SPECIAL SECTION: Message For Our “Friends” In The Middle Kingdom
I normally save this for near the end, but…basically…up your shit-kicking barbarian asses. Yes, barbarian! It took a bunch of sailors in Western Asia to invent a real alphabet instead of badly drawn cartoons to write with. So much for your “civilization.”
Yeah, the WORLD noticed you had to borrow the Latin alphabet to make Pinyin. Like with every other idea you had to steal from us “Foreign Devils” since you rammed your heads up your asses five centuries ago, you sure managed to bastardize it badly in the process.
Have you stopped eating bats yet? Are you shit-kickers still sleeping with farm animals?
Or maybe even just had the slightest inkling of treating lives as something you don’t just casually dispose of?
中国是个混蛋 !!!
Zhōngguò shì gè hùndàn !!!
China is asshoe !!!
And here’s my response to barbarian “asshoes” like you:
OK, with that rant out of my system…
Justice Must Be Done.
The prior election must be acknowledged as fraudulent, and steps must be taken to prosecute the fraudsters and restore integrity to the system.
Lawyer Appeasement Section
OK now for the fine print.
This is the WQTH Daily Thread. You know the drill. There’s no Poltical correctness, but civility is a requirement. There are Important Guidelines, here, with an addendum on 20191110.
We have a new board – called The U Tree – where people can take each other to the woodshed without fear of censorship or moderation.
And remember Wheatie’s Rules:
1. No food fights
2. No running with scissors.
3. If you bring snacks, bring enough for everyone.
4. Zeroth rule of gun safety: Don’t let the government get your guns.
5. Rule one of gun safety: The gun is always loaded.
5a. If you actually want the gun to be loaded, like because you’re checking out a bump in the night, then it’s empty.
6. Rule two of gun safety: Never point the gun at anything you’re not willing to destroy.
7. Rule three: Keep your finger off the trigger until ready to fire.
8. Rule the fourth: Be sure of your target and what is behind it.
(Hmm a few extras seem to have crept in.)
Spot Prices
All prices are Kitco Ask, 3PM MT Friday (at that time the markets close for the weekend).
Last week:
Gold $1762.00
Silver $22.65
Platinum $981.00
Palladium $2000.00
Rhodium $14,050.00
This week, markets closed for the weekend at 3:00 PM Mountain Time
Gold $1758.00
Silver $22.75
Platinum $1031.00
Palladium $2167.00
Rhodium $14,850.00
Gold and Silver are holding steady…ridiculously so in fact. I read speculation that they’re going to bust out and surge. Why shouldn’t they? Inflation is galloping, the economy is headed for trouble once (some of) the companies out there actually stick to the jab mandate.
Platinum and palladium have taken decent jumps. Rhodium is up $800. That’s not too shabby either.
Personally? I’m liable to end up unemployed. I should buy a “I can’t afford to fix or replace this because of J** B*d*n” bumper sticker for my rear-ended car.
Part XXI: Nuclear Physics Uses The Hammer
Introduction
Last time I said that this time I’d take up stars. But I did some preliminary research on the history, and realized that we’re not quite there from a historical standpoint as our narrative is basically in the 1930s (except when I run ahead to finish something that started in the 1930s, like I did with neutrinos).
So I’m going to pick up the story of neutrons. Discovered in 1932 by James Chadwick, they turn out to be the “other” nucleon in the nucleus, supplementing protons. Similar in mass but with no electric charge, they were the actual occupants of the place in the nucleus that we had imagined held proton-electron pairs.
Because a neutron bears no electric charge, it has no trouble getting close to a nucleus and sticking to it, whereas a proton is repelled by any nucleus it approaches. If it can get close enough it will stick…but first it has to get close enough, and that’s a challenge. The same is true of alpha particles (which are bundles of two protons and two neutrons).
The “sticking” is provided by the strong nuclear force.
It’s as if you had two magnets, and were trying to bring the north poles close together. They push each other apart pretty hard, but if the magnets were covered with velcro, they’d stick together…once you overcame that repulsion.
Free neutrons are basically a new form of radiation, by the way; we have alpha and beta radiation (that bundle of four nucleons, and an electron, respectively), gamma radiation (a very high energy photon, X-rays on steroids), and now, we have free neutrons.
Free neutrons are scary. They’ll simply wander around until they find a nucleus to stick to…and they will more than likely make that nucleus radioactive. I don’t mind being around alpha and beta sources (so long as they’re not inside of me); they’re trivial to shield against. Gamma rays are intimidating because they penetrate very thick shielding. All three of these, if they get to you, will blast some chemical bond to smithereens which can either mean nothing or cause big problems, depending on what it was they hit. They won’t make you radioactive. But the neutrons just sort of wander aimlessly through matter, unaffected by very much until they find a nucleus–and nuclei don’t take up much space, in fact they take up virtually none of it. Whatever nucleus they hit becomes a new (and likely radioactive) isotope.
That nucleus, with the extra neutron, may find itself with “too many” neutrons, and one of the neutrons will then change into a proton, via the weak force. This has the effect of making that atom a different element, the one next over to the right on your handy-dandy periodic table. That increases the atomic number, Z, by 1, while leaving the mass number (the total number of protons and neutrons) the same.
OK, that’s the end of the review. Now on with the story, which is complicated. I apologize in advance if this is completely un-followable. And if I somehow managed to garble it in trying to simplify it, I apologize for that as well. [Most of this is from the Wikipedia article on Lise Meitner, and the article on the discovery of fission.]
Transmutation
Nuclear physicists had all kinds of fun playing with neutrons through the 1930s (and beyond). Enrico Fermi, in Rome, made a hobby of bombarding different elements with neutrons to see what would happen; first creating a more neutron rich isotope of the starting element, then monitoring the beta decay, determining half lives and energies, which are different for each isotope. Sometimes there’d be multiple decays, because one wasn’t enough to get to a stable isotope.
Remember, each such beta decay moves you one to the right, to the next higher atomic number. This led to an irresistibly tantalizing question.
What happens if you pick the element with the highest atomic number, uranium with Z=92, and bombard it with neutrons?
Shouldn’t you get element 93, previously utterly unknown, in fact, previously nonexistent?
Fermi tried it. And he got a whole bunch of different kinds of beta radiation out of it. He concluded that he had created a “transuranic” element. Not so fast though. Aristid von Grosse suggested that what Fermi had found was a new isotope of protactinium (element 91, not 93). This “wait a minute” wasn’t enough to prevent Fermi’s winning the 1938 Nobel Prize for Physics for this work, not just with uranium but the other elements as well.
But there was enough controversy that someone needed to dig in and figure out if we were looking at element 93 or protactinium.
And who better to do that than Lise Meitner and Otto Hahn, the discoverers of protactinium? Their collaboration at Kaiser Wilhelm institute in Berlin had lapsed, but this question got the two of them back together. From 1934-1938 the two of them, along with Otto Frisch, dug into the matter.
Initially, Meitner and Hahn believed they had created elements 93, 94, 95 and even 96. But as time went on Meitner became less certain.
Part of the muddle came from the fact that it was wrongly believed that only the lanthanide elements had that special row at the bottom of the table, pulled out from the main body so it would fit nicely on a landscape piece of paper. Actinium was placed two spots below yttrium, thorium below hafnium, protactinium below tantalum and uranium below tungsten (or as the Germans called it, “wolfram”). Indeed the chemical behavior of these elements could be a bit confusing, but it would eventually turn out that that stopped with element 93, which behaved more like a lanthanide. That whole sequence of elements in fact belonged in a second footnote row below the lanthanides.
For example, Fermi had found a rhenium-like element in his experiments and, in the belief that element 93 was directly below rhenium in the periodic table, concluded that that is what he had found. (In fact, he had found technetium, the then-undiscovered element above rhenium in the table, and didn’t realize it–but I’m getting ahead of myself here.)
This mistaken belief, at the time, bunged up any attempt to chemically analyze the products of the neutron bombardment. When element 93 is expected to behave like rhenium, for instance, rather than like a rare earth, it’s kind of difficult to figure out what’s going on.
One thing Meitner and Hahn wer fairly confident of: when they bombarded uranium, which was mostly uranium-238 (92 protons and 146 neutrons), they were indeed getting, as step one, uranium-239, with a 23 minute half life. They were able to do chemistry on it and prove that it was, indeed uranium.
After that it was a muddle. There seemed to be three different reactions, all from uranium-239, one with a ten second half life, one with a twenty second half life, and one with a 23 minute half life.
In 1937 Meitner and Hahn each published a report. Hahn was emphatic that they had found transuranic elements (“Above all, their chemical distinction from all previously known elements needs no further discussion”); Meitner was pretty certain almost everything was a product of uranium-238, somehow, but figured the three most prominent products were isomers.
Er, what’s an isomer?
As if it isn’t difficult enough to recall that elements come in isotopes, with the same number of protons but different number of neutrons, it turns out that some of the isotopes themselves come in different forms, some more energetic than others, and that the more energetic form eventually just blasts out pure energy (a gamma ray photon) and settles down to become the less energetic, and (usually) more stable form, having kept all of its protons and neutrons intact (but, likely, having dropped mass a bit). An isotope like this gets an “m” after the number.
For example, consider protactinium-234m, which has a 1.17 minute half life, and ejects a photon as it settles down to become protactinium-234, with a half life of 6.70 hours. When Pa-234m was discovered in 1913, we weren’t clear on the concept of isotopes, so it was considered a new element and named brevium for its brief half life.
When “regular” Pa-234 was discovered in 1921, that marked the discovery of nuclear isomers; it was the first such distinction between an “m” isotope and a “regular” isotope. And, interestingly, the discoverer was Otto Hahn, who later on in 1937 found his colleague using the concept to argue against his interpretation of the U-239 decay products!
[Side note: Probably the most useful isomer today is technetium-99m. It’s a decay product of molybdenum-99, which has about a 30 hour half life. Mo-99 is sent to hospitals, which extract the Tc-99m chemically, embed it in larger molecules, perhaps favored by muscles, then inject that into patients and watch where the gamma rays come from. This can be used to diagnose heart problems, though it does mean the patient is a source of gamma rays for a while. Tc-99m has a six hour half life, after which it blasts out a fairly weak gamma ray and settles down to Tc-99, which has a much longer half life (hundreds of thousands of years) and will ultimately beta decay and become ruthenium-99. The patient generally gets rid of the technetium-99 within days, so no digging up bodies to try to get the ruthenium, please.]
Meitner concluded her report with the following: “The process must be neutron capture by uranium-238, which leads to three isomeric nuclei of uranium-239. This result is very difficult to reconcile with current concepts of the nucleus.”
Another group in Paris decided to investigate as well. They ultimately found a product that was chemically very similar to lanthanum (element 57). (It turned out it couldn’t be more similar, as it was lanthanum, but I get ahead of myself again.)
Did I just almost forget to mention Meitner was Jewish?
What does that matter? Normally it wouldn’t matter in the slightest, but in mid 1930s Berlin, it mattered a great deal. And it was mattering more and more as time passed.
Meitner Has To Flee
Meitner had been kept safe, somewhat, by the fact that she was an Austrian, but on March 12, 1938, Austria was annexed by Germany. Her Austrian citizenship was moot as there was no Austria to be a citizen of. Niels Bohr and Paul Scherrer invited her to take positions in Denmark and Switzerland, respectively, but Carl Bosch at KWI said she could remain. By May, though, Meitner learned that her situation was being looked at by the no-doubt misnamed Reich Ministry of Science, Education and Culture.
Although many people outside of Germany wanted to give her refuge, there were all sorts of bureaucratic snafus. For instance, she couldn’t go to Denmark no matter how much Niels Bohr wanted her there, because Denmark considered Austrian passports to be invalid. Germany also forbade academics to leave the country.
By July the situation was critical. Dirk Coster, a Dutch scientist, convinced the Netherlands to accept Meitner, and on July 12, she showed up for work at KWI as usual, staying late to mark up an associate’s paper for publication. The next day she and Coster took a train on a lightly used rail line to the Dutch border. Otto Hahn had given her his mother’s ring and “Frau Professor” was apparently thought to be the wife of the Dutch professor, so the German border guards didn’t stop her. She got out, with ten marks and her summer clothes, and the ring she could sell for money if needed. (The story is much more complex, and given in the Wikipedia article on Meitner.)
Once Meitner was safely out of Nazi Germany, work continued long-distance. Hahn and Strassman at KWI decided to try to replicate the Paris group’s results, and found what they thought was radium (element 88).
Figuring that the neutron hitting uranium-238 was creating uranium-239, which then gave up two alpha particles to become radium-231, they dug a little more carefully, and decided to extract the radium from the sample.
Radium lies directly under barium (element 56) on the periodic table (it was properly understood back then, unlike uranium), and the two elements have an affinity with each other. If there was any radium in the products, barium could be used to draw it out, then it could be separated from the barium without interference from all the other “stuff” in the sample.
Indeed, the barium came out radioactive, indicating that there was radium in it. So it looked like they had found their radium, and the two alpha decays.
But then they couldn’t separate the radium from the barium.
The extraction process used was tested by putting known samples of radium into the barium, and they were separated out without any trouble.
The Light Dawns
Finally they were forced to conclude that the reason they couldn’t find any radium in the barium, is that it was barium.
A radioactive isotope of element 56 was coming out of uranium-239.
Meitner and Frisch finally realized that what was happening. They had gotten together for Christmas in 1838, and were out cross-country skiing having a rather atypical conversation.
What if, they thought, the uranium nucleus were simply splitting? The prevailing model of the nucleus was called the “liquid drop” model, treating it as similar to a drop of liquid; if it were under enough tension that it wanted to break up, a neutron could add just enough “oomph” to happen, just like a very large drop of water wants to split into smaller drops. (Incidentally the liquid drop model, though not the most advanced model, is still good enough to be of some use today.)
However the two pieces would find themselves outside the range of the strong nuclear force, and repel each other quite forcefully. About 200 million electron volts–about a fifth the mass/energy of a proton–would be released as the two pieces flew apart. Where would it come from?
Meitner was able to figure out that the two pieces’ binding energy was high enough compared to the uranium’s binding energy that the 200 MeV would be supplied by that.
It fit.
Nuclear fission was real.
Uranium could be induced to split and release a lot of energy. The lanthanum, technetium and barium were real. It just depended on exactly how the split happened, which particular smaller elements you’d get.
When Frisch told Niels Bohr of this, Bohr literally smacked his own forehead and exclaimed, “What idiots we have been!”
Fermi was also embarrassed; that part of his work bombarding things with neutrons that had to do with uranium turned out to be misinterpreted, and the 1938 Nobel prize he had just been told he would receive was in part awarded for his transuranium “discoveries.” Just in time though; he added a footnote to his acceptance speech to explain what they had just figured out.
In the fullness of time, it developed that those 10 and 20 second reactions Meitner, Hahn and Frisch were seeing were fission products. But the 23 minute reaction really was a decay into element 93, isotope 239.
And it was the small amount of uranium-235 that was fissioning, not the uranium-238.
And we now had a new form of radioactive decay: fission, spontaneous fission. Uranium 236’s most common decay mode is this.
The Bomb
The rest of the story is much more famous, though at the time it was shrouded in secrecy. The US government, alerted by none other than Albert Einstein’s letter to FDR concerning the potential of such massive releases of energy, created the Manhattan Project to build a nuclear bomb.
Much of this early research had been done in Nazi Germany. What if they, too, were working on The Bomb?
It turns out that when uranium-235 is hit by a fairly slow moving neutron, it becomes, for just an instant, uranium-236, which is what fissions into two large pieces. But there are also two or three bare neutrons released; if they can be slowed down and then induced to hit more uranium-235, you can have a chain reaction, each step doubling or even tripling the energy release as more and more uranium-235 catches neutrons and releases yet more neutrons.
However, you need a fairly substantial amount of U-235 for this to work. If it’s a small lump of the stuff, the freed neutrons will probably exit the sample before they find a nucleus to hit. There’s a critical mass that must be brought together for the chain reaction to take off.
The bigger difficulty, of course was that uranium-235 is only a small fraction, less than one percent, of uranium.
So one of the two approaches taken was to try to extract “enriched” uranium-235 from uranium by reacting the uranium with fluorine to create uranium hexafluoride gas, which could then be centrifuged to separate out the slightly lighter U-235. This work was done at Oak Ridge, Tennessee. Once you have the enriched uranium-235, it’s dead easy to make a bomb. Bring two small masses together, enough to make a critical mass, make sure there’s a neutron source nearby, and, KABOOM!!
The other approach involved those transuranics. Uranium-239 was beta decaying into neptunium-239; neptunium 239 was in turn beta decaying into plutonium-239. The two new elements were named to continue the series. Uranium had been named after the planet uranus; the next two elements were named after neptune and pluto (then believed to be a planet).
And it turns out that plutonium-239 is easy to produce; just bombard regular uranium with neutrons–and it too will fission when struck by a neutron. The trick is to get enough of it together close enough that the excess neutrons will find another plutonium-239 atom before exiting the mass. As it happens, Pu-239 must be compressed for it to work. And getting that to happen precisely right was a challenge.
It’s a good thing that the easy-to-make bomb requires the hard-to-make material, and the easy to make material is hard to make a bomb out of.
But we did both.
The U-235 bomb was deemed so simple it wouldn’t need a test. Thus it was dropped on Hiroshima on August 6, 1945 (Germany had surrendered in May of that year, after millions had given their lives to put the mad dictator Hitler down). It worked beautifully, releasing energy equivalent to about 20 thousand tons of TNT, all at once.
The Pu-239 bomb, however, was the first nuclear detonation. It was tested at Trinity site in New Mexico on July 16th, then a second example was dropped on Nagasaki on August 9th.
The bombs killed tens of thousands of people, but likely saved at least ten times as many lives. Had the United States needed to invade the Japanese home islands, there likely would have been two million casualties.
The neutron had gone from being an abstract thing cared about only by some physicists trying to figure out what keeps atoms together…to something as impossible to ignore as a slap across the face.
And this wasn’t even the end of that road.
Obligatory PSAs and Reminders
China is Lower than Whale Shit
Remember Hong Kong!!!
中国是个混蛋 !!!
Zhōngguò shì gè hùndàn !!!
China is asshoe !!!
China is in the White House
Since Wednesday, January 20 at Noon EST, the bought-and-paid for His Fraudulency Joseph Biden has been in the White House. It’s as good as having China in the Oval Office.
Joe Biden is Asshoe
China is in the White House, because Joe Biden is in the White House, and Joe Biden is identically equal to China. China is Asshoe. Therefore, Joe Biden is Asshoe.
But of course the much more important thing to realize:
Joe Biden Didn’t Win
乔*拜登没赢 !!!
Qiáo Bài dēng méi yíng !!!
Joe Biden didn’t win !!!









Thank you for your nice work 🙂
I guess everyone is still in their fast and prayer day?
I was up early need to sleep good night.
Am involved in a WA state public records request lawsuit. Actually eight of the varmints. Guess who is trying to intervene?
None other than the Democrat Central Committee (state version of the DNC).
Guess who they hired as their attorney?
None other than Perkins Coie. Yep, the firm that is under criminal investigation by John Durham, special counsel.
Perkins Coie is trying to muddy Durham’s water and create some exculpatory evidence to show that Durham is on a revenge witchhunt for us DJT voters.
It will fail. Keep us in your prayers.
Will do.
Perkins Coie are TRAITORS of the highest order.
May they all FLEE TO CHINA before this is over.
LEVIATHAN. Run to the belly of LEVIATHAN, you PESTS – you FREAKS – you TRAITORS.
The same group of people every time.
Perkins Coie was who Dominion Antifa Boy Eric Coomer ran to, when he was being put under oath.
They’re anti-American SCUM.
Sinking Ship
The hand is all wrong.
It’s either too low, or not low enough.
😁 😁 😁
…. please Lord make it sooooooooon … 🥺🙏🏼 …
Doubt if Hoe took that much coaxing.
Covid-1984 (@NeverSleever) Tweeted:
A Medical Health Professional Sent Me This…
Clear As Day… A Financial Incentive To Inject People…
So Is This About Health? Or…. https://t.co/sz4MeZUZpl
Medicine has been CORRUPTED beyond belief.
Hitler’s exile of Jewish scientists was one of the most significant “self-pwns” in history.
And Jewish engineers and scientists (Einstein etc)
Alternatively, it was one of Stalin’s messiest victories.
For all our retired or exmil. 😁
Kay (@KayTXTX) Tweeted:
From my Son… 😂😂🤣🤣 https://t.co/ELemRSp2sU
Nuclear weapons have been used in war only twice (so far). Here’s the story of a guy who experienced both of them, and survived.
https://www.history.com/news/the-man-who-survived-two-atomic-bombs
(It is speculated that he was the inspiration for the bowl of petunias.)
That poor dude was studied and his blood and DNA is still studied today.
wtf didn’t he die?
wtf didn’t he develop cancers?
Life it TOUGHER than people think.
SOME SURVIVE.
I read a similar story back in the 70s or 80s (so it wasn’t this guy, who didn’t speak about it until the 00s); the man managed to get home to Nagasaki, stumbled into his house, said “there was a big blue flash” and…there was another big blue flash.
“….just like that one….”
They were both Japanese industrial cities not far apart, and one could expect a fair amount of day trips between the two for salesmen, technicians, truckers, and the like. That nuclear bombs were used was unique, but one wonders if some of those caught up in the relentless bombing of Berlin, February 3rd 1945, went home to try and recover in Dresden when the bombs started falling on February 13th.
Wth does nonce not valid mean?
Ffs!
it posted!
now I know I’m being messed with.
like the endless cycle of you have subscribed to the q tree.please confirm your subscription
We have no end of parties who would “mess with us”. Again, it depends on who is saying what. To complicate things, the same “error glitch” can throw three different error messages to three different people looking at it.
Woo….
OK, it’s cryptography.
When you’re trying to establish a trusted link on a non-trusted network, you play with a “nonce” to give your other party something to play with and hand back to you. It’s part of a protocol between two or more players.
The meaning of “nonce not valid” depends on which player is saying it.
It appears when I try to post. Sometimes
other times stuff vanishesinto the vid and I get an email saying confirm subscription
So you can read this blog without it, but it only shows when you try to post here?
Ahhh Soooo…..
Read and feed… 🙂
Mostly yes.
and FYI over here a nonce is a rock spider/ paedo
That is quite amusing. It’s funny how the rest of the world thinks we speak the same language.
OK, so Wikipoo’s articles on this are overall quite suboptimal. First, you have https://en.wikipedia.org/wiki/Cryptographic_nonce , which sort-of defines it in a way that it might be used for practically anything.
In the article https://en.wikipedia.org/wiki/Transport_Layer_Security , which purports to document the process where it probably bit you, “nonce” occurs precisely once in a sad muddled stew of incomprehensible glorp. The usage is in: “In May 2016, it was reported that dozens of Danish HTTPS-protected websites belonging to Visa Inc. were vulnerable to attacks allowing hackers to inject malicious code and forged content into the browsers of visitors.[266] The attacks worked because the TLS implementation used on the affected servers incorrectly reused random numbers (nonces) that are intended to be used only once, ensuring that each TLS handshake is unique.”
In order to appreciate just what is going on, I’d recommend https://www.cloudflare.com/learning/ssl/what-happens-in-a-tls-handshake/ — which has no uses of “nonce”, but uses better language with “client random”, “server random”, and “premaster secret”, all of which might be considered nonces.
This also clarifies why it is so important to understand WHO is saying “nonce not valid” in order to interpret the error message.
Same language?
Terry Irwin’s job on the Steve Irwin videos was to translate! 😀
Why am I being allowed to comment?
is a hostile takeover of the information superhighway occurring?
Yeah, there is a war, and you poor saps are in CHINA-OCCUPIED TERRITORY.
….as if we aren’t?
It’s hard to say who’s further along, but Australia is looking pretty Poland right now.
China. In a world of hurt and the only way out is to eat crow
Or bats…
Beautiful. From Rural Life group on Gab.
Fall Harvest time.

is some of that pumpkin butter ?
I 💖 pumpkin butter !
and pumpkin ice cream, pumpkin bread, pumpkin pie, pumpkin cake.
and the color of pumpkin.
warm
delicious
nutritious
comfort
🎃
It’s the color of F# to me… warm…
Pumpkin Butter, Apple Butter, Date Butter… but Pumpkin is the warmest, and comforting when it’s getting cold and frosty…
I love pumpkin soup.
When I was a child a East Prussian grandmother adopted me she made the best pumpkin soup.
I learned how to spin wool and knit socks from her. I was poor as hell material wise as kid but very rich with good people in my life and experiences one cannot buy with money.
God is good He put good people thougout my life I am able to draw from the good that life offers that gives life. The simple life is the biggest gift God gives to us.
do you happen to have your Grandmother’s pumpkin soup recipe ?
pretty please ?
I wish I did. I have made pumpkin soup but never taste the same as hers.
She was not my grandmother but I called her that and she embraced me like her own.
I was blessed many people were my grandpa and ants and uncles even though we were not related. My family had issues not good parents but people but I was loved by many people. I was poor but I was rich 🙂
If I find a good pumpkin soup I share. Need to get on the German search engine.
👍 💞
If you are interested I found the East Prussian pumpkin soup on Internet. The recipe is in German I can translate.
The sweet pumpkin soup is a cross of dessert and soup.
One can find normal P soups on internet made with chicken stock and some have apple in. That seems more traditional soups
the East Prussian recipe !
thanks !
I found a recipe that had also English here is the URL Let me know if it works for you ?
https://www-webkoch-de.translate.goog/rezept/kuerbissuppe-aus-ostpreussen-23455?_x_tr_sl=de&_x_tr_tl=en&_x_tr_hl=en&_x_tr_pto=nui,sc
Love it almost looks like my collection 🙂
My daughter told me because people were locked in many women are preserving instead of freezing their produce and fruit.
May we enjoy the ingatherings of this season.
The pictures you post are nourishing to the soul. Thank you 🙂
Where I live surround the countryside looks like that . From my home I can see the hills from KY.
Wow 😊 What talent !!!!!!!!!!!!
Enjoyable! The amazing history of that time.
We’re seeing it now, as CHINAZIA annexes the West by STEALTH.
And now for some FUN!!! 😃
https://rumble.com/vnhkyh-lets-go-brandon-is-paged-at-chicago-ohare.html
👍
wow
check out La Palma right now…Live feed…
La Palma Volcano Eruption, the Canary Islands (Feed #2)” width=”1280″ height=”720″ src=”https://www.youtube.com/embed/HtpEE276x_Y?feature=oembed” frameborder=”0″ allow=”accelerometer; autoplay; clipboard-write; encrypted-media; gyroscope; picture-in-picture; web-share” referrerpolicy=”strict-origin-when-cross-origin” allowfullscreen>
unreal
western flank blows out…yesterday…watch…
One of the other videos offered after the first one, alludes to the possibility of the banana crop being lost.
Pat Frederick needs to take heed.
Pat’s on a banana run this morning.
Good weather for marshmallows 👍
c’mon man…not to be Deb Downer or anything but…
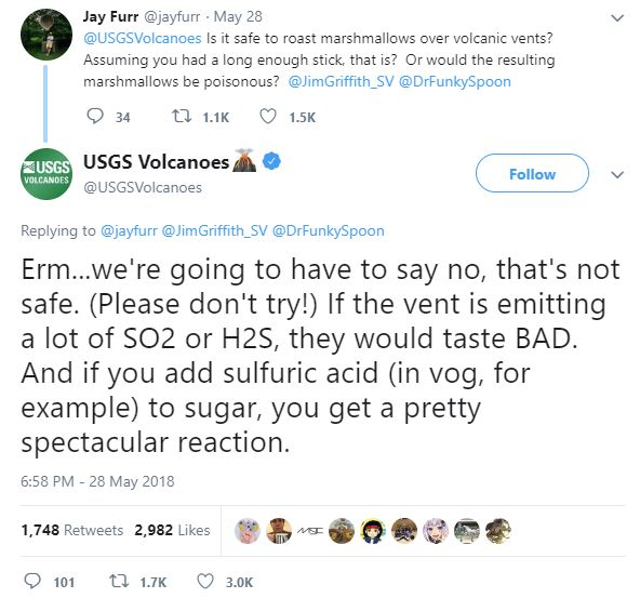
How about kebabs?
sheesh ?
I remember having one of those “genius of Disney” moments when I first encountered the Ohana restaurant at WDW.
“Food on a stick over a fire” has been the prototypical form of cooking since Homo erectus [“H. erectus is associated with the Acheulean stone tool industry, and is postulated to have been the earliest human ancestor capable of using fire {emph added}, hunting and gathering in coordinated groups, caring for injured or sick group members, and possibly seafaring and art” — Wikipoo].
It is generally held that the bulk of modern lifestyles are descended from six cradles of civilization — https://en.wikipedia.org/wiki/Cradle_of_civilization . “Food on a stick over a fire” was valid in all six, and continues to be valid today worldwide.
WDW was designed for a substantially more international mix of guests. It is old-style Disney genius that they included a restaurant with THE most universal cuisine on the planet to cater to all guests. Of course, then they dressed it up in some goofball pseudo-Polynesian trappings….
But, assuming you showered and clothed them, you could have Homo erectus, Neanderthals, Cro-Magnons, Incas, Olmecs, Egyptians, Sumerians, Harappans, and Xia Dynasty Chinese scattered throughout the dining room and notice them just as much as the Japanese, Arabians, Europeans, and Hispanics that are actually there today (amongst the non-Domestic US guests).
IIRC, however, they actually use a hardwood firepit instead of a volcanic vent. That might have been a bridge too far with their Polynesian theming.
food-on-stick-over-fire + homo erectus = just one of many primal urges
the paleo diet for homo erectus…

Ringo knew how to pound out a rhythm.
As mentioned, of course, the volcanic vent would have made the food smell and taste like farts and rotten eggs.
Like food from a California homeless camp?
Apparently it’s okay for cooking Dino omelettes though…
The New Zealand maoris have been cooking in steam vents since they got there.
seriously, when you first go to Rotorua (highly active area) the first thing you notice is the stench of sulpha. It is literally retch inducing. After 24 hours you can’t smell it at all and you can eat food cooked in steam vents and only taste the food flavours.
they have one of the earliest geothermal power station at Waikato.
Take that back!!! Kalbo is our FREN!!!
That’s it! “Kalbokalbs’ Kebab Korner!” Sure to be a Kommercial success!!!
Well, the other thing we used to do with marshmallows when we were kids is put them on a stick, and hold the marshmallow directly in the fire, until it caught fire.
At which point you had a kind of sticky flaming napalm that would stick to anything you flung it at.
Or just use the stick to launch the flaming marshmallow up into the night sky, like a Roman Candle.
So… good weather for marshmallows… 👍😂
And hope the fuck it didn’t land on you. Or someone else’s land on you. Even if the flame went out it burnt like hell
Lots of volcanic activity the past few months.
listen to brandon goofus speaking in Illinois on Thursday…
who does it better ?
FJB
Gail Combs did a HUGE transcript of Professor David Clements interviewing Col. John Mills – I put it up as a post with the video embedded.
Enjoy!
https://www.theqtree.com/2021/10/09/transcript-professor-david-clements-interviews-colonel-john-mills-the-deep-state-up-close-and-personal/
Thanks, Steve, for another great Saturday thread!
👏 😁
Good stuff…very informational.
Love the rant!
😄 👍
And yes…𝕁𝕠𝕖 𝔹𝕚𝕕𝕖𝕟 𝕕𝕚𝕕𝕟’𝕥 𝕨𝕚𝕟!
Found these on Gab:

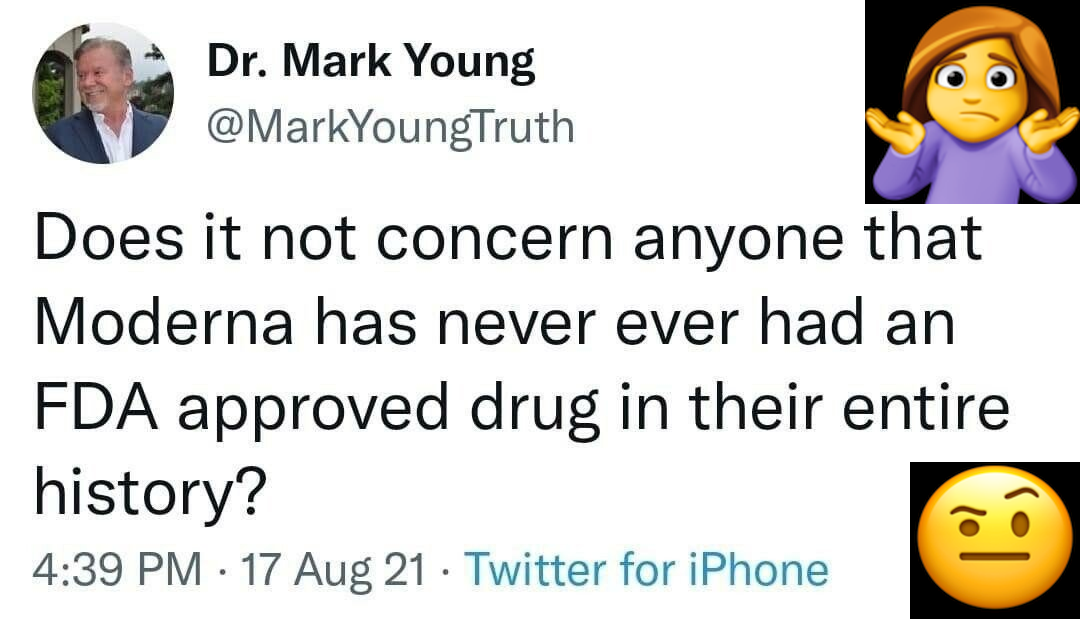
So I did a little checking…
Moderna hasn’t been around very long.
Moderna – Wikipedia
…………….From Wikipoo:
In 2010, ModeRNA Therapeutics was formed to commercialize the research of stem cell biologist Derrick Rossi. Rossi had discovered the works of Hungarian biochemist Katalin Karikó on RNA-mediated immune activation, which resulted in the co-discovery with American immunologist Drew Weissman of the nucleoside modifications that suppress the immunogenicity of RNA. This technology would later be licensed and used by Moderna and BioNTech to develop COVID-19 vaccines.[14]
[…]
In March 2013, Moderna and AstraZeneca signed a five-year exclusive option agreement to discover, develop, and commercialize mRNA for treatments in the therapeutic areas of cardiovascular, metabolic, and renal diseases, and selected targets for cancer.
[…]
In 2018, the company rebranded as “Moderna Inc.” and further increased its portfolio of vaccine development
[…]
The Moderna COVID‑19 vaccine (pINN: elasomeran[33]), codenamed mRNA-1273 and sold under the brand name Spikevax,[34] is a COVID-19 vaccine developed by American company Moderna, the United States National Institute of Allergy and Infectious Diseases (NIAID) and the Biomedical Advanced Research and Development Authority (BARDA). It is authorized for use in people aged twelve years and older in some jurisdictions and for people eighteen years and older in other jurisdictions to provide protection against COVID-19 which is caused by infection by the SARS-CoV-2 virus.[35][34][36][37] It is designed to be administered as two or three 0.5 mL doses given by intramuscular injection at an interval of at least 28 days apart.[38][39][40][41]
It is an RNA vaccine composed of nucleoside-modified mRNA (modRNA) encoding a spike protein of SARS-CoV-2, which is encapsulated in lipid nanoparticles.[42]
……………………….More at link.
Moderna and Theranos have a lot of similarities at the founding and board levels. GLOWING similarities.
Hy am I not surprised?
Clip from Hannity’s thursday night show:
https://youtu.be/2FRAbTBas-Q
Progress.
And if you haven’t had Covid, you don’t need to take the vaccine either.
So in conclusion, whether you’ve had Covid or you haven’t, you don’t need to take the vaccine — and you shouldn’t.
Why would you turn yourself into a medical experiment for Big Pharma and their untested experimental gene-therapy project?
Nevermind the Nazi lawlessness of experimenting on humans without their knowledge or consent, but they’re making billions, and they’re not even paying you a nickel to be their lab rat.
Why would you want to be a volunteer lab rat for Big Pharma?
Were you dropped on your head when you were little?
Or… more recently?
Steve Bannon: ‘I stand with Trump and the Constitution
’https://populist.press/steve-bannon-says-stand-trump-tells-jan-6-committee-not-comply-subpoena-html/
Virginia Judge Hands Victory to Parents, Sides Against Prosecutor on School Board Recall
.
https://xyz.frankspeech.com/article/virginia-judge-hands-victory-parents-sides-against-prosecutor-school-board-recall
https://twitter.com/disclosetv/status/1446489655973761026
does he grow poppies at any of those ostentatiously extravagant show places ?
Nope!
Just from back in the “old” country.
He grows them FOR to have the show places, not to grow AT them.
But “poppies” is certainly the special *password* for his dealings …
From our tax money ? Just asking for a friend I. 🙂
“Leaked documents reveal just how concerned the Marine Corps was about Lt. Col. Stuart Scheller’s call for ‘revolution”
https://taskandpurpose.com/news/marine-corps-lt-col-stuart-scheller-court-martial/
If this is correct I can see some things differently.
Revolution that is new news !!!
I think they just seized on that loose word but he de-fanged them and the fake news.
They are making an example of him so no other will critique the military .
The military knows they are participating in an silent revolution in this country. The takeover is apparent and without the military this would not happen. They project what they are doing on their victims just like Stalin.
Buried in the article is Scheller calling out Marine Corps, General McKenzie, Commander Central Command.
^^^^ Marine Corps protecting its turf. A major aspect in the butt hurt feelings over Scheller calling out the ENTIRE Chain of Command over Afghanistan debacle.
I think he saw what was coming and forestalled the fake news and shyster lawyers creating any fiction.
For Caturday on the Qtree 🙂 (H/T TheTrueNolan on chiefio’s)
Buitengebieden (@buitengebieden_) Tweeted:
Sound on.. 😂
#caturday https://t.co/G0l9konuHB
https://twitter.com/buitengebieden_/status/1446757023551479809?s=20
And another from TTN, looks like whoever does predictions/modeling for the NHS in the UK were/are doing it for the CAGW madness…
Bad science begets bad science…
4,000,000 Americans WILL DIE…… 😱 😱 😱
Shades of James Hansen and his “death trains”…..
Babylon Bee has some help for those confused as to how they identify 


(H/T Bayou Renaissance Man via another ian over on chiefio’s )
https://babylonbee.com/news/what-is-your-intersectional-identity-use-this-handy-chart-to-find-out
What Is Your Oppressed Intersectional Identity?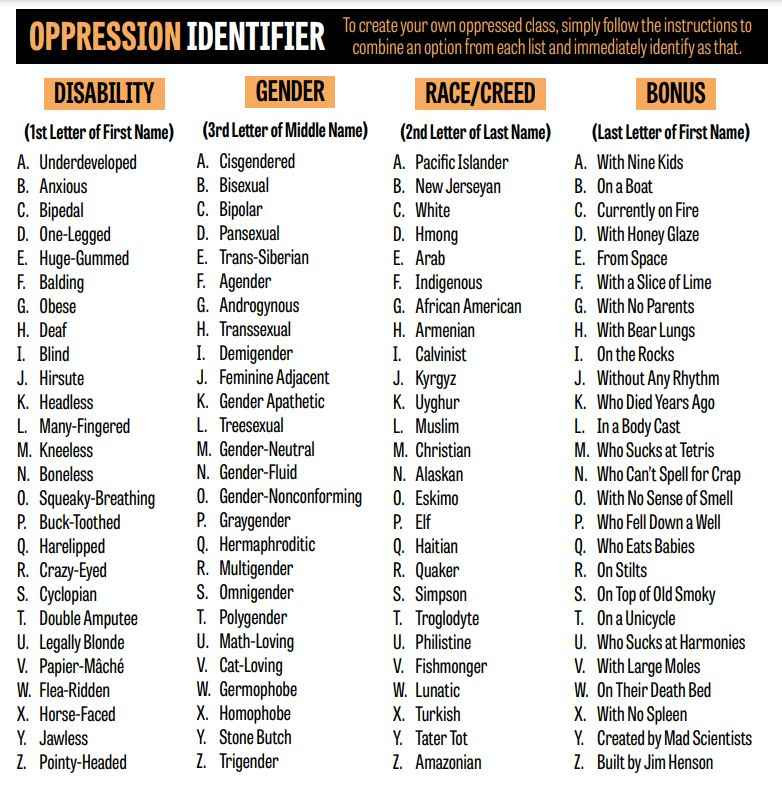
Use This Handy Chart To Find Out!
So that makes me a bipedal demigender Eskimo with nine kids 🙂
I always wondered why I like the cold so much … ❄ ☃ 🎄
What did you use for a middle name? The others are consistent with “Cuppa Covfefe.”
Irish 😀 🍀 ☕ 🍀
Some days, heavy on the Irish, hold the covfefe 🙂
I didn’t think it was “idiot.” Doesn’t fit.
Thanks 🙂
I was thinking of “Hot” as a “middle name”, but polygender is even worse then demigender (at least it’s not hemidemisemigender – like in music with hemidemisemiquavers).
That “Bonus” column would be good for a poem or a G&S parody patter song…
We’ll just stop at Cyclopian.
Careful lest you auto-doxx.
Exactly.
Almost as bad as Auto-Tune… 🎶 🎵
Hey Cuz!! 👋👋👋

No trigger finger discipline!
One eye, one trigger, whaddaya expect??
Perfection, of course. 😎
That’s our PGROUP!! 💖
No issue with cross-dominance here.
.. 😳 .. 👁 … 👀
the ‘eyes’ have it .. bwahahahahaha
… 👁 ..
I can’t ride a bike anyways .. 😑🤚 … bwahahahahaha
…. seriously … total coward ..
https://twitter.com/YoussefDisla/status/1446239675904151561?s=20
Only 7.60 a gallon for regular so what’s he bitching about?
/sarc.
First time I’ve seen US prices higher than here…
Not that the greens aren’t trying (in a number of ways…)…..
More seriously, I remember the headaches gas stations had when they had to post prices over a dollar for the first time. Many would just post the cents and expect the customers to assume the dollar, at least until their signs got replaced with ones that had three useful digits (the 9/10ths of a cent was usually unchangeable). Others would hang a kludged up 1 before the sign.
With digital pumps, not being able to input 10.009 could be a big issue!
When I moved to US gas was 25cent-35cent a gallon same as bread I think my memory is not failing me ?
Anyone remember?
It certainly was at that level at some point…60s maybe?
Yes I came in 64.
Ha my husband’s first job he made $95 a week.
So prices were accordingly.
San Jose, CA. Started driving in 71. So, it was 71-72. Routinely paid $.30- $.35. Occasionally ranged $.25 – $.35. A brief period, gas was in the teens. Lowest I ever paid was $.14, on one occasion. Many a run to Santa Cruz. 🙂
Teenager flipping burgers in them days, always hawked the cheapo gas stations.
In the mid-late 80s gasoline in Southern CA was generally a dime cheaper than Colorado.
singing…I worked in Palo Alto from early 70s to early 80s. Lived in Santa Cruz, and my commute was 112 miles a day round trip.
i clearly remember gasoline being $.25/gallon.
and waiting in line for 45 minutes to tank up.
Not a memory problem.
Oh good it seemed like a nice dream. 🙂
Yes, I remember. In the 1950s and 60s. Went up to $0.60 in the seventies with the oil FAUX embargo. (Trucker told mr the NJ refineries didn’t know where to put all the gas. the panic WAS ALL MADE UP in order to increase the price dramatically.)
WTH ever happened to gas wars?
sigh….
And the stuff they used to give you (as did the banks)…
Steak knives, books, glasses, etc. And they’d send out a grease-monkey (or apprentice) or two to clean the windshield, check oil and water (and top up, if needed), and sometimes even clean the other windows and/or check your tire pressure. AND pump the gas, too…..
Back in the good old days, when the customer was king (or queen) [and KNEW WHICH ONE THEY WERE!!!]…
What happened?????
Seems all that money floating around back then floated away from us… into the pockets of the Hyper-Rich…
We used to drive coast-to-coast every summer (but one) between 1960 and 1973… and that on ONE INCOME. And we neither starved to death, nor slept in fleabag motels, nor scrimped on seeing attractions/museums/etc. And Stuckey’s Pecan Log Rolls were not out of reach either… sigh….
We don’t just need the diamonds back; we need the pearls, rubies, emeralds, and all the other gems that we used to have back in the 1950s and 1960s that the deep state took away…
TIME TO TAKE BACK WHAT IS RIGHTFULLY OURS!!!!!!!
AMEN!!!
Just looked it up. Gorda by the sea resort area. Just north of san simeon ie Hearst Castle area. They are the only place until you get to Big Sur then on to Carmel and Monterey.
They are engaging in gouging which is reportable, but good luck getting the state to help.
According to the reviews, whoever owns this place engages in routine unchecked gouging. $5 can drinks that run 2 bucks, charging for bathroom use with purchases, etc.
They’ve always been high and gougers… and then go back to Highway 1 and get stuck behind “the Dorothy Chandler Pavilion on wheels” for the rest of the way (or most of it)…
…. Rosebud…..
If 8 go up that way as beautiful as it is, its a long long drive to Monterey. Ill just cruise the 101 and the five.
The coast road between Los Angeles (Pacific Coast Highway) and San Francisco (Highway 1) is one of the most beautiful drives on the planet.
It’s also quite dangerous, challenging, prone to road closures, and slow (it’s almost impossible to do in one day) — not to mention that there are a couple of day’s worth of adventures just off the road — like Malibu, downtown Santa Barbara, Hearst Castle, Big Sur, Carmel, Monterey, and Santa Cruz.
We went up pch to hearst and then to carmel. One of my top 5 drives ever. And a nice trip.
https://www.discover-central-california.com/mcway-falls.html .
Past big sur not sure where we saw a few cars parked at the side of the road. I made mr gil stop. It was a little trail going inland, not on the beach. Turns out it was a beautiful little creek and pond. A slight hike just further back wS a natural hot springs. On the walk it i was covered with 🐞. Natures beautiful moments.
Verse of the Day for Saturday, October 9, 2021
✟
“But the meek shall inherit the earth; and shall delight themselves in the abundance of peace.”
Psalms 37:11 (KJV)
Thank You, Jesus, for blessings received and prayers answered!!!
BE MY VOICE
PRAYING ON THE ARMOR OF GOD
Father God, I now follow your command to put on the full armor of God, because my battle is not against flesh and blood but against rulers, authorities, the powers of this dark world and against spiritual forces of evil in the unseen world.
I first pray on the Belt of Truth that it may be buckled around my waist, may I be centered and encircled by your truth dear Lord. Hem me inside all that is true and right, and may I be protected and held up by the truth of your living word, in my Lord Jesus name.
I pray on the Breastplate of righteousness, please protect my vital organs and my inner man, cover my integrity, my spirit, and my soul. Guard my heart for it is the wellspring of life, please strengthen and guard the most vulnerable places in my life with that which is right, good, and noble that I might not receive a fatal blow from the enemy, in my Lord Jesus name.
I pray on the Gospel Shoes of Peace. I choose to stand in the shoes of your good news, and on the firm foundation of my Lord and Savior Jesus Christ, the solid eternal rock. All other ground is sinking sand, I pray that I will not slip or fall, but that my feet would be firmly fitted on your lordship, my Lord Jesus. I choose to stand on you, so that the peace of God, which transcends all understanding will guard my heart and mind in Christ Jesus, the eternal Rock of Ages. I receive your holy peace now my Lord, from the sole of my feet to the crown of my head, in my Lord Jesus name.
I pray the Shield of Faith into my hand now. As I take up the shield of faith, I ask that you might extinguish every dart and arrow, that is launched from the enemy to take me down spiritually, physically, mentally, emotionally, and every attempt of the enemy to destroy my joy. I ask that my faith in you would make it flame out. Extinguish every flaming arrow that would come against me, my life, my family, my home, or my ministry. May my faith always be out in front of me like a shield. Give me the courage to “faith my fears” by choosing to walk by faith and not by sight, in my Lord Jesus name.
I pray on the Helmet of Salvation, that you might protect my mind from the thoughts that can lead me astray. I choose to take every thought captive, and arrest all intentioned ideas and motives that would harm others, or distract me from your holy will for me. I submit every captured thought to the Lordship of my Lord Jesus Christ, and ask that you would imprison those thoughts that are not of you my Lord. Transform my mind and renew my thinking that I may think God thoughts, and have a sober mind that is focused on your glory. Please protect me from being double minded that I may allow my mind, I reject to live an earthly life, because I choose to live a holy one, governed by you My Lord Jesus, the prince of peace, please have my mind to be saturated with the holy mind of Christ, in my Lord Jesus name.
Finally, I take up the Sword of the Spirit which is the holy word of God, I pray this powerful offensive weapon into my hand, and ask that your holy word would be fitting for every encounter I face. As the enemy gets close to me, please give me the insight, wisdom, and skill to wield the word of God to drive away the enemy, in my Lord Jesus name.
May the enemy and his team flee from me, upon hearing the word of God spoken by the power and direction of the Holy Spirit. Give me the sword of the spirit to cut through the wiles of the devil, so that I may discern the schemes of the enemy when he is near.
With all kinds of prayers, supplication, and intercession I pray to you my Lord God as the one who fights my battles. Now that I’m in your holy powerful armor, I walk away covered and ready to face my day as you go before me, and please protect me in the midst of the spiritual warfare in this unseen world, in my Lord Jesus name.
Thank you my Lord, for the spiritual weapons of armor and prayer that you have given me. It is written no weapon formed against me shall prosper, and you will refute every tongue that accuses me.
Thank you Father God, my Lord Jesus and the Holy Spirit, that I am more than a conqueror in my Lord Jesus. I pray all of this in the mighty name of my Lord God and Savior Jesus Christ, amen.
Glen lives, up
Barrels of fun 🙂 Good golly, Ms. Holl(e)y…
(12?)…
Three deuces (2-barrels).
OK, thanks… been a loooong time since I’ve seen a carb…
(On a low-carb diet 🙂 )…..
Had a Taurus SHO with the Yamaha engine that looked like a plate of lasagna on the top. Great engine, lousy car (first year Ford had water-based paint, and it showed)… I heard someone had their engine balanced and modded and could reach 11K RPM with it… not sure for how long 🙂
There wasn’t much room for a clutch, too, so the first one only lasted a few months, after which Ford acknowledged the design problem, and put in a MUCH stiffer, quasi-racing clutch. Felt like I was doing leg presses every time I shifted…
.. all around the town,
‘the kids play ring-a-rosie
London bridge is falling down
boys and girls together
me and Rosie O’Roark
trip the light fantastic on the sidewalks of …
New York .. 🎶🎵

Jabbed Jesuits Catch COVID, Suffer Brain Clots
.
Dean in coma as virus hits fully vaxxed Pontifical Gregorian University
.
https://www.churchmilitant.com/news/article/jabbed-jesuits-catch-covid-suffer-brain-clots
I didn’t think Gregorian was actually Jesuit. I mean, Jesuits attend, but it’s not strictly one order.
The dean is in an induced coma and according to sources in the linked article, half of his brain is dead. He was in good health before getting “fully vaccinated.”
Laramie, WY.
Well that area is off my list of places to move.
Robby Starbuck (@robbystarbuck) Tweeted:
Meet Grace. She’s a 16 year old girl who’s showing more courage than most men right now. She’s refusing to comply with the mask mandate her Wyoming school board is trying to force. She was fined and arrested. Here’s her arrest. She’s a hero.
Support her: https://t.co/reUHkcHjHx https://t.co/fs0Xr5mz03
University town.
The area around Jackson Hole, and much of I-80, tend to be more “liberal” as I understand it, though I suspect the I-80 corridor (minus Laramie) is still pretty conservative by nationwide standards.
Ok. All these uni towns are destroying the areas.
Asshoe school LOCKED DOWN over a student not wearing a mask.
Arrested the 16 yo.
School “officials” over the top idiots.
LE 100% should NOT enforce mask anything.
Congrats to the youngster and parent for standing up for her rights.
WHERE ARE THE OTHER PARENTS of students.
Here’s some truth. These people are Nazis. People who are living there are living with literal Nazis. But these particular Nazis are run by ELECTED school boards. People WANT this shit.
We are Americans. We don’t HAVE to live where the literal Nazis live. Of course it isn’t EASY to move to where your kids aren’t treated like Jews in Occupied France. But it wasn’t EASY for our ancestors to settle the West by wagon train, either.
It’s time to ABANDON the places where these Nazis congregate, and form sane communities where they are VERBOTEN.
Yes indeed. These people are intent on perversion of society.
Seems there’s a herd of geldings in Wyoming ..

Good Morning Mr. Steve.
I just found 2 1980 gold South African Kruggerands in my Dad’s belongings.
Whatever shall I do with them?
Here’s some snacks while you’re thinking it over. 😁😋🥓☺🤗🥰


If it were ME, I’d keep them. (I am assuming your dad has no further use for them.) But then, I tend to accumulate such things over the years.
I don’t know how desperately you need money. But I do have a suspicion the price is going to break out of its current narrow range. It’s up to you to judge which way it will break, up or down: Hold for a while if you think upwards, dump if you think downward.
TY!
And thanks for the bacon!!
YW.
Crap!!! That’s what I forgot at the grocery store. 🙁
AHHHHHH!!!

Never fear, plan B is nearly as good as Plan A.
Bacon and guacamole sandwiches. There’s an idea.
What, no love for bacon meatballs?? 😜
My husband bought bacon sausage. I roasted them in the oven with red cabbage wedges and served with apple sauce. He had a beer with it I a class of water. Was very delicious.
Saturday at 11:30 p.m. ET
Sunday at 2 p.m. ET
This Week: Joe Biden’s bumbling speech fantastic faceplant in Michigan draws hundreds of protesters chanting “Let’s go Brandon.” Fauci, the mean Christmas elf, walks back his holiday COVID-19 restrictions. George Soros’ new funded protesters tactic: harassing senators like Kyrsten Sinema in public places! The world’s first male Playboy cover model will make thousands of men actually just “read the articles” this month. A scary indoctrination song that even this group of preschoolers want no part of.
Archbishop Viganò: Vaccines made with fetal tissue are a ‘human sacrifice of innocent victims offered to Satan’
.
‘The most innocent and defenseless creature, the baby in the womb in the third month of gestation, is sacrificed and dismembered in order to extract tissue from his still palpitating body with which to produce a non-cure, a non-vaccine, which not only does not heal from the virus, but in all likelihood causes a greater percentage of death than Covid itself, especially in the elderly or those who are sick.’
.
https://www.lifesitenews.com/blogs/archbishop-vigano-vaccines-made-with-fetal-tissue-are-a-human-sacrifice-of-innocent-victims-offered-to-satan/
Some of these babies were sacrificed much later than three months in the womb.
There are places, at least over here, that are pushing after-birth abortion. There was mention of something like six months in the Netherlands…
These people are SICK.
Yes
They do not know it but in my mind they are just as evil as Nazis.
Same mindset just a different century. All that happened is the EVIL went underground for a few decades.
They can’t unload, and when they finally DO unload, they aren’t given time to load empty containers to take back with them. So there’s even a container shortage in some places while others are buried under empty containers.
In la, plenty of workers, even per diem, but not allowed to unload, just sitting.
Chiefio has a deep dive on the problem. Seems Newscum and the oppressive DEMONRAT regulatory mora$$ in Kalifornistan in making matters much worse. For instance, drayage (and other) trucks must replace their engines with more “efficient” ones, drivers are severely limited in the hours they are allowed to drive and even where they can park to take their breaks, etc., etc., etc. But NAZI-Nan and her NAZI-Nephew will get whatever they want, whenever they want…
https://chiefio.wordpress.com/2021/10/06/logistics-california-diesel-truckers-you
(He’s heading out of CA, but lots of folks are doing the same thing – so costs are pretty high to escape high costs… sigh…)…..
My Sherwin Williams buddy says after weeks of circling the ports, containers are put in 30day quarantine (WuFlu) at/near the port before they’re made accessible. A huge amount of paint ingredients/additives, solvents, etc., come from Europe and Asshoes. Alternatively, they can load a plane and have inventory delivered PRONTO for $20K a trip with no delay. Guess WuFlu doesn’t enter US on cargo planes.🙄
They’re out of many products for the automotive industry with no date in sight of inventory replenishment. As you know, WuFlu and lack of electricity has caused manufacturing to slow/cease in other parts of the world. Other big manufacturers are faring even worse and trying to buy inventory from them. So much for globalization eh? The whole world suffers, especially the US because we sent our mfg over there. Remember when the highest bidder got the PPE first???
So FYI, don’t have a wreck! You may not get your car back very quickly because they don’t have the ingredients to paint your parts to match your vehicle. And oh yeah, they’re implementing their 3rd price increase(11%) for the year this month, so expect price of repairs, Insurance premiums, and new cars to increase. After all, Big Corporations don’t just suck up higher taxes, they pass them down to their customers, me and you. More Build Back Better Bidenflation Bull$hit $$$!! 🤬 🤬
Sort of a Billed-Back Brandon…
Too many Buh Buh Buh Beez is BBBBBAD!!!
Yep. Like BeezleBubba…
BuhZackly.
>>”So much for globalization eh?”<<
They can turn the spigot OFF, and they can turn the spigot ON.
Just like the “vax” / “boosters” ongoing scam – where one puts ones very LIFE into the hands of those who don’t cherish life (for other than themselves, of course!).
No one in their right mind would allow others to have such power – it ALWAYS ends up with slavery.
The TRUE power belongs NOT to megalomaniacal humans – but solely to God.
Is Sherwin Williams trying to “cover the earth” with container ships? 😆


My daughter worked some time ago at Sherwin Williams in Chicago. She was working in department developing Automotive paint.
Hope it wasn’t the stuff that peeled off of my Taurus SHO 😀
(Although that could have been something to do with the, erm, 5000 watts of car-stereo amps I had in it, and the 15″ subwoofers… Yep, young and geeky was I )…..
(And, yes, it seems that audio watts are not the same as REAL watts, maybe there’s some extra power coming in from hyperspace… cf Rodriguez from Stereo Review)…
(And in my own defense, I NEVER paid $100.00 for a three-foot Monster Cable 😀 )…
She is a physical chemist specializing in surface enhancement. She must be good she makes lot of money. She is very good in what she does.
Sorry about your Taurus. We used to own one like that it was an experience 🙂
Here’s one of the things I loved about that car 🙂 😎

And an article about it (from a motorcycle magazine, no less – Yamaha being a motorcycle specialist). Seems the redline was 7500 RPM, could go much higher except the alternator, A/C, air pump, etc. couldn’t handle it)..
https://www.rideapart.com/news/264846/that-time-yamaha-built-ford-an-engine-and-changed-the-world
The motor look need 🙂
Mine spend lots of time with the mechanic otherwise was a fun car.
Yep. Spot on post. good insights. A little clarification on the “quarantine” required before entering port.
Merchant Mariner son has been on a two ships. Seven month obligation on each ship. He has told me quarantine varies between country. Usually five to seven days. Has been two weeks. This usually, not always, INCLUDES time at sea from the last port.
CA and other dumb ass states may dictate 30 day quarantine. But at some point, the backlog balances out and becomes fairly steady.
With Fall arriving, Thanksgiving, Christmas and New year, ports are prolly the busiest of the year.
Im seeing lots of meat going bad. No one will pay that. Pasta routinely low stock. Ground beef and ground turkey low.
CHINA JOE.
China’s satanic threat – if we can’t run the world, we’ll FUCK IT UP.
Don’t forget the corollary – if we CAN run the world, we’ll FUCK IT UP.
THAT’s how we know who the Enemy is (or at least the form that the Enemy chooses to cloak himself in, for now).
The Shy-Knee are feeling the HEAT.
It’s unfortunate for ALL peoples – even their own populace.
Like a bad divorce between couples who have come to hate each other. “If I can’t have the house, I’ll burn it down.”
“There’s no Jesus in that.”
Local Smith’s (Krogers) meat prices are stoopid high. Just before they have to toss the meat, the meat gets a healthy mark down.
Folks like me swoop in, buy up the BEST deals, most affordable meat.
Meat good for at least a year. Have read two years.
I look at ralphs but their good markdowns are too late in the freshness. I found 10 half lb bags of pistachios 2 days expired and asked for a sale price. They said they had to throw them out.
My Dad made the best steaks in the world. His rule was to always buy the after-date steaks. Aged meat is better.
I always buy the steaks that are on sale because of the date.
My grandfather in West-by-God, who farmed for most of his 96 years in addition to his regular job, wouldn’t eat bananas unless they were pretty much brown. Said they were sweeter that way. I figure he knew what he was talking about, as he (and my Dad) could grow anything; they could probably make bricks take root…
[Folks over here think bananas are ripe when they’re still green. If they get brown, they get pitched… sigh… ]
Grocery store bananas are never the same after you’ve eaten an apple banana, or an ice cream banana, or any of the other varieties that grow in tropical places like Hawaii. And most of them you eat when they are brown.
Yep. Some of the ones here look like plantains 🙂
I’ve heard from a Scottish friend of mine that they can grow bananas and pineapple on the west coast of Scotland due to the mild climate caused by the Gulf Stream. Not sure if it’s every year, but that sounds interesting, even if they don’t produce enough to export to beautiful downtown Deutschland (to paraphrase Johnny Carson)…
(I used to live near “Beautiful Downtown Burbank” 😮 )…..
Wow, that’s cool. Most of Scotland is COLD.
Years ago, my Dad gave me 7 little banana plants from his neighbor across the street. I planted in my back yard. They spread like crazy! Took many years, but i finally got a stalk. I cut it down and let it ripen on the back porch. I had no idea if they were edible. My Dad came over and had to eat one in front of me to convince me. OMG!!! Those little things are some of the MOST DELICIOUS fruit I’ve ever had!!! 🤸♀️ 🤸♀️ 🤸♀️
I’ve finally learn to go look for petals of that flower on the ground, then look up to see if there’s a stalk. Haven’t had any for many years. The plants only make bananas if they get YUGE!! The plants die back in the winter and come back bigger the following year. BUT, if you have a freeze, that seems to set them back to zero and they need 2 years before they get big enough to produce fruit. So this year Texas had an Ice Age and there was no summer fruit. If this winter is mild w/no freeze, I could have some yumminess next summer. 🤞 😋 🤞
Dwarf Bananas
The great majority of commerical bananas are clones of the “Cavendish” variety. They became extremely popular because they shipped well, NOT because they tasted particularly good.
I’ll bet they are all radioactive, thanks to the potassium…and the inevitable K-40 tagalongs.
The banana equivalent dose is based on the K in a banana.
It’s actually kind of silly, but it IS geek humor after all.
If I eat a banana, the K in it either replaces K already in my body…or passes right through.
The K in the body, of course, is also radioactive due to being the same mix of isotopes as the banana’s K. So you get no actual dose from a banana as eating it changes nothing, potassium wise (unless you’re deficient, in which case you have bigger problems than the dose).
Yes these are best tasting aromatic bananas you can get.
Nothing like the “Cavendish” type.
I also have them in my garden.
After you harvest the bunch. You need to cut the tree. The new baby ones will quickly grow and take their place.
If you want more plants you need to cut the baby ones from their mother once they are about 2 foot high. Cut off the root of the baby right next to its mother stem (it is connected at the stem, but has developed separate roots) and plant the baby at a different place.
Yeah, the little bitty ones are GOOD.
My husband went to Kroger bought organic chop meat the price has not changed.
Good steaks at Whole Foods I pay for two $31- $37. I am not sure what it is now.
I ask my husband how prices are he has not noticed much difference here . He said ” what we buy has not increased or very little. People put a lot of junk in their cards.
Bacon is $ 5- $6 for a pound without nitrite and uncured.
I wonder if prices are adjusted higher where one lives?
A case study of real results from AZ shot.
Including ct.
https://mobile.twitter.com/DrJohnB2/status/1446581695717724161
We CAN break the tyrannical spell that has been cast over the world—but we need you to stand behind us, stand beside us, and, if needed, stand in front of us.
Stucky QOTD: Colluders – The Burning Platform
.
I wish we could all be more specific and careful with languaging like this.
It’s the CCP, and even more specifically the willing members of the CCP, who are lower than whale shit, not “China” itself.
To say “China” throws any chinese that would oppose the CCP in the same toilet with willing members of the CCP.
Worse, China is bigger than the CCP, so to say “China” makes the CCP bigger than it really is. The CCP loves to be identified as “China”, rather than “the regime controlling the nation of China”.
While I’m at this, let me also say I am guilty of doing this exact and very same thing. No glass houses here.
The CCP is our enemy, not “China”. China is merely a place, a location. It’s the communist douchebags running that place who are lower than whale shit.
>>”I am guilty of doing this exact and very same thing”<<
As am I.
But at the same time, I absolutely LOATHE their adoption of “Duality” as their primary credo – to my mind, it stinks of the “As above. so below” fallacious theology, so that’s an extra reason to condemn their misguided culture.
>>”The CCP is our enemy”<<
I would say that they are the obvious FACE (a front) of our true Enemy, which still remains somewhat hidden. Our REAL enemy loves them some proxies / masks!
China – the country – is captured, and even Xi has his masters.
The real target is those who pull the (Xi’s, et al.) strings.
The CB is WW; the CCP is but a puppet.
IMO.
From 12-07-17 (#299):
“[…]
Rothschilds (cult leaders)(church)(P)
Banks / Financial Institutions
WW Gov Control
Gov Controls People
SA
Oil Tech Sex/Children
SA Controls (assigned) US / UK Politicians / Tech Co’s (primary)
Soros
Controls organizations of people (create division / brainwash) + management / operator of slush funds (personal net worth never reduces think DOJ settlements Consumer Iran Enviro pacts etc etc)
/_\ – Rock (past)(auth over followers)
_\ (present)
(Future)
Order is critical.
Strings cut to US/UK.
Expand your thinking.
Swamp drain.
1 – sexual harassment exit + future
….
[R] – No.
Bomb away.
[…]”
Vigilant Citizen has a number of articles on what the elite think of children. These are “Do Not Look Away” articles and situations…
And they’re appalling.
THIS is what we’re up against. No wonder they’re going after the children…
https://vigilantcitizen.com/latestnews/zara-removes-suggestive-photoshoot-featuring-a-child-model
https://vigilantcitizen.com/latestnews/something-is-terribly-wrong-with-the-pepper-pics-instagram-account
https://vigilantcitizen.com/latestnews/something-is-terribly-wrong-with-the-childrens-clothing-line-caroline-bosmans
https://vigilantcitizen.com/latestnews/the-2018-met-gala-because-the-industry-loves-blasphemy
https://vigilantcitizen.com/latestnews/the-met-gala-2019-a-perfect-reflection-of-the-showbusiness-agenda
https://vigilantcitizen.com/latestnews/the-met-2021-gala-another-display-of-the-elites-insanity
https://vigilantcitizen.com/moviesandtv/the-meaning-of-netflixs-brand-new-cherry-flavor-a-celebration-of-occult-hollywood
And, of course, NetFlix:
https://vigilantcitizen.com/moviesandtv/squid-game-the-meaning-of-its-messages-and-symbolism
https://vigilantcitizen.com/moviesandtv/the-sick-twisted-messages-in-chilling-adventures-of-sabrina
I think China was grabbed by the elite during the Opium Wars. One day I may post the bits and pieces of evidence I have managed to find.
I think this is a big reason why we can’t come together against a common enemy … we are having trouble identifying the true core enemy. CCP? Cabal? DS? Are they just 3 heads of the Hydra?
I think the moment we put a face or name or identity to the core evil is the moment we all come together to defeat the evil.
I always call them The Hated Chicoms.
I think this makes the distinction clear.
Why assume that the enemy is ONE enemy?
Isn’t it possible that there are factions of bad guys that are fighting each other as they fight us (or at the very least maneuvering for position)?
That’s part of the beauty of calling them “mr global” … it’s non-specific enough to allow for an expanding definition.
Cyber Polygon, Dark Winter, Digital ID, And The Hunger Games Have More In Common Than You Might Think – The Burning Platform
More from NYC.
Rally today. Totally slipped my mind with stuff going on, not the least of which is shaving down the dog with only the Couch Commando around to hold her. She is NOT happy, and I’m not done yet. And he won’t put the darn phone down.
Okay, so do we want one? I can take a little time from some other projects to put one together.
I’ll be back for the answer.
Always.
Meanwhile they’re already lining up without us…Oh wait… this was the last one? Better you do this than me..
https://citizenfreepress.com/breaking/lines-already-forming-for-trump-maga-rally-in-des-moines-photos-video/
Okay, let me get through something real quick and I will put one up.
Yeah. Some old hippie asked of Joe Bye,Done:
“Hey, man, what’s your sign?”
“Vacant”…..
“Help Wanted.”
“Condemned.”
“Noxious Vapors.”
YES, PLEEZ!!!😍💖😍
Yes please
❤️
EXCLUSIVE: Experts BASH Big Tech for Reported Demonetization of So-Called ‘Climate Denial’ | Newsbusters
Here you go. I need to get some other stuff done before this commences. Later.
https://www.theqtree.com/2021/10/09/rally-thread-save-america-des-moines-iowa-october-9-2021/
New Political Moonshine article:
It All Starts With a Code: Part 2 – The ICDM-10 Coding Manual Mapped for COVID-19
October 9, 2021
Intro: “Part one of this sub-series of articles was written for the informed reader and veteran Moonshiner to delineate evidentiary slides relative to established work and positions on COVID-19. It’s purpose was to establish the ‘ICD-10-CM Official Guidelines for Coding and Reporting FY 2021 – UPDATED January 1, 2021’ as a primary data driver for the enterprise fraud construct.
Part two is no different. Part one contains 41 evidentiary slides respective to comprehending COVID-19 as a construct of enterprise fraud and it is required reading to make sense of the evidentiary exhibits that follow here. A longer but shallow dive connecting to primary, secondary and so on sources can be found in our timeline if further contextual backdrop is needed for comprehension.
This exhibit builds on firmly established, existing and exhibited evidence of COVID-19 being an enterprise fraud construct leveraging a closed-loop system to propagate harvested co-morbidity data as COVID-19 data. The RT-PCR test calibrated to faulty high cycle thresholds and the NVSS diagnostic memos serve as established primary data drivers within the construct and the ICD-10-CM lane is established through the NVSS.
The following exhibits depict the ICD-10-CM as a data driving lane established by the WHO/Tedros [directly] and China [indirectly via Tedros (installed by secret electronic vote in 2017)] to impact internal U.S. COVID-19 data and thus effectuate control over the direction of the nation and its policies respective to the pandemic.”
[continued at title link above]
I wonder what would happen if a blm supporting ‘protest group’ did some midnight shopping/protesting in a grocery store.
Would they arrest or turn away like summer 2020?
IDK… with no white communists to lead them, will they even come out in numbers? Looking more and more like BLM was not a grass roots movement, but media driven instead. Okay, always looked that way from here, but it should be obvious to more people now.
It’s being noticed that there really aren’t any orders. An announcement was made and a whole lot of people assumed it was real.
It isn’t.
Businesses are being good little sheep.
Businesses with injection mandates are following the RUMOR. NOT following the law, regulation or anything else.
IF fake news was doing their job, they’d report there are no injection mandate laws, regulations…ONLY rumors.
They all should sue specially if business is lying.
That’s the problem. There is no regulation to sue over.
To bad. People need to be informed.
“There is no regulation to sue over.”
Doesn’t matter, a worker can SUE if he is fired or forced to quit BECAUSE HE CAN NOT BE FORCED TO TAKE AN EXPERIMENTAL DRUG AS AN EMPLOYMENT REQUIREMENT!!!
Plague of Liars: Nuremberg Code Outlaws Forced Medical Procedures, Which INCLUDES Mandatory Vaccinations
Let me know when someone wins a case
That’s the problem, it’s going to take a while.
Rumors and media statements by the pResident of the senior living center.
“IF fake news was doing their job,”
_____________
Fake news *is* doing their job, it’s just not the job we think it is.
We think (because we’re conditioned) their job is to investigate and report news and hold public figures accountable.
But their real job is to cover up and propagandize and make sure public figures are never held accountable.
People are tricked and its a scam.
As far as I know that’s businesses only.
Federal employees and contractor employees are in fact covered by an EO (albeit an illegal one).
https://twitter.com/GallopingGaye/status/1446458107102736388?s=20
Awesome. Nurse dressed in operating room scrubs begins to sing as she devests herself of her glasses, and then all her medical gear, (two masks, cap, stethoscope, thermometer, ID badge, dropping each on the ground as she cheerfully sings her song. Finishes with loosening her long brown hair, then shaking that out, all while the text boxes tell her story of doing 4yrs of health science, 2 years accelerated graduate school for nursing, Then 19 months as an ICU nurse in patient and COVID wards caring for grandparents, wives, husbands, daughters and sons all while reusing hospital protection gear spending holidays, weekends, sleep time taking care of others while away from her own family, only to be on leave 1 Oct and to be terminated come 1 November because she won’t take the jab.
Won my internets award for fall season for sure.
para, Thanks for finding this!
I agree, a prize winner!!
WOW.
Beautiful.
And Beautiful.
And sad.
And unfair.
And IT HAS TO STOP!!!!!!!!!!!!!!!
It will stop when med schools get sued for not warning about it before they take your tuition money.
“And IT HAS TO STOP!!!!!!!!!!!!!!!”
____________
But it doesn’t have to stop… in fact, why would it ever stop, unless someone stops it?
People used to understand this better. It was probably most obvious in the old Western movies.
Bad guys would roll into town, take it over, do as they pleased, rape, murder, whatever they wanted.
They liked it.
They weren’t ever going to stop.
Until some Sheriff came to town, or a whole posse, or a bounty hunter, and confronted the bad guys, and killed them all.
That’s the basic outline of probably 90% of all the Westerns ever made… 😁
It didn’t take a lot of imagination to write the script, because there are only two ways the situation could be resolved, either the bad guys win, or the good guys win.
And it never once happened without the good guys confronting the bad guys.
Because if the good guys never confront the bad guys, the bad guys will never stop.
The fact that the bad guys MUST be confronted (and stopped!) is the premise for the whole movie… because if no one ever confronts the bad guys, then you just have approximately two hours of misery and injustice, like we’re all experiencing every day.
And who wants to pay money to go see that?
🤣 😂 🤣
Luthien standing before Morgoth.
Wow, the China-bots have flooded the comments below that video, just one hateful remark after another, to make sure the zombies don’t wander off the reservation.
Standing by for indictments, arrests, court trials….
Not holding my breath.
We are eleven plus months after the stolen election. No indictments. No arrests.
Hopeful, albeit with some jaded skepticism.
Durham will die of old age, the day before he was going to make his first big indictment 👍
So we have that goin’ for us… 😂 🤣 😂
I like the tone on that one!
because FJB and the traitors he rode in on
Yep.
One might say FJB and the whores he rode in on…
(or with)…
Yep.
RECEIPTS: DoD Joint Artificial Intelligence Center Monitoring Vaxx Deaths!Karen Kingston has done it, AGAIN. Kingston is a biotech analyst who joined Stew Peters with RECEIPTS, proving that the theory of Cyrus Parsa is NOT a “crazy conspiracy theory”, but the DoD is actually using their Joint AI Intel Center to monitor “vaccine” deaths! Why?
https://rumble.com/vnhqap-receipts-dod-joint-artificial-intelligence-center-monitoring-vaxx-deaths.html
I hope for Nuremberg and military tribunals. But I won’t hold my breath.
FWIW, Austria’s Premier, Sebastian Kurz, has resigned due to a corruption scandal. He is/was one of the WER’s rising young stars, so perhaps they have “special training” in nefarious deeds, and he missed a few bits 🙂
He will be replaced by Außenminister (Secretary of State) Alexander Schallenberg , a fellow member of the ÖVP. This is just breaking, and I’m hitting the hay, but it may prove to be interesting. Kurz was pretty conservative, but controlled by the likes of Sauros, et. al… Controlled opposition?
The fact that he was pretty conservative as you say is likely the only reason he stepped aside. If he was a Dem from here he’d just ignore anything having to do with ethics or criminality.
Jacksonville, FL – Pilots walk out over vaccine mandate….
Happy for the airlines misery…
Stop the injection demands.
“Is there a media blackout on last night’s air traffic controller walkout in Jacksonville, FL?”
______________
Watch, they’ll hire illegal aliens to take their places.
What could go wrong?
👍😂🤣😂
Hey, I thought Ronnie D. signed a law or an executive order preventing mandates in Florida, didn’t he?
And if not, why not?
Air Traffic Controllers are federal employees, so they’re subject to the actual executive order applying to federal employees.
“Air Traffic Controllers are federal employees, so they’re subject to the actual executive order applying to federal employees.”
_____________
Has anyone actually found the executive order?
I keep seeing articles claiming that nobody can find any such mandate on the OSHA website or anywhere else.
But OSHA would implement the OTHER order, the one directed at private business.
There are two orders (or pseudo orders) in question here, one directed to federal employees and contractors, the other at private industry.
What we famously cannot find is the one directed at private industry, and that fact (that we can’t find that order) is not relevant here, because ATC is not private industry.
I understand now, thank you! 👍
GA/FL…oughta be fun this Holiday Season. By that time, who knows what staffing will be like.
Not to mention people just not wanting to fly w such odious restrictions.
Airlines gonna lose big bucks.
Study suggests Delta variant is being spread FROM VACCINATED healthcare workers TO VACCINATED healthcare workers.
If so, what is the purpose of the vaccine mandates? – Rep. Thomas Massie –
Five of the most vaccinated countries now have the highest rates of new infections – Singapore, Seychelles, Bermuda, Gibralter, Israel
Had a patient frothing at the mouth about the unvaccinated. He came to hospital with a broken arm and got covid in ER. Wanted all unvaccinated locked up. I asked him if he was double dosed and he said YES! Told him it was the vaccines job to stop him getting covid and that there were plenty of people who couldn’t/ shouldnt or don’t want to get vaccinated. He said that then they shouldn’t get health care. I said really? So fat porkers don’t get stents when they get heart disease? And smokers treatment for lung cancer? What about people who are carriers of a disease who have kids-do the kids get kicked to the curb if they inherit the disease?
put him back in his box
Good on ya!!!!!
It’s stunning how people lose all sense of logic and reason with regard to the clot shot. The damned Big Pharma Industry and the politicians have redefined what a vaccine is, and how it’s supposed to work.
Never mind the fact that the clot shot is NOT a vaccine, rather it’s a means of gene modification and replacement “therapy”.
Have people really forgotten how to think?????!!!!!! 😮
Sometimes I think this madness is “the delusion at the end of the age”…. Sure seems like it…..
P.S. Hang in there. Pray everything’s OK for you and yours…
Say what? Is there any sauce on this. It doesn’t sound like something Pompeo would say.
I didn’t see any. But Jack has a pretty reliable advanced scoop record. Guess we gotta wait and see.
Besides, I like Kansas and all, but it ain’t ever gonna happen! Anyone who dares run against Trump is a stupid fool!!
Why is Kansas in the same organization Pence is in and Kelly Ann Conway ?
I know there are some other questionable people on there but forgot their names. Would I trust Kansas 100% No. For me his earning my trust is still out.
Jack why would jack lie and put out misinformation? Would he do it on purpose I do not think so. Is someone misleading him maybe. He is smart who knows the game lintel play.
We have put our bet on many eople I am not counting them up and it was misplaced trust in them.
Kansas has been great so far. Supportive of 45, always put out 45’s message and not his own, tough on our enemies, pro MAGA policies, Godly man, doesn’t suffer fools. BUT….. I have a nagging spidey sense about him. 1) He’s too friendly with too many RINOs, which may or may not be ok.🤨 AND.. I’m not happy with 2) his tenure at CIA, appointing Traitor Haspel after her role in Crossfire Hurricane, AND 3) He didn’t drain any of the State Dept cesspool. They were allowed to continue nasty swamp bizness as usual and they still haven’t stopped. 🤬🤬
Much to contemplate on. I see some warning signals could be just me have not figured it out.
Link or it didn’t happen.
NOT calling you out PR. Calling Jack out.
I know…
There have been rumors about Jack…
I can’t find anything about his comment anywhere else…
perhaps Jack is the problem here?
Yikes..the replies are disturbing. If he did..is it a Rino ploy to split the base ?
But just look at how he puts that. How can you primary someone who isn’t currently in office??? And if Trump was, it would be his last term. So how do you primary him? Pompeo wouldn’t say that.
It doesn’t sound like something Pompeo would say but it sure sounds like something the feckless traitorous GOP would say. I think they know Pence is dead in the water . They also know Pompeo is well liked.
Wonder if we unravel the mystery? Somehow the truth will come out .
Clearly, “primary” here would mean to run against Trump for the nomination, with the intention of beating him. The bit about the target being the incumbent is secondary in this context.
Bingo 🙂
no way the base would split…
there is NO 22 or 24 election unless Trump returns and soon…
Jack emphasized “really”
” Is Pampeo really telling people he might primary Trump?”
All reporters rely on sources and all reporters are eager to put out new information. This source likely needs more scrutiny, but that’s between him and his source. I’d be willing to bet Jack used this for a throw away even knowing he was being baited with a ridiculous notion. Even in Jacks mind this is stupid and he’s not afraid to put it out just to watch it catch flame and be turned to cinder.
Meanwhile for us though just looking at Kansas we all know he’d not be able to handle the rigors of the Presidency and we have been lucky he did not expire from a stroke while serving the best President ever. Now if Kansas suddenly drops fifty pounds we can take it serious but even then it would be laughable.
I heard he had lost weight Para…
I’ve heard rumors Poso is sea eye eh …
‘course this a.m. the claim is lots of sources
and I agree it is laughable… but they all, GOP included live in a bubble of corruption
U.S. Military Academy and Harvard Law grad.