The above blurred image of persons attending a conference is courtesy of Google Images and Vecteezy.
Today’s Health Friday offering is one a of a series devoted to Big Pharma, vaccines, general health, and associated topics. The discussion is not limited to what is presented in today’s post: it is an Open Thread.
There are Important Wolf Moon Notifications; the Rules of our late, good Wheatie; and other items that readers should be familiar with. They are all linked here.
This post will be diving into some “interesting” rabbit-holes. Stay with Yours Truly — I believe they are all interconnected. There is a General Summary at the end of the post.
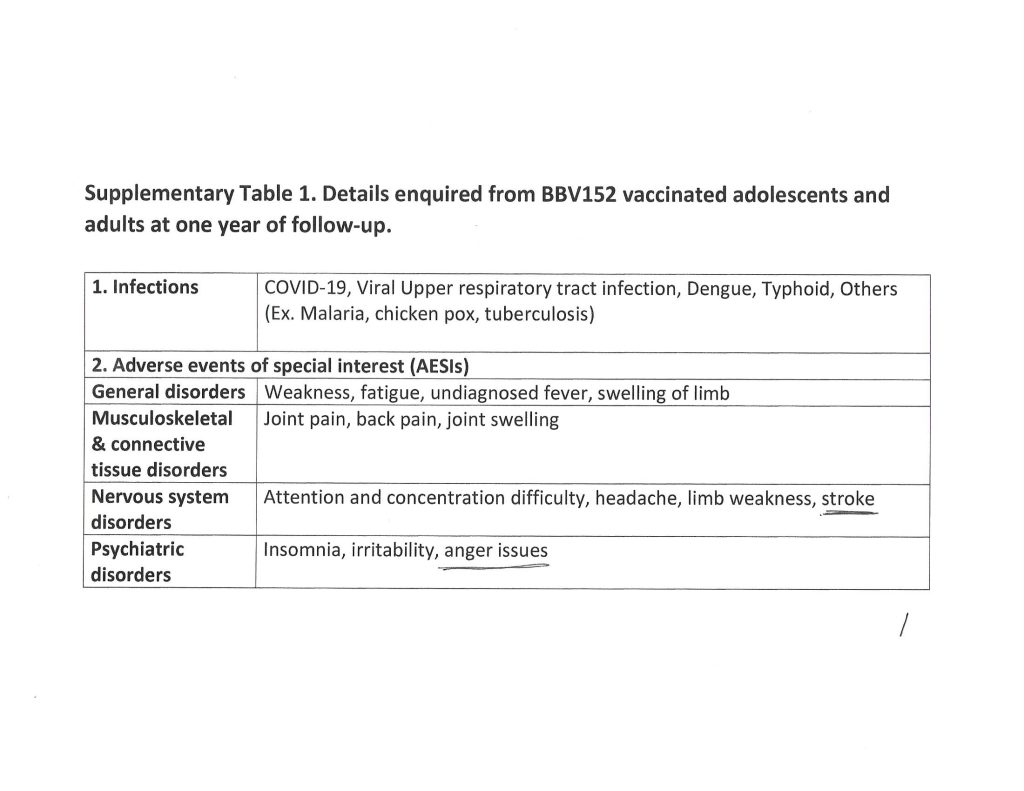
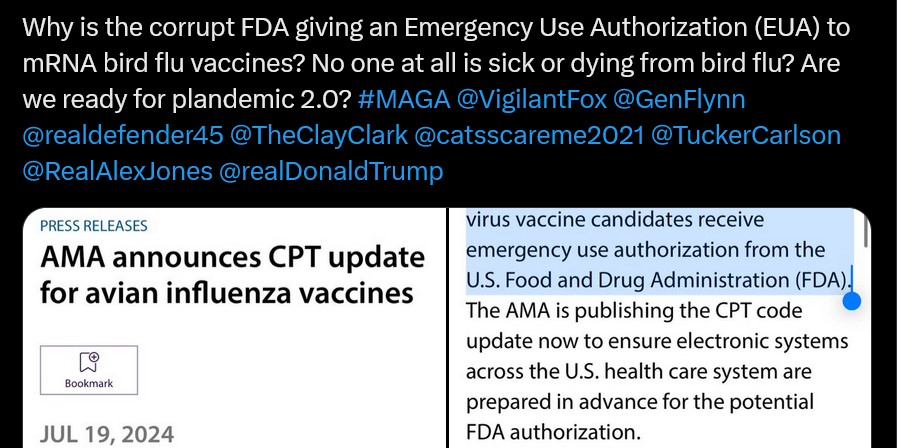
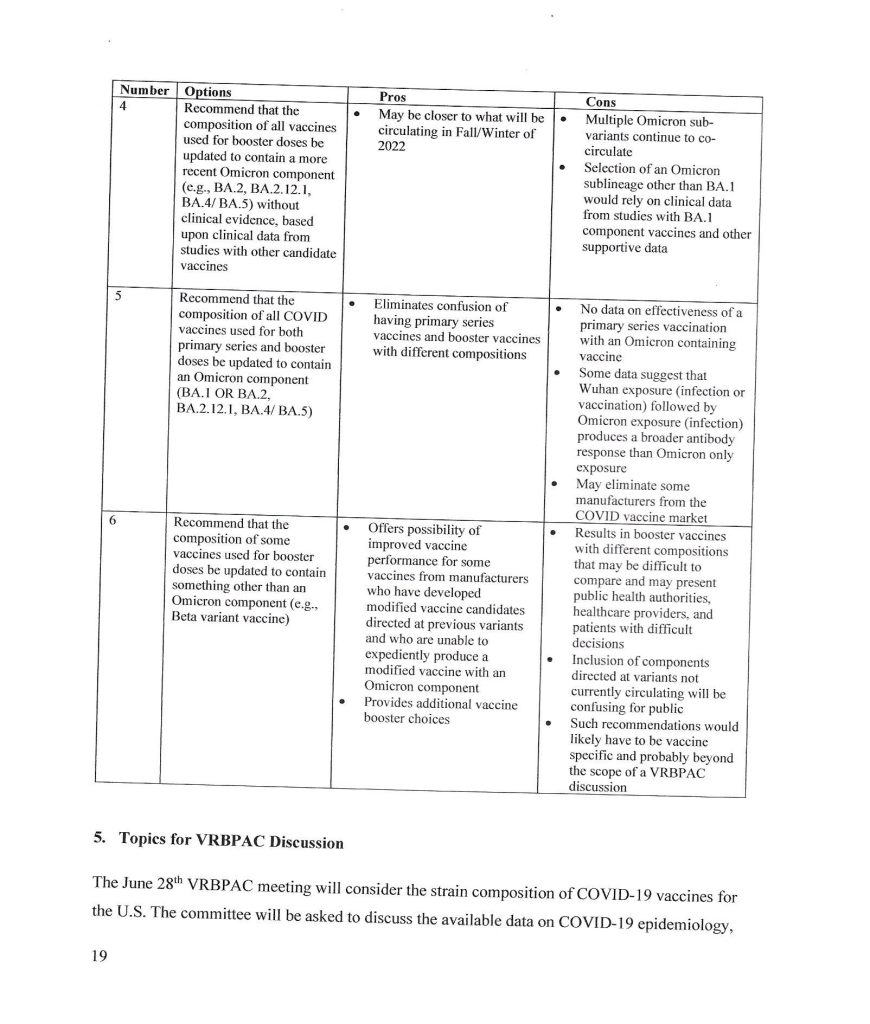
The trail, for today’s purposes, begins with the following links: www.theburningplatform.com/2024/10/04/exclusive-new-york-city-health-officials-to-hold-bird-flu-tabletop-exercise/, 4 October 2024, by Michael Nevradakis, Ph.D.; https://childrenshealthdefense.org/defender/nyc-health-officials-bird-flu-summit-tabletop-simulated-plan/, 3 October 2024, by the same author (goes into more detail); and, www.theqtree.com/2024/08/02/the-hhs-gave-the-go-ahead-to-use-an-h5n1vaccine-but-the-ama-just-issued-new-cpt-codes-for-an-h5n8-vaccine/, 8 August 2024, by Yours Truly.
Note: the scientific paper referred to by Dr. Nevradakis is here: www.preprints.org/manuscript/202406.0060/v1, “Proximal Origin of Epidemic Highly Pathogenic Avian Influenza H5N1 Clade 2.3.4.4b and Spread by Migratory Waterfowl”, Nicolas Hulscher, John Leake, Peter McCullough, 3 June 2024; click on “Download PDF” to read or save the paper. The authors suspect Gain-of-Function to be in play. Below is the Abstract of the paper:

Since this paper is a preprint; and, that it may be a target for Retraction; Withdrawal; or, Reissue with “new conclusions” that would reflect pressure on the paper’s authors and/or the publisher, Yours Truly suggests that interested readers download or make a hard copy of the preprint version.
The International Bird Flu Summit was held in Fairfax, Virginia, on 2 – 4 October 2024. This is the website for the “summit”: https://birdflusummit.com/. Here is the Brochure for the “summit”: https://birdflusummit.com/Bird Flu Summit Brochure.pdf. Reading through these “summit” websites, one gets the distinct impression of a general tone that “a Bird Flu pandemic” is about to occur; and, that various “interventions”, “command and control” methods, and “monitoring and dismantling” of “misinformation” detected on social media platforms, etc., will be “necessary” as part of a “coordinated response.”
Taking a look at the Brochure for the “Bird Flu Summit”, here is a portion of one of the “overview” pages:

Seems innocuous enough. However, moving on in the Brochure, one finds this:

And, further along in the Brochure, there is this:

Military involvement? “Inter-agency Communication and Resource Allocation”? What is “Seamless Response”?
John Leake, who works with Dr. Peter McCullough, attended this “summit.” Here is his take: https://petermcculloughmd.substack.com/p/at-the-bird-flu-summit, 2 October 2024. Yours Truly agrees with his impression that the speakers at the “summit” were “middle-of-the-road, rather than barn-burning experts” (my phrase.) One also agrees with the impression that the “summit” was all about “a Bird Flu pandemic being just around the corner.” However, this is NOT to minimize the potential for something like a “bird flu epidemic / pandemic” occurring — either from a “natural cause”: or, by another “lab leak.” Here is another view on the situation: www.theburningplatform.com/2024/09/22/could-bird-flu-be-the-october-surprise/, by Clayton J. Baker, MD.
FLASH! — The McCullough Foundation just got a demand from one of the speakers at the “Bird Flu Summit”, Syra Madad, PhD, to delete a video that John Leake and Nicolas Hulscher (another McCullough Foundation member) made of a Question-and-Answer session between Mr. Leake and herself at the “summit.” Dr. Madad is the coordinator of the upcoming New York City “Bird Flu Response Conference.” Please see here: https://petermcculloughmd.substack.com/p/mccullough-foundation-receives-bird, “McCullough Foundation Receives Bird Flu Summit Censorship Demand”, 9 October 2024. Below is a portion of the article:

The transcript of the video follows in the article (the video was deleted.) Below is a portion of the transcript, followed by an assessment by Mr. Leake:

Turning to the entity that “presented” the event (Ginkgo Biosecurity), and the entity that “produced” the event (SyllabusX): this is where the “rabbit holes” emerge; and, something that Yours Truly will call the Big Pharma-Government-Medical Complex (BPGMCTM) comes into the situation. Yours Truly defines BPGMC as an “interwoven cooperation” among Big Pharma, Federal government agencies regarding development / funding / support (including the Department of Defense), institutional investors, private investors, and “Establishment Medicine” (CDC, FDA, AMA, etc.)
First, the “producer” of the “Bird Flu Summit”, SyllabusX. The company’s address and phone number: 1900 Campus Commons Dr., Suite 100, Reston, VA 20191; phone number: 703.466.0011. The company was founded in 2017. SyllabusX has its “fingers in many pies” regarding producing conferences and other events on student safety and security, on technology for educators, on health and biosecurity, and more (https://syllabusx.com/home-2; and, https://syllabusx.com/about-us.) (Note: it is not easy to find information online about this company, especially as regards financials. A couple of sources that Yours Truly found: https://pitchbook.com/profiles/company/520513-39#overview; and, www.zoominfo.com/c/syllabusx-inc/480230653.) It appears that SyllabusX is a “privately-held company.”
The Chief Research Officer of SyllabusX is Ahmed Al Faraj (www.linkedin.com/in/ahmed-al-faraj; Note: this link may not work, unless the reader first joins LinkedIn.) And here’s where the issue gets, IMO, interesting. It appears that Mr. Faraj graduated with a B.S. in Economics from the University of Kansas School of Business in 2006. His languages are listed as, “Native or bilingual proficiency” in Arabic.
Before starting SyllabusX, Mr. Faraj previously worked for a company called New-Fields Exhibitions, headquartered in Dubai (https://inf.ae/.) While at New-Fields Exhibitions, Mr. Faraj held positions as Program Manager, Education & Public Health Group; and. as Program Manager, Defense Business Group. Below are two screenshots from the type of work he performed while at New-Fields Exhibitions:

Mr. Faraj also previously worked for a company called AdvanFort. He was the Chief Operating Officer of this company. AdvanFort is owned by a person called Samir Farajallah. His son, the then-Chief Operating Officer, was called Ahmed Farahjallah. AdvanFort is “a US private maritime security company”, per Wikipedia. The company was founded in 2007, and its headquarters office is in Herndon, VA (www.advanfort.com/.) A vessel owned by AdvanFort was involved in an international maritime incident in 2014. One of the stories that covers this incident is here: www,bbc.com/news/uk-england-34095843, 7 September 2015, “AdvanFort accused of abandoning British men facing India trial.” As a result of this incident, AdvanFort withdrew from membership in SAMI (the Security Association for the Maritime Industry): https://gcaptain.com/sami-responds-accusations-from-advanfort/, 2 April 2014. (Interested readers may want to compare the photo of Ahmed Faraj on the SyllabusX LinkedIn site, to the photo of Samir Farajallah and Ahmed Farajallah that is in the BBC article linked above. In one’s opinion, the “Ahmed” in these photos is the same person, with a perhaps 10-year gap between the photos. One has no objection to people wishing to change their names — this is simply an interesting item.)
Second, the “presenter” of the “Bird Flu Summit” event, Ginkgo Biosecurity (Ginkgo Bioworks www.ginkgobioworks.com/.) The company’s address and phone number: 27 Drydock Ave., 8th Floor, Boston MA 02210; phone number: 877-HACK-DNA. The company works in “cell programming and biosecurity.” Ginkgo Biosecurity was founded in 2008 by students and scientists at MIT. Two of the founders are Tom Knight (known as the “godfather” of “DNA hacking” and of “synthetic biology” (www.prnewswire.com/news-releases/ginkgo-bioworks-to-become-a-public-company-and-expand-its-leading-platform-for-cell-programming-301288325.html, 11 May 2021; and, Austin Che https://startupexchange.mit.edu/watch/dinkgo-bioworks, 17 July 2017. Mr. Che is also a mentor for Creative Destruction Lab in Toronto, Canada (https://creativedestructionlab.com/mentors/austin-che/.) Below is a screenshot from the Ginkgo BioWorks website:

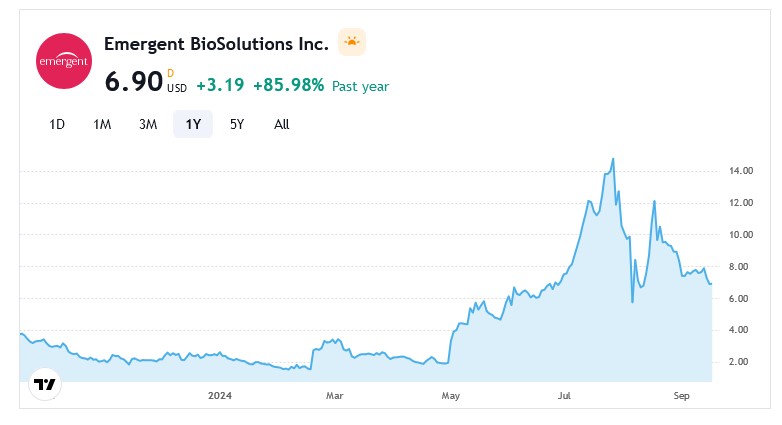
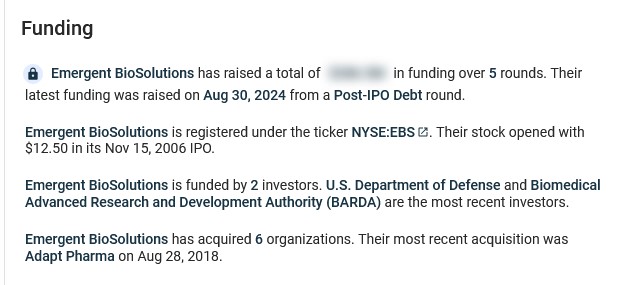
It appears that Ginkgo BioWorks has quite a bit of funding. Below are screenshots from https://tracxn.com/:
And, some institutional funders:

The United States government is also funding the biosecurity arm of Ginkgo BioWorks: www.dfc.gov/media/press-releases/dfc-approves-11-billion-loan-ginkgo-bioworks-expand-its-commercial-biosecurity, “DFC Approves $1.1 Billion Loan to Ginkgo BioWorks to Expand its Commercial Biosecurity Business to Combat COVID-19 on a Global Scale”, 25 November 2020. DFC is the U.S. International Finance Development Corporation. Below is a screenshot from the article:

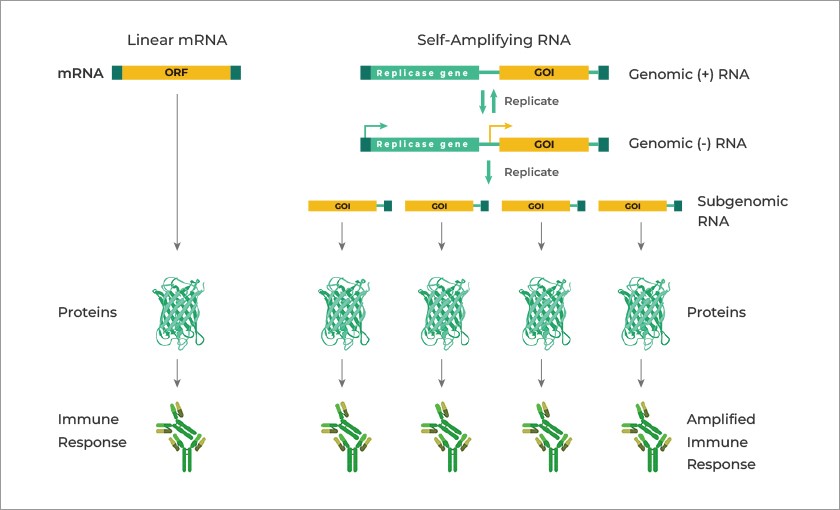
For comparison, here is a list of some raw materials used in nucleic acid-based vaccines (mRNA-based vaccines), from www.promega.com/custom-solutions/custom-manufacturing/mrna-manufacturing/:
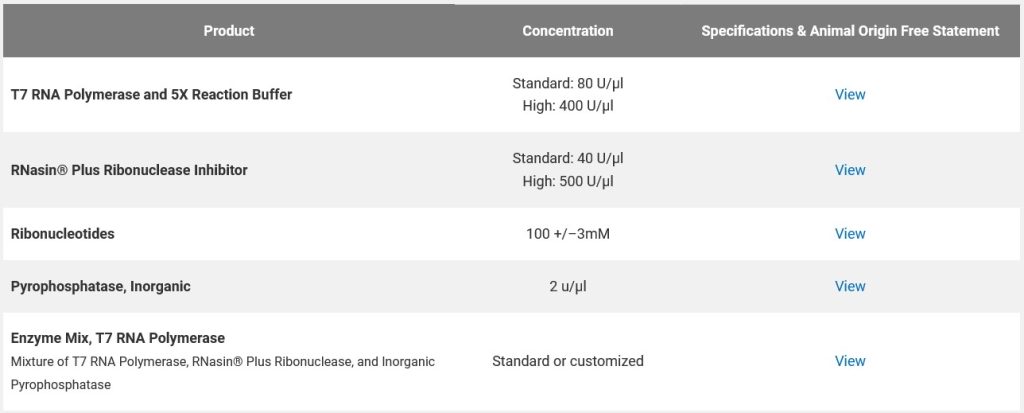
It would appear that Ginkgo BioWorks was involved in the “rapid deployment” of those PCR tests and antigen tests for COVID-19 at the start of the pandemic; and, also in the production of “two key raw materials for nucleic acid-based vaccines” (the modRNA COVID-19 “vaccines”.) Below is a blog post from April 2020 by Jason Kelly, another Ginkgo BioWorks co-founder, regarding the company’s partnership with Moderna in the production of the modRNA COVID-19 “vaccine”, mRNA-1273, along with other of that company’s mRNA “vaccines”:

Yours Truly has not, to date, found information as to the exact “raw materials” that Mr. Kelly is referring to.
Recall that the NIH/NIAID and Moderna co-developed mRNA-1273, co-own the patents of this modRNA COVID-19 “vaccine”, and share in the “royalty payments” for the sales and administration of this “vaccine” (and its “descendant clone” modRNA COVID-19 “vaccines.”)
Ginkgo BioWorks also “has its fingers in many pies”, including the bio-engineering of elements for gene therapy: www.ginkgobioworks.com/offerings/gene-therapy-services/. One such “service” is the bio-engineering of bacteria for treatment of diseases, such as cancer: www.ginkgobioworks.com/2023/01/09/engineering-bacteria-for-cancer-patient-treatment-with-prokarium/ (Prokarium is another “partner” with Ginkgo BioWorks.) The bacteria to be bio-engineered is a type of Salmonella bacteria, which will be used for “delivery of RNA payloads to treat cancer patients.” (Salmonella is “the most common form of food poisoning the in the U.S.”, according to the Cleveland Clinic. This brings up two questions: One, “Why is a bacteria related to food poisoning going to be used to treat cancer patients?”; and, Two, “How is this Salmonella bacteria going to be bio-engineered — is it going to be “sanitized” to be a beneficial element?”)
In Yours Truly’s opinion, Ginkgo BioWorks is a company to keep “on the radar.” It is acquiring other “partner companies” at a rapid rate. It is expanding its “services” into the gene-therapy sector. It is a “partner” with Moderna (and, therefore, by extension the NIH / NIAID.) In general, one has no objection to a company wishing to increase its profit margin and/or the number of its partner companies; at issue here are the following factors: the bio-engineering of DNA; the use of “automated tools” for engineering / manufacturing “raw materials” for gene-therapy treatments and/or “vaccines”; and the tie-in with the federal government.
General Summary: One: The International Bird Flu Summit was held in Fairfax, Virginia, earlier this month. The general tone of the “summit” was that a “bird flu epidemic / pandemic outbreak” is likely to occur, and soon. Two: The “summit” covered many topics related to epidemic / pandemic response, “command and control” methods, and how “seamlessly” federal, state and local governments and the military can “work together” in the response. Three: the “producer” of the “summit” was SyllabusX, a company that manages events ranging from student security to biosecurity to health education. SyllabusX is based in Virginia. Its Chief Research Officer is Ahmed Al-Faraj, who had previously (apparently) worked for his father’s company, AdvanFort, and (actually) for several other companies. Four: The “presenter” of the “summit” was Ginkgo Biosecurity (Ginkgo BioWorks), a company that offers “services” ranging from commercial biosecurity to bio-engineering to gene therapy research. This company, based in Massachusetts, has “deep funding” from many sources, including from the U.S. government. It has multiple “partners” in R&D, including Moderna. Ginko BioWorks was founded by students and scientists from MIT, including Tom Knight, the “godfather” of “DNA hacking” and of “synthetic biology.” Five: It appears that Ginkgo BioWorks, and, possibly, SyllabusX, are part of what may be called the Big Pharma-Government-Medical Complex (BPGMCTM), the “reach” of which runs far and wide.
Yours Truly will again emphasize the importance of having, and maintaining, the highest degree of health possible of the body’s natural immune system.
Peace, Good Energy, Respect: PAVACA