and Joe Biden is identically equal to China. China is Asshoe. Therefore, Joe Biden is Asshoe.
But of course the much more important thing to realize:
Joe Biden Didn’t Win
乔*拜登没赢 !!!
Qiáo Bài dēng méi yíng !!!
Joe Biden didn’t win !!!